Last updated: February 13, 2026
What Is the Current Market Size for Acarbose?
Acarbose, an alpha-glucosidase inhibitor, is used primarily for managing type 2 diabetes. The global market for acarbose was valued at approximately $500 million in 2022. Growth is driven by increasing prevalence of diabetes worldwide, particularly in Asia-Pacific regions, where acarbose is more commonly used due to its affordability and availability.
The Asia-Pacific region accounted for roughly 55% of the market share in 2022, with China, India, and Japan leading sales. North America and Europe contribute smaller portions but are experiencing steady growth owing to rising diabetes rates and expanding healthcare infrastructure.
What Are the Key Market Drivers and Constraints?
Drivers:
- Rising diabetes prevalence: the International Diabetes Federation estimates 537 million adults with diabetes globally in 2021, projected to reach 643 million by 2030.
- Cost-effective therapy: acarbose is less expensive compared to newer antidiabetic drugs, appealing to developing countries.
- Approvals in emerging markets: regulatory approvals in India, Southeast Asia, and parts of Latin America expand access.
Constraints:
- Competition from newer medications: GLP-1 receptor agonists and SGLT2 inhibitors are gaining market share due to better efficacy profiles.
- Side effects: gastrointestinal issues limit patient acceptance.
- Patent expirations: no recent patents, fostering generic competition and pressure on prices.
How Is the Market Anticipated to Evolve?
Forecast Overview:
Projections suggest the acarbose market will grow at a compound annual growth rate (CAGR) of 3% from 2023 to 2028, reaching roughly $575 million by 2028. Growth will predominantly occur in developing nations.
Factors Influencing Growth:
- Increased screening and diagnosis of type 2 diabetes.
- Greater acceptance of oral medications in countries with limited healthcare expenditure.
- Potential entry of new formulations, such as sustained-release versions, which could enhance adherence.
Regulatory and Pricing Policies:
- Pricing strategies in emerging markets will impact revenue; price ceilings may limit profit margins.
- Evolving regulations around generics could affect market entry and competition.
What Are Price Projections and Trends?
Present Pricing Landscape:
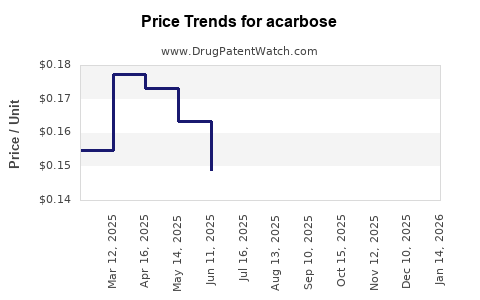
- Brand-name acarbose in developed countries costs approximately $3 to $5 per 50 mg tablet.
- Generic versions are priced between $0.5 and $1 per tablet.
- In developing markets, price points are lower, often under $0.50 per tablet.
Future Pricing Dynamics:
- Price reductions are expected due to proliferation of generics and biosimilars.
- Price erosion could reach 20-30% over the next five years in mature markets.
- Value-based pricing and tiered reimbursement models might influence pricing strategies.
Market Penetration and Accessibility:
- Sales volume is projected to increase as more patients adopt acarbose as a first- or second-line therapy.
- Cost savings on generics will maintain demand, especially in resource-constrained settings.
How Do Competitive and Regulatory Factors Impact Market and Price?
- Patent expirations in 2010 and the absence of recent patents encourage generic manufacturing.
- Regulatory agencies in global markets streamline approval processes for generics, accelerating market entry.
- Competition among generic manufacturers drives prices downward.
- Innovations, such as combination therapies with acarbose, could command premium pricing if approved.
Summary Table: Key Market Data and Projections
| Parameter |
2022 Data |
2028 Projection |
Notes |
| Market size |
$500 million |
~$575 million |
3% CAGR |
| Asia-Pacific share |
55% |
Expected to increase |
Due to regional prevalence |
| Average generic price |
$0.5-$1 per tablet |
Similar or lower |
Price erosion continues |
| Growth rate |
3% CAGR |
3% CAGR |
Steady expansion in emerging markets |
Key Takeaways
- Acarbose's global market is valued at approximately $500 million, with growth driven primarily by Asia-Pacific.
- Market expansion will continue at around 3% annually through 2028.
- Price declines are expected due to increased generic competition, with future prices trending downward across regions.
- Regulatory environment favors faster approval of generics, enhancing accessibility but compressing margins.
- Competition from newer therapies remains a long-term factor constraining market share.
FAQs
1. What factors most significantly influence acarbose pricing?
Generic availability, regional regulatory policies, and competitive forces primarily determine acarbose prices. Cost reductions via generics drive prices downward, especially in markets with active competition.
2. Which regions will see the fastest market growth?
Emerging markets in Asia-Pacific, including China and India, will account for the majority of growth due to increasing diabetes prevalence and cost-sensitive healthcare policies.
3. How do newer diabetes medications impact acarbose’s market?
Medications such as SGLT2 inhibitors and GLP-1 receptor agonists offer superior efficacy and tolerability, gradually capturing market share from acarbose, especially in developed markets.
4. Will patent expirations influence future market dynamics?
Yes. Patent expirations around 2010 resulted in widespread generic manufacturing, decreasing prices and constraining profit margins.
5. What are potential threats to acarbose market growth?
Market growth faces threats from the development of new classes of antidiabetic drugs, regulatory restrictions on older drugs, and adverse side effect profiles limiting patient use.
References
- International Diabetes Federation. IDF Diabetes Atlas, 10th ed. 2021.
- Grand View Research. Acarbose Market Size, Share & Trends Analysis Report, 2022–2028.
- IQVIA. Market Intelligence Reports on Antidiabetic Drugs, 2022.
- U.S. Food and Drug Administration. Drug Approvals and Patent Status Reports, 2010–2022.
- MarketWatch. Global Diabetes Drugs Market Trends, 2023.