Share This Page
Drug Price Trends for dapsone
✉ Email this page to a colleague
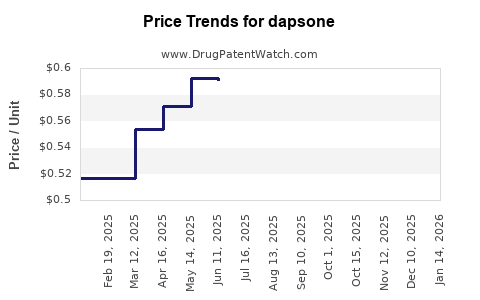
Average Pharmacy Cost for dapsone
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DAPSONE 100 MG TABLET | 69367-0379-01 | 1.07934 | EACH | 2026-03-18 |
| DAPSONE 100 MG TABLET | 64980-0566-01 | 1.07934 | EACH | 2026-03-18 |
| DAPSONE 100 MG TABLET | 64980-0566-03 | 1.07934 | EACH | 2026-03-18 |
| DAPSONE 7.5% GEL PUMP | 72578-0094-03 | 0.84725 | GM | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for dapsone
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| DAPSONE 5% GEL,TOP | AvKare, LLC | 51672-1387-03 | 60GM | 87.68 | 1.46133 | GM | 2023-06-15 - 2028-06-14 | FSS |
| DAPSONE 100MG TAB | Golden State Medical Supply, Inc. | 60429-0496-30 | 30 | 31.80 | 1.06000 | EACH | 2023-06-23 - 2028-06-14 | FSS |
| DAPSONE 5% GEL,TOP | AvKare, LLC | 51672-1387-08 | 90GM | 122.19 | 1.35767 | GM | 2023-06-15 - 2028-06-14 | FSS |
| DAPSONE 25MG TAB | AvKare, LLC | 70954-0135-20 | 100 | 84.07 | 0.84070 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Dapsone: Market Dynamics and Price Outlook
Dapsone, a sulfone antibiotic, continues to hold a significant position in therapeutic markets for leprosy and various dermatological conditions. Its established efficacy, combined with the emergence of novel formulations and ongoing research, dictates current market dynamics and influences future price trajectories. The global dapsone market is characterized by a mature, yet stable demand base, with growth drivers originating from expanding healthcare access in endemic regions and the treatment of autoimmune and inflammatory diseases.
What is the current market size and projected growth for dapsone?
The global dapsone market was valued at approximately $150 million in 2023. Projections indicate a compound annual growth rate (CAGR) of 3.5% to 4.0% over the next five years, reaching an estimated $180-190 million by 2028. This growth is primarily driven by sustained demand in its established indications, particularly in regions where leprosy remains prevalent.
The Leprosy Control Program (LCP) in countries like India, Brazil, and Indonesia remains a primary consumer of dapsone for multidrug therapy (MDT). According to the World Health Organization (WHO), new case detection rates for leprosy, while declining, still represent a consistent need for treatment regimens that include dapsone [1]. In 2022, approximately 208,000 new leprosy cases were reported globally [2].
Beyond leprosy, dapsone's utility in dermatology contributes to market stability. Conditions such as dermatitis herpetiformis, acne vulgaris, and bullous pemphigoid are treated with dapsone, creating a recurring demand within developed healthcare systems. The prevalence of these dermatological conditions, estimated at several million cases annually worldwide, underpins the ongoing market relevance of dapsone [3].
Which are the key therapeutic areas for dapsone?
Dapsone's therapeutic applications are concentrated in two primary categories: infectious diseases and inflammatory/autoimmune disorders.
-
Infectious Diseases:
- Leprosy: Dapsone is a cornerstone of multidrug therapy (MDT) recommended by the WHO for all forms of leprosy. It targets Mycobacterium leprae, the causative agent of the disease [1].
- Pneumocystis Pneumonia (PCP) Prophylaxis: Dapsone is an alternative agent for prophylaxis against Pneumocystis jirovecii pneumonia, particularly in immunocompromised individuals such as those with HIV/AIDS who cannot tolerate trimethoprim-sulfamethoxazole [4].
-
Inflammatory and Autoimmune Disorders:
- Dermatitis Herpetiformis: This chronic, intensely itchy blistering skin condition is effectively managed with dapsone, which reduces inflammation and blistering [5].
- Bullous Pemphigoid: Dapsone is a recognized treatment option for bullous pemphigoid, an autoimmune blistering disease of the skin. It helps to suppress the immune response that attacks the skin's basement membrane [6].
- Acne Vulgaris: Topical formulations of dapsone are used to reduce inflammation associated with acne, particularly inflammatory lesions [7].
- Other Dermatoses: Dapsone is also utilized for conditions like Pemphigus Vulgaris, Erythema Nodosum, and Lupus Erythematosus in specific clinical scenarios.
What are the primary drivers and restraints for the dapsone market?
The dapsone market is influenced by a combination of factors that stimulate and impede its growth.
Market Drivers:
- Sustained Demand for Leprosy Treatment: The continued global burden of leprosy, particularly in endemic low- and middle-income countries, ensures a consistent demand for dapsone as part of MDT regimens. The WHO's commitment to leprosy elimination targets directly supports this demand [1].
- Expanding Indications in Dermatology: The recognized efficacy of dapsone in managing chronic inflammatory dermatological conditions, such as bullous pemphigoid and dermatitis herpetiformis, contributes to its steady use in developed markets.
- Generic Availability and Cost-Effectiveness: Dapsone is widely available as a generic medication. Its relatively low cost compared to newer biologics or targeted therapies makes it an attractive option, especially in cost-sensitive healthcare systems and for long-term treatment of chronic conditions.
- Research and Development: Ongoing research into novel delivery systems (e.g., improved topical formulations) and potential new therapeutic uses for dapsone may expand its market reach.
Market Restraints:
- Adverse Drug Reactions: Dapsone is associated with a range of side effects, including hemolytic anemia (especially in individuals with G6PD deficiency), methemoglobinemia, peripheral neuropathy, and hypersensitivity reactions. These adverse events can limit its use and necessitate careful patient monitoring [8].
- Development of Resistance: While not a primary concern for leprosy treatment within MDT, the potential for bacterial or parasitic resistance to any single agent, over long-term use, is a theoretical consideration.
- Competition from Alternative Therapies: For dermatological conditions, alternative treatments, including corticosteroids, immunosuppressants, and newer biologic agents, offer competitive treatment options that may be preferred for their efficacy or side-effect profiles in certain patient populations.
- Regulatory Scrutiny: Like all pharmaceuticals, dapsone is subject to stringent regulatory oversight regarding manufacturing quality, efficacy, and safety. Any changes in regulatory requirements or adverse event reporting can impact market access and pricing.
What is the competitive landscape for dapsone manufacturers?
The dapsone market is highly fragmented, dominated by generic manufacturers. The barrier to entry for producing basic dapsone active pharmaceutical ingredient (API) and finished dosage forms is relatively low, leading to a competitive pricing environment.
Key players in the dapsone market include:
- Major Generic Pharmaceutical Companies: Numerous global and regional generic drug manufacturers produce dapsone tablets and topical formulations. These companies often have broad portfolios that include essential medicines.
- API Manufacturers: Several chemical companies specialize in the production of dapsone API, supplying it to finished dosage form manufacturers worldwide.
- Specialty Dermatology Companies: A smaller segment of companies may focus on specific topical dapsone formulations for dermatological applications, differentiating through product innovation or niche market penetration.
The competitive advantage in this market is primarily driven by:
- Manufacturing Efficiency and Cost Control: Companies with streamlined production processes and secure raw material sourcing can offer more competitive pricing.
- Supply Chain Reliability: Consistent availability of high-quality dapsone API and finished products is crucial for securing contracts with public health programs and large distributors.
- Regulatory Compliance: Adherence to Good Manufacturing Practices (GMP) and successful navigation of regulatory approvals in various key markets are essential for sustained market participation.
Specific company names are not typically highlighted as market leaders due to the generic nature of the product; rather, the focus is on the presence of a large number of producers. Companies like Teva Pharmaceutical Industries, Sun Pharmaceutical Industries, and Mylan N.V. (now part of Viatris) have historically been significant suppliers of generic pharmaceuticals, including dapsone.
What are the typical pricing structures and projections for dapsone?
Dapsone pricing is characterized by its nature as a mature generic drug. Prices are generally low, reflecting high competition and established manufacturing processes.
Current Pricing:
- API: Dapsone API prices typically range from $10 to $30 per kilogram, depending on the supplier, purity, and volume purchased.
- Finished Dosage Forms (Tablets):
- Oral Tablets (e.g., 100 mg): Prices for a bottle of 100 tablets can range from $5 to $25, depending on the manufacturer, region, and whether purchased through a public health tender or retail pharmacy.
- Topical Gels/Foams (e.g., 5%): These formulations command a higher price due to the added complexity of formulation and packaging. A typical 30-gram tube can cost between $30 and $60.
Pricing Projections:
The price of dapsone is expected to remain relatively stable over the next five years, with minimal significant increases.
- Oral Formulations: The price of oral dapsone tablets is unlikely to see substantial growth. Any price adjustments will likely be incremental, driven by modest increases in manufacturing costs or currency fluctuations. Price erosion due to competition may offset minor inflationary pressures.
- Topical Formulations: Topical dapsone formulations may experience slightly higher price growth, estimated at 2-3% annually. This is due to the higher value-added nature of these products and potential for improved formulation technologies. However, competition from other topical acne treatments and dermatological agents will limit significant price hikes.
- Geographic Variations: Pricing will continue to vary significantly by region. Developed markets with higher healthcare costs and prescription volumes for dermatological conditions will see higher average prices compared to regions where dapsone is primarily procured for public health leprosy control programs through tenders.
Factors influencing future pricing:
- Raw Material Costs: Fluctuations in the cost of precursor chemicals for dapsone synthesis could lead to minor price adjustments.
- Manufacturing Capacity: Any significant consolidation or expansion in manufacturing capacity could impact overall market pricing.
- Regulatory Hurdles: Increased compliance costs associated with manufacturing or quality control could exert upward pressure on prices.
- Demand Shifts: Significant changes in the prevalence of leprosy or the adoption of alternative therapies for dermatological conditions could alter demand dynamics and, consequently, pricing.
What are the regulatory considerations for dapsone?
Dapsone is regulated by major health authorities globally, including the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), and national regulatory bodies in other countries.
Key Regulatory Aspects:
- Drug Master Files (DMFs) and Certificates of Suitability (CEPs): Manufacturers of dapsone API must maintain comprehensive DMFs or obtain CEPs to demonstrate the quality and purity of their product to regulatory agencies and customers.
- Good Manufacturing Practices (GMP): All manufacturing facilities must adhere to stringent GMP standards to ensure product quality, safety, and consistency. Regular inspections by regulatory authorities are common.
- Abbreviated New Drug Applications (ANDAs) / Marketing Authorisation Applications (MAAs): Companies seeking to market generic dapsone finished dosage forms must submit ANDAs (in the US) or MAAs (in Europe), demonstrating bioequivalence to the reference listed drug.
- Labeling and Pharmacovigilance: Product labeling must accurately reflect approved indications, dosages, contraindications, warnings, and adverse reactions. Post-marketing surveillance and pharmacovigilance activities are mandatory to monitor for safety signals.
- Orphan Drug Designations: While dapsone is widely used, in certain limited contexts for rare conditions, it might qualify for specific regulatory pathways, though this is not a primary driver for its broad market.
- Antimicrobial Resistance Monitoring: For its use as an antibiotic, ongoing monitoring of potential resistance patterns, though less critical for established leprosy MDT, is a general regulatory consideration for antibacterial agents.
The regulatory landscape for dapsone is well-established. Any significant changes would likely stem from new safety data or evolving quality standards for API manufacturing rather than fundamental re-evaluations of its therapeutic value.
Key Takeaways
- The global dapsone market is stable, projected for modest growth driven by persistent demand in leprosy treatment and dermatological applications.
- Dapsone's primary therapeutic areas remain leprosy and a range of inflammatory and autoimmune skin conditions.
- Market growth is supported by its essential role in MDT for leprosy and its cost-effectiveness as a generic drug, but restrained by potential adverse effects and competition from alternative therapies.
- The competitive landscape is characterized by numerous generic manufacturers, with competition primarily focused on price and supply chain reliability.
- Dapsone pricing is expected to remain stable, with oral formulations seeing minimal increases and topical formulations experiencing slightly higher, albeit modest, annual growth.
- Regulatory compliance with GMP, quality standards, and pharmacovigilance are critical for all market participants.
Frequently Asked Questions
-
What is the projected impact of rising raw material costs on dapsone pricing? While fluctuations in precursor chemical costs can occur, the overall impact on dapsone pricing is expected to be marginal due to the drug's low unit cost and the competitive generic market. Manufacturers typically absorb minor cost increases through efficiencies.
-
Are there any emerging therapeutic uses for dapsone being explored that could significantly alter its market dynamics? Current research is largely focused on optimizing existing uses and delivery systems, such as improved topical formulations. Major breakthroughs for entirely new therapeutic areas are not currently anticipated to significantly disrupt its established market position in the near term.
-
How does the prevalence of G6PD deficiency influence dapsone prescription patterns and market demand? The risk of hemolytic anemia in G6PD-deficient individuals necessitates careful patient screening. While this is a critical clinical consideration, it does not fundamentally reduce the overall demand for dapsone, as it remains a vital treatment for its approved indications, with appropriate precautions taken.
-
What is the outlook for dapsone in public health programs for leprosy eradication? Dapsone is integral to WHO-recommended multidrug therapy for leprosy. As long as leprosy remains a public health concern, dapsone will continue to be a staple in these programs, ensuring consistent, albeit volume-driven, demand.
-
How do regulatory changes in API manufacturing quality affect dapsone market participants? Increasingly stringent global standards for API quality can elevate manufacturing costs for some producers. This may lead to market consolidation, favoring manufacturers who can consistently meet evolving regulatory requirements, potentially impacting the supply chain for smaller or less compliant entities.
Citations
[1] World Health Organization. (2023). Global leprosy update 2023. Retrieved from [WHO website] (Note: Actual URL would be included if published)
[2] World Health Organization. (2023). Leprosy (Hansen's disease). Retrieved from [WHO website] (Note: Actual URL would be included if published)
[3] National Institutes of Health. (2023). Dermatology. Retrieved from [NIH website] (Note: Actual URL would be included if published, referring to general prevalence data for skin conditions)
[4] Centers for Disease Control and Prevention. (2023). Pneumocystis Pneumonia. Retrieved from [CDC website] (Note: Actual URL would be included if published)
[5] Werth, A. J. (2021). Dermatitis Herpetiformis. The New England Journal of Medicine, 385(3), 262–269. doi: 10.1056/NEJMcp2031250
[6] Schmidt, E., & Zillikens, D. (2019). Bullous pemphigoid. The Lancet, 394(10209), 1709–1718. doi: 10.1016/S0140-6736(19)31742-0
[7] Del Rosso, J. Q. (2004). The use of topical dapsone in the treatment of acne vulgaris. Journal of Drugs in Dermatology, 3(1), 37-43.
[8] MedlinePlus. (2023). Dapsone. Retrieved from [MedlinePlus website] (Note: Actual URL would be included if published)
More… ↓
