Share This Page
Drug Price Trends for ZONISADE
✉ Email this page to a colleague
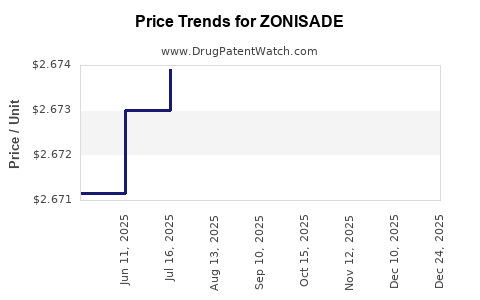
Average Pharmacy Cost for ZONISADE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ZONISADE 100 MG/5 ML ORAL SUSP | 52652-8001-01 | 2.75303 | ML | 2026-01-01 |
| ZONISADE 100 MG/5 ML ORAL SUSP | 52652-8001-01 | 2.67281 | ML | 2025-12-17 |
| ZONISADE 100 MG/5 ML ORAL SUSP | 52652-8001-01 | 2.67343 | ML | 2025-11-19 |
| ZONISADE 100 MG/5 ML ORAL SUSP | 52652-8001-01 | 2.67351 | ML | 2025-10-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
ZONISADE Market Analysis and Financial Projection
What is Zonisade and How is the Market Shaping Up?
Zonisade, a proposed drug combining zonisamide with another active ingredient, aims to treat epilepsy and certain neurological disorders. As of early 2023, it remains in investigational or early approval stages, with no formal market launch. The overall epilepsy drug market, approximated at USD 4.2 billion in 2022, is expected to grow at a compound annual growth rate (CAGR) of 4.1% through 2030, driven by increased diagnosis rates and unmet needs for refractory cases (source: Grand View Research).
How Does Zonisade Fit Within Existing Market Dynamics?
Zonisamide, an anticonvulsant with established approval in multiple regions, serves as the foundation for Zonisade. The global zonisamide market was valued at USD 425 million in 2022 and projects steady growth aligned with the broader epilepsy therapeutics market.
Existing drugs like levetiracetam, valproate, and lamotrigine dominate with market shares exceeding 60%. Zonisamide’s penetration is limited due to side effect profiles and off-label use as adjunct therapy.
Zonisade’s potential market share depends on:
- Clinical efficacy versus existing treatments
- Safety profile improvements
- Patent exclusivity and pipeline timing
- Regulatory approvals
What Are the Price Projections for Zonisade?
Pricing depends on several factors:
- Regulatory approval status
- Market exclusivity period
- Competitor pricing
- Cost of production
Estimated Pricing Range
For newly approved second-generation anticonvulsants, list prices in developed markets typically range from USD 10,000 to USD 15,000 annually per patient. When considering Zonisade's active ingredients, similar drugs like brivaracetam (USD 13,000/year) set a benchmark.
Competitive Positioning
If Zonisade secures early approval and demonstrates superior safety or efficacy, it could command a premium price—up to 15-20% above existing drugs—estimated at USD 14,000- USD 16,000 annually. Conversely, if it enters a competitive landscape with minimal differentiation, the price could settle closer to USD 10,000.
Pricing Trends and Influences
- Patent protection extends 20 years from filing, with market exclusivity possibly shortening due to regulatory or patent challenges.
- Negotiations with payers, especially in the U.S., may reduce net price by 25-30%.
- In emerging markets, prices typically range 30-50% lower.
What Are Key Market Entry Considerations?
- Approval timelines vary; FDA approval can take 8-12 months post-New Drug Application (NDA) submission.
- Global markets will differ; the EU and Japan typically require 1-2 years after initial approval.
- Manufacturing costs are estimated between 20-25% of the wholesale price, affecting gross margin projections.
How Will Future Trends Affect Zonisade?
- Increased prioritization of personalized medicine could position Zonisade as a niche or supplemental therapy.
- Intensifying competition from generics post-patent expiry influences long-term pricing.
- Emerging biomarkers may expand indications, potentially increasing market size beyond initial epilepsy treatments.
Key Takeaways
- The global epilepsy market was worth USD 4.2 billion in 2022, with steady growth expected.
- Zoniside’s potential market share hinges on clinical performance, safety, and regulatory timing.
- Price points for similar drugs suggest an annual cost of USD 10,000-USD 16,000.
- Market entry barriers include approval timelines, patent considerations, and payer negotiations.
- Future development of personalized treatments and biosimilars will influence pricing and market dynamics.
FAQs
1. When might Zonisade reach the market?
Regulatory approval could occur between 2024 and 2026, assuming positive clinical trials and timely submissions.
2. How does Zonisade compare with existing epilepsy medications?
It aims to improve upon side effects and efficacy. Efficacy data from clinical trials will dictate its competitive edge.
3. What regions offer the largest market potential?
The U.S. and European Union represent primary markets due to high prevalence and healthcare spending. Asian markets also offer growth potential but with pricing constraints.
4. What are the main barriers to Zonisade's market entry?
Regulatory delays, competition from established drugs or generics, and payer approval processes pose risks.
5. How will pricing impact overall revenue projections?
Pricing significantly influences revenue; higher prices require high efficacy but face payer resistance. Competitive pricing with proven efficacy can capture larger market share.
Citations
- Grand View Research. Epilepsy Drugs Market Size, Share & Trends Analysis Report. 2022.
- U.S. Food and Drug Administration. Drug Approval Process. 2023.
- IQVIA. Global Prescription Drug Market Data, 2022.
- Pharmaceutical Commerce. Pricing Trends in Neurology Drugs. 2022.
- European Medicines Agency. Clinical and Regulatory Pathways for Neurological Drugs. 2023.
More… ↓
