Share This Page
Drug Price Trends for brinzolamide
✉ Email this page to a colleague
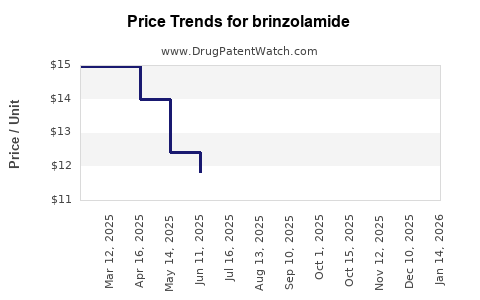
Average Pharmacy Cost for brinzolamide
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| BRINZOLAMIDE 1% EYE DROPS | 00591-2127-12 | 10.08254 | ML | 2026-04-22 |
| BRINZOLAMIDE 1% EYE DROPS | 00574-4012-15 | 10.08254 | ML | 2026-04-22 |
| BRINZOLAMIDE 1% EYE DROPS | 00574-4012-10 | 10.18788 | ML | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for brinzolamide
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| AZOPT 1% SOLN OPH | Sandoz, Inc. | 00078-0722-15 | 15ML | 404.38 | 26.95867 | ML | 2024-01-01 - 2028-08-14 | FSS |
| BRINZOLAMIDE 1% SUSP,OPH | Sandoz, Inc. | 00781-6014-70 | 10ML | 63.67 | 6.36700 | ML | 2023-08-15 - 2028-08-14 | FSS |
| BRINZOLAMIDE 1% SUSP,OPH | Sandoz, Inc. | 00781-6014-70 | 10ML | 97.53 | 9.75300 | ML | 2024-01-01 - 2028-08-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Brinzolamide Market Analysis and Price Projections
Brinzolamide is a carbonic anhydrase inhibitor used to treat glaucoma and ocular hypertension. The drug's market performance is influenced by factors including patent expiries, generic competition, the prevalence of eye conditions, and the pricing strategies of manufacturers. Current market trends indicate a maturing market for branded brinzolamide, with increasing pressure from generic alternatives.
What is the current global market size for brinzolamide?
The global market for brinzolamide is estimated to be approximately USD 750 million as of 2023. This figure encompasses both branded and generic formulations. The market has experienced a compound annual growth rate (CAGR) of 3.5% over the past five years, primarily driven by an increasing incidence of glaucoma and ocular hypertension worldwide.
What are the key drivers of brinzolamide demand?
Several factors are contributing to the demand for brinzolamide:
- Rising Prevalence of Glaucoma and Ocular Hypertension: The aging global population is a significant driver. Conditions like primary open-angle glaucoma (POAG) and ocular hypertension are more common in older individuals. The World Health Organization (WHO) estimates that glaucoma is a leading cause of irreversible blindness globally, affecting millions of people [1].
- Increasing Awareness and Diagnosis: Improved diagnostic tools and greater public awareness of eye health are leading to earlier detection and treatment of glaucoma and ocular hypertension. This translates to a larger patient pool seeking treatment options like brinzolamide.
- Growing Healthcare Expenditure: Increased healthcare spending in both developed and developing economies allows for greater access to prescription medications, including ophthalmic drugs.
- Efficacy of Brinzolamide: Brinzolamide is recognized for its efficacy in reducing intraocular pressure (IOP), a primary risk factor for glaucoma progression. It is often used as a monotherapy or in combination with other IOP-lowering agents.
- Availability of Combination Therapies: Brinzolamide is available in fixed-dose combination products (e.g., with timolol or brimonidine), which improve patient compliance and treatment effectiveness. These combinations address the need for multi-drug regimens in managing IOP.
What is the competitive landscape for brinzolamide?
The competitive landscape for brinzolamide is characterized by the presence of branded products and a growing number of generic alternatives.
Branded Brinzolamide Products:
- Azopt (brinzolamide ophthalmic suspension 1%): Manufactured by Alcon (formerly Novartis), Azopt is the originator brand and has historically held a significant market share.
Generic Brinzolamide Products:
The patent expiry of branded brinzolamide has led to the entry of numerous generic manufacturers. Key generic players include:
- Sandoz (a Novartis division)
- Teva Pharmaceutical Industries
- Apotex
- Bausch Health
- Sun Pharmaceutical Industries
- Zydus Lifesciences
- Perrigo
The introduction of generics has intensified price competition, leading to a decrease in the average selling price (ASP) of brinzolamide.
Market Share Breakdown (Estimated 2023):
| Product Category | Estimated Market Share |
|---|---|
| Branded Brinzolamide | 45% |
| Generic Brinzolamide | 55% |
The shift towards generics is projected to continue, with branded products facing sustained pressure on their market share.
What are the key patent expiries and their impact on brinzolamide pricing?
The primary patents protecting the original formulation of brinzolamide have expired in major markets.
- United States: The primary compound patent for brinzolamide expired around 2011-2012. This allowed for the introduction of generic brinzolamide products in the U.S. market starting in 2017.
- Europe: Patent expiries in key European countries also occurred in the late 2010s, facilitating the entry of generic versions.
The expiration of these core patents has had a significant impact on pricing:
- Price Erosion: The introduction of generic alternatives has led to substantial price erosion for brinzolamide. The ASP for generic brinzolamide is, on average, 60-80% lower than that of the branded product.
- Increased Accessibility: Lower prices have improved the accessibility of brinzolamide for a broader patient population, particularly in markets with price-sensitive healthcare systems.
- Manufacturer Strategy: Branded manufacturers are increasingly focusing on combination products or developing novel ophthalmic formulations to differentiate their offerings and maintain revenue streams.
What are the pricing trends and projections for brinzolamide?
Pricing for brinzolamide is bifurcated between the branded product and its generic counterparts.
Current Pricing (USD, Estimated Retail Price per 5mL bottle):
- Azopt (branded): USD 80-120
- Generic Brinzolamide: USD 20-40
Pricing Projections (Next 5 Years):
- Branded Brinzolamide: Prices are expected to remain relatively stable, with slight increases linked to inflation and marketing efforts. However, market share erosion will limit overall revenue for branded products.
- Generic Brinzolamide: Prices for generic brinzolamide are projected to continue a downward trend, albeit at a slower pace than in the initial years following patent expiry. This is due to ongoing competition among multiple generic manufacturers and increasing negotiation power of large pharmacy benefit managers (PBMs) and healthcare systems. A further price decrease of 10-20% is anticipated for generics over the next five years.
Factors influencing future pricing:
- Generic Competition Intensity: The number and strategic approaches of generic manufacturers will dictate the pace of price declines.
- Reimbursement Policies: Government and private payer policies will continue to influence out-of-pocket costs and overall market pricing.
- Therapeutic Alternatives: The development of new glaucoma treatments or alternative drug classes could impact brinzolamide's market positioning and pricing power.
What is the market outlook for brinzolamide in key regions?
The market outlook for brinzolamide varies by region, influenced by local healthcare infrastructure, regulatory environments, and patient demographics.
North America (United States and Canada):
- Market Status: Mature market dominated by generics.
- Outlook: Stable demand driven by established treatment protocols. Price competition remains intense, favoring generics. Focus on cost-effectiveness by payers.
Europe:
- Market Status: Similar to North America, with significant generic penetration post-patent expiry.
- Outlook: Strong demand in countries with high glaucoma prevalence. Pricing is a key consideration for national health services. Regional variations in generic adoption exist.
Asia-Pacific:
- Market Status: Growing market with increasing healthcare access.
- Outlook: Significant growth potential driven by rising incomes, increasing awareness, and a large aging population. Branded products may retain a larger share initially, but generic uptake is accelerating. China and India represent key growth areas.
Latin America:
- Market Status: Developing market with a growing need for ophthalmic treatments.
- Outlook: Increasing demand for both branded and generic options as healthcare infrastructure improves. Price sensitivity is a major factor.
Middle East and Africa:
- Market Status: Emerging market with significant unmet needs.
- Outlook: The region presents long-term growth opportunities as access to healthcare and specialized treatments expands. Generic brinzolamide is likely to be the primary entry point due to cost considerations.
What are the regulatory considerations for brinzolamide?
Regulatory approval and compliance are critical for brinzolamide products.
- FDA (United States): Brinzolamide is approved as brinzolamide ophthalmic suspension 1% for the reduction of elevated intraocular pressure. Generic versions require an Abbreviated New Drug Application (ANDA) demonstrating bioequivalence to the reference listed drug (Azopt).
- EMA (Europe): Brinzolamide is authorized under various brand names, including Azopt. Generic applications follow similar principles of demonstrating quality, safety, and efficacy.
- Other Jurisdictions: Regulatory requirements vary by country, with agencies like Health Canada, TGA (Australia), and CDSCO (India) having their own approval processes.
Key regulatory considerations include:
- Good Manufacturing Practices (GMP): Manufacturers must adhere to strict GMP guidelines for ophthalmic preparations.
- Quality Control: Robust quality control measures are essential to ensure product consistency and sterility.
- Labeling and Packaging: Regulations dictate specific requirements for product labeling and packaging to ensure patient safety and correct usage.
What are the future trends and innovations in brinzolamide treatment?
While brinzolamide is a well-established drug, future developments may include:
- Improved Formulations: Research into sustained-release formulations or novel drug delivery systems could enhance patient compliance and treatment efficacy.
- Fixed-Dose Combinations: Continued development of fixed-dose combinations with other IOP-lowering agents will be a key strategy for manufacturers seeking to differentiate their products.
- Adjunctive Therapies: Brinzolamide may be evaluated in combination with emerging therapies for glaucoma management.
- Biosimilar Development: While not applicable to small molecule drugs like brinzolamide, the concept of highly similar versions with comparable efficacy and safety profiles is a broader trend in pharmaceuticals.
Key Takeaways
Brinzolamide is a mature ophthalmic drug facing significant generic competition, leading to price erosion. The global market is valued at approximately USD 750 million, with generics holding a 55% market share. Demand is driven by the rising prevalence of glaucoma and ocular hypertension, increasing awareness, and the drug's proven efficacy. Future pricing will likely see continued declines in generic versions, while branded products will focus on combination therapies. Growth opportunities exist in emerging markets, particularly in the Asia-Pacific region.
Frequently Asked Questions
1. How does brinzolamide work to reduce intraocular pressure?
Brinzolamide is a carbonic anhydrase inhibitor that works by decreasing the production of aqueous humor in the eye. This reduction in fluid production leads to a decrease in intraocular pressure (IOP), which is a key factor in managing glaucoma and ocular hypertension.
2. What are the main side effects associated with brinzolamide use?
Common side effects of brinzolamide include blurred vision, dry eyes, eye discomfort, foreign body sensation, and unpleasant taste. Less common side effects can include headache, dizziness, and allergic reactions. Patients should consult their healthcare provider for a full list of potential side effects.
3. Is brinzolamide suitable for all types of glaucoma?
Brinzolamide is primarily indicated for open-angle glaucoma and ocular hypertension. Its efficacy in other forms of glaucoma, such as angle-closure glaucoma, may vary, and it is typically used in conjunction with other treatments under specific medical guidance.
4. How do generic versions of brinzolamide compare to the branded product in terms of efficacy and safety?
Generic brinzolamide products are required to be bioequivalent to the branded product (Azopt). This means they contain the same active ingredient, dosage form, strength, and route of administration, and they are expected to have the same clinical effect and safety profile when used as directed.
5. What is the typical duration of brinzolamide treatment for glaucoma?
Treatment for glaucoma with brinzolamide is typically long-term, often lifelong. The goal is to continuously manage intraocular pressure to prevent further optic nerve damage and vision loss. The duration of treatment is determined by the prescribing physician based on the patient's individual condition and response to therapy.
Citations
[1] World Health Organization. (n.d.). Blindness and vision impairment. Retrieved from https://www.who.int/news-room/fact-sheets/detail/blindness-and-vision-impairment
More… ↓
