Last updated: November 21, 2025
Introduction
Ambrisentan is an endothelin receptor antagonist primarily indicated for the treatment of pulmonary arterial hypertension (PAH). As of 2023, it has established itself as a critical therapeutic option within the niche of PAH management, balancing efficacy with a relatively favorable safety profile. This analysis explores the current market landscape, competitive positioning, key drivers, challenges, and forecasts of ambrisentan's price trajectory over the next five years, offering actionable insights for stakeholders.
Market Landscape of Ambrisentan
Therapeutic Indication and Market Need
Pulmonary arterial hypertension is a progressive, often fatal, disorder characterized by elevated pulmonary arterial pressure leading to right heart failure. The global PAH market is projected to grow at a compound annual growth rate (CAGR) of approximately 7% between 2023 and 2028, driven by increased diagnosis rates, expanding treatment guidelines, and the approval of novel therapies [1].
Ambrisentan, marketed under brands such as Letairis, holds a significant share within endothelin receptor antagonists (ERAs), a class that includes bosentan and macitentan. Its selectivity for the endothelin A receptor offers distinct advantages, including reduced hepatotoxicity compared to its predecessors.
Market Size and Revenue
Based on current sales data, the global ambrisentan market generated approximately USD 350 million in 2022, with North America accounting for the largest share due to high diagnosis rates and reimbursement coverage. Emerging markets like China and India represent significant future growth opportunities but currently have limited penetration owing to pricing and regulatory hurdles [2].
Competitive Landscape
The dominant competitors include bosentan and macitentan, both of which have broader indications but also come with distinct safety considerations. Ambrisentan's targeted safety profile and dosing convenience position it as a preferred choice in many cases. Patent expirations of key drugs and the introduction of generic formulations will influence market dynamics, potentially expanding access but lowering average prices.
Regulatory and Patent Status
Ambrisentan received FDA approval in 2007 for PAH, with subsequent approvals in other jurisdictions. Its patent protection is projected to conclude around 2025, although patent challenges and regulatory exclusivities may modify this timeline. Once patent expiry occurs, biosimilar or generic manufacturers are expected to enter the market, exerting downward pressure on prices.
Key Market Drivers
- Increasing Diagnosis and Awareness: Improved screening protocols lead to earlier detection, expanding the patient base.
- Expansion of Indications: Ongoing research may extend ambrisentan's use in other forms of PAH or related conditions.
- Reimbursement Policies: Favorable insurance coverage in high-income regions enhances drug accessibility.
- Patient Preference: Once-daily dosing and safety profile favor adherence, positively impacting sales.
Price Dynamics and Projection
Historical Pricing Trends
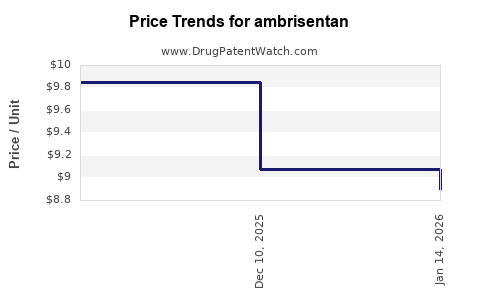
In the United States, the list price of ambrisentan has hovered around USD 4,300 per month per patient in 2022, with net prices varying based on rebates, discounts, and insurance negotiations. Historically, drug prices for PAH therapies have been high, driven by the specialized patient population and manufacturing complexity.
Factors Influencing Price Trajectories
- Patent Expiry and Biosimilar Entry: Predicted around 2025, likely to induce significant price reductions.
- Market Competition: Introduction of generics may reduce prices by 20–50% within 1–3 years of market entry.
- Regulatory Changes: Price regulation policies, particularly in Europe and Asia, could further influence pricing.
- Manufacturing Costs: Steady technological advances might lead to reduced production costs over time, marginally impacting prices.
Future Price Projections (2023–2028)
| Year |
Estimated Average Monthly Price (USD) |
Key Factors |
| 2023 |
4,250 – 4,500 |
Pre-patent expiration, stable demand, moderate competition |
| 2024 |
4,100 – 4,400 |
Patent nearing expiry, increasing generic discussions |
| 2025 |
2,500 – 3,000 |
Patent expiry, first generics entering, price decline begins |
| 2026–2028 |
1,500 – 2,000 |
Market stabilization with multiple generics, competitive pressures |
Note: These projections are approximate, subject to regulatory developments, patent challenges, and market entry timelines.
Challenges and Opportunities
Challenges
- Patent Expiry and Biosimilars: The impending expiration could dilute revenue streams.
- Pricing and Reimbursement Constraints: High drug costs create barriers in emerging markets.
- Competitive Dynamics: Superior efficacy or safety profiles of existing drugs like macitentan could limit ambrisentan's market share.
Opportunities
- Combination Therapies: Co-administration with other PAH agents can drive volume.
- Expanding Indications: Potential use in other endothelin-related conditions widens the target population.
- Pricing Strategies: Tiered pricing and value-based models could optimize revenue streams.
Implications for Stakeholders
For pharmaceutical companies, strategic planning around patent cliffs and biosimilar entries is crucial. Price projections support making informed decisions on market entry, licensing, or negotiations with payers. Healthcare providers and payers should consider these trends when assessing formulary inclusions and reimbursement policies.
Key Takeaways
- The ambrisentan market is poised for moderate growth, driven by increased PAH diagnosis and expanding indications.
- Patent expiration around 2025 is a pivotal event expected to significantly reduce drug prices through generic competition.
- Pricing projections indicate a decline from approximately USD 4,300/month in 2022 to below USD 2,000/month by 2028.
- Staying ahead of patent expiries with strategic product positioning and diversification into combination therapies can sustain revenue.
- Global market access requires tailored approaches, considering regional pricing regulations and reimbursement landscapes.
FAQs
-
When will generic versions of ambrisentan enter the market?
Patent protections are expected to expire around 2025, opening the door for generics that could significantly lower prices within 1–3 years post-expiry.
-
How does ambrisentan compare economically with other PAH therapies?
Currently priced around USD 4,300/month, ambrisentan’s cost is comparable to alternatives like bosentan but may offer savings post-patent expiry when generics emerge.
-
What factors could delay price reductions?
Regulatory hurdles, patent litigation, or limited generic manufacturing capacity could delay price declines.
-
Are there emerging competitors that threaten ambrisentan’s market share?
Yes; drugs like macitentan and novel agents under development may challenge its dominance, especially if offering improved efficacy or safety.
-
How can payers leverage this information?
Anticipating price reductions allows for strategic formulary management, negotiated discounts, and informed coverage decisions, especially around patent expiration timelines.
Sources
[1] Grand View Research. Pulmonary Arterial Hypertension Market Size, Share & Trends Analysis Report (2022).
[2] IQVIA. Pharmaceutical Market Insights and Sales Data (2022).