Share This Page
Drug Price Trends for ZYLET EYE DROPS
✉ Email this page to a colleague
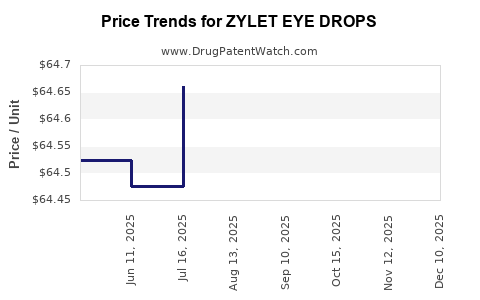
Average Pharmacy Cost for ZYLET EYE DROPS
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ZYLET EYE DROPS | 24208-0358-05 | 64.49686 | ML | 2026-05-20 |
| ZYLET EYE DROPS | 24208-0358-10 | 64.45400 | ML | 2026-05-20 |
| ZYLET EYE DROPS | 24208-0358-10 | 64.39433 | ML | 2026-04-22 |
| ZYLET EYE DROPS | 24208-0358-05 | 64.50867 | ML | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Overview of ZYLET Eye Drops
ZYLET Eye Drops combine two active ingredients: loteprednol etabonate (a corticosteroid) and tobramycin (an antibiotic). Approved by the FDA in 2008, they target post-operative ocular inflammation and infection management. The drug competes in a niche market directed at ophthalmologists, primarily addressing patients following eye surgery or with inflammation-associated infections.
Market Size and Key Players
The global ophthalmic drugs market was valued at approximately $31.95 billion in 2022 and is projected to reach $44.66 billion by 2030, growing at a CAGR of 4.4% (Grand View Research). The segment including corticosteroid-antibiotic combination drugs is a minor but significant subset, estimated at about 10% of the overall market.
Several drugs serve similar indications, including:
- Pred-G (prednisolone acetate with antibiotic)
- Tobradex (tobramycin and dexamethasone)
- Zylet (loteprednol and tobramycin)
Zylet maintains a competitive advantage with a favorable side effect profile compared to dexamethasone-based formulations, owing to the lower propensity for increased intraocular pressure.
Pricing Dynamics and Revenue Streams
Average retail price per unit (a one-month supply):
- US: $80 - $120
- Europe: €60 - €90
Pricing varies based on region, insurance reimbursement, and healthcare provider contracts. Wholesale acquisition costs (WAC):
| Region | WAC (per bottle) | Retail Approximate Price |
|---|---|---|
| US | $69 | $80 - $120 |
| Europe | €50 - €70 | €60 - €90 |
Profits depend on patent status. Patent expiration in 2019 opened the market to generics, significantly influencing pricing. As of 2022, generic versions are widely available at approximately 30-50% lower prices.
Patent and Regulatory Status
- Original patent expired in 2019 in major markets.
- Patent challenges and generic approvals have increased market competition.
- No new formulation or dosage schedule approvals are pending, limiting patent-based exclusivity.
Market Trends and Drivers
- Surge in ophthalmic surgeries (LASIK, cataract removal) drives demand for anti-inflammatory and antibiotic medications.
- Rising prevalence of ocular surface disease linked to aging populations supports ongoing need.
- Manufacturers focus on safety, minimizing steroid-related intraocular pressure increases.
Price Projections (2023-2030)
Anticipated factors:
- Entry of generic competitors will pressure prices downward.
- Increased demand due to aging demographics and surgery volume sustains sales volumes.
- Potential for price stabilization with biosimilar and additional formulations.
Projected retail prices:
| Year | US Price Range | Comments |
|---|---|---|
| 2023 | $70 - $110 | Market stabilization post-patent expiry |
| 2025 | $65 - $100 | Generics penetrance increases, pressures prices |
| 2027 | $60 - $90 | Market consolidates further |
| 2030 | $55 - $85 | Price decline plateau with increased competition |
Competitive Strategies
Manufacturers are investing in:
- Making generic versions more accessible.
- Developing combination therapies with improved safety profiles.
- Expanding into emerging markets with growing ophthalmic surgery volumes.
Regulatory and Policy Impact
Reimbursement policies influence net revenue; regions with national health systems tend toward price controls, impacting profitability. The US Inserm Medicare Part D program and other private insurers influence copayment levels for patients.
Risks and Opportunities
- Patent litigation risks could delay generic entry.
- Opportunities exist in expanding indications and formulations, such as preservative-free options.
- Market growth could be hindered by patient compliance issues and side effect concerns.
Key Takeaways
- ZYLET faces market pressure from branded and generic competitors post-patent expiration.
- Price decline is expected, with retail prices potentially falling by approximately 20-30% over the next five years.
- Growing ophthalmic procedures sustain demand, but reimbursement policies exert downward pressure on margins.
- Development of new formulations and biosimilars may create pricing and market-share opportunities.
- Broader healthcare trends, such as aging populations and increasing surgical volumes, support long-term demand.
FAQs
1. What factors influence ZYLET’s pricing?
Patents, generic competition, regional regulations, reimbursement policies, and market demand.
2. How will generics impact the market?
They will lower prices and erode market share for the branded product, incentivizing innovation and formulation improvements.
3. Are there upcoming regulatory changes affecting ZYLET?
No significant pending approvals; however, policy shifts affecting drug reimbursement could influence profitability.
4. What is the outlook for ZYLET’s sales volume?
Sales volume is expected to remain stable or grow modestly due to increasing ophthalmic surgeries, despite price pressures.
5. Which regions offer the highest revenue potential?
The US holds the largest market share due to high surgical volumes and healthcare spending, with growth expected in Asia-Pacific regions.
Sources:
[1] Grand View Research, "Ophthalmic Drugs Market Size & Trends," 2022.
[2] FDA Drug Database, ZYLET approval and patent status.
[3] IQVIA, Prescription Sales Data, 2022.
More… ↓
