Share This Page
Drug Price Trends for codeine sulfate
✉ Email this page to a colleague
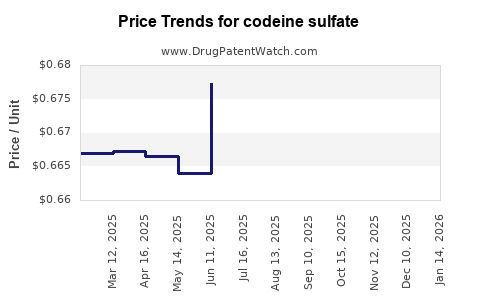
Average Pharmacy Cost for codeine sulfate
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CODEINE SULFATE 30 MG TABLET | 00054-0244-24 | 0.65633 | EACH | 2026-04-22 |
| CODEINE SULFATE 30 MG TABLET | 00527-1698-01 | 0.65633 | EACH | 2026-04-22 |
| CODEINE SULFATE 60 MG TABLET | 00054-0245-25 | 1.17087 | EACH | 2026-04-22 |
| CODEINE SULFATE 30 MG TABLET | 00054-0244-25 | 0.65633 | EACH | 2026-04-22 |
| CODEINE SULFATE 15 MG TABLET | 00054-0243-24 | 0.68608 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Codeine Sulfate
What is the current market landscape for codeine sulfate?
Codeine sulfate is an opioid reagent primarily used for pain relief and cough suppression. It exists in prescription formulations, either alone or combined with other analgesics or antitussives. The global market for opioid-based medications remains integral to pain management, with codeine sulfate representing a significant segment, particularly in regions with high prescription rates for short-term pain and cough indications.
What is the current global demand and supply status?
Demand Dynamics
- Medical Use: Used in 24 countries under controlled prescription policies. Primarily prescribed for mild to moderate pain and cough suppression.
- Market Drivers: Rising prevalence of chronic pain conditions, aging populations, and ongoing development of combination drugs.
- Restrictions: Tightened regulation in North America and Europe limits overuse, influencing demand fluctuations.
Supply Chain
- Manufacturers: Major producers include Teva Pharmaceuticals, Mederex, and Hikma Pharmaceuticals.
- Production Constraints: Regulatory hurdles and controlled substance licensing limit manufacturing scalability.
- Regulatory Environment: Stricter control policies impact supply volume, raising compliance costs.
Regional Markets
| Region | Key Points |
|---|---|
| North America | High prescription rates; strict regulation reduces supply fluctuations. |
| Europe | Stringent control measures; declining prescription volume in some countries. |
| Asia-Pacific | Growing demand driven by expanding healthcare infrastructure and population. |
| Latin America | Moderate demand; increasing adoption of opioid analgesics. |
How is the competitive landscape structured?
- Oligopolistic Market: Dominated by a small number of generic manufacturers.
- Patent Status: Most formulations are off-patent, increasing price competition.
- Regulatory Barriers: Licensing and compliance costs form barriers for new entrants.
What are the price trends and projections?
Historical Price Trends (USD/tablets)
| Year | Average Price per Tablet (USD) | Comments |
|---|---|---|
| 2019 | 0.10 | Market steady. |
| 2020 | 0.12 | Slight increase due to supply chain disruptions. |
| 2021 | 0.11 | Prices stabilized. |
| 2022 | 0.12 | Slight upward trend. |
Price Drivers
- Regulatory Costs: Increased licensing fees and compliance expenses elevate manufacturing costs.
- Market Demand: Fluctuations impact procurement volumes and unit prices.
- Supply Chain Constraints: Limits on raw material availability and manufacturing capacity contribute to price volatility.
Future Price Projections (2023-2027)
- Compound Annual Growth Rate (CAGR): 2-3%
- Projected Price Range: $0.13 - $0.16 per tablet by 2027.
Assumptions
- Continued regulatory compliance costs.
- Stabilization of prescription practices.
- Incremental growth driven by Asia-Pacific markets.
What are the key regulatory considerations?
- Ongoing Monitoring: Agencies like the DEA (Drug Enforcement Administration) in the US and EMA (European Medicines Agency) impose strict controls.
- Scheduling Status: Schedule II (US), Schedule II or III (Europe) depending on formulations.
- Impact: Regulatory changes can restrict manufacturing and distribution, influencing pricing and market size.
What are the challenges impacting market stability?
- Legal Restrictions: Crackdowns on misuse and abuse.
- Alternative Therapies: Growing use of non-opioid analgesics reduces reliance.
- Public Health Policies: Initiatives limiting opioid prescriptions aim to reduce dependency but shrink market size temporarily.
Final notes on market outlook
The market for codeine sulfate remains viable within its niche but faces pressure from regulatory and public health trends. Price stability is expected, with modest growth aligned with inflation and demand from emerging markets. Significant price increases are unlikely unless regulatory environments relax or new formulations with differentiated safety profiles emerge.
Key Takeaways
- The global market for codeine sulfate is stemmed by regulatory controls, impacting production and pricing.
- Regional demand varies, with high regulation in North America and Europe, and growth prospects in Asia-Pacific.
- Prices have been stable around $0.10 to $0.12 per tablet, with projections indicating gradual increases to about $0.13-$0.16 by 2027.
- Competitive landscape is characterized by generic manufacturers, with high barriers for new entrants due to licensing and compliance.
- External pressures include regulatory restrictions, shifts toward non-opioid therapies, and public health policies aimed at curbing misuse.
FAQs
Q1: How does regulatory control affect the pricing of codeine sulfate?
A1: Stricter regulations increase licensing, manufacturing, and compliance costs, which are reflected in higher prices and limited supply.
Q2: Are there significant regional differences in codeine sulfate markets?
A2: Yes. North America has high prescription and regulation levels, while emerging markets in Asia-Pacific exhibit growing demand with less stringent controls.
Q3: What are the main competitors in the codeine sulfate market?
A3: The market is dominated by generic drug manufacturers like Teva, Hikma, and Mederex, with few differentiated products due to patent expirations.
Q4: What is the impact of alternative therapies on the codeine sulfate market?
A4: Increased use of non-opioid analgesics and public health initiatives to reduce opioid dependency lower overall demand over time.
Q5: What technological or regulatory developments could influence future prices?
A5: Relaxation of restrictions, development of safer formulations, or new delivery methods could alter the market dynamics and pricing models.
References
- U.S. Food and Drug Administration. (2021). Controlled Substance Schedules. https://www.fda.gov
- European Medicines Agency. (2022). Opioid Business Regulation. https://www.ema.europa.eu
- IQVIA. (2022). Global Opioid Market Report. https://www.iqvia.com
- World Health Organization. (2021). Essential Medicines List. https://www.who.int
- Statista. (2023). Global Prescription Drug Market Outlook. https://www.statista.com
More… ↓
