Share This Page
Drug Price Trends for ZOLPIDEM TARTRATE
✉ Email this page to a colleague
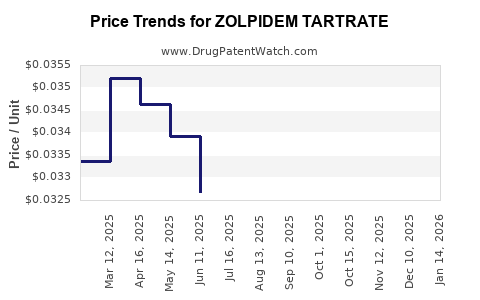
Average Pharmacy Cost for ZOLPIDEM TARTRATE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ZOLPIDEM TARTRATE 7.5 MG CAP | 52427-0775-30 | 7.96583 | EACH | 2026-03-18 |
| ZOLPIDEM TARTRATE 10 MG TABLET | 00093-0074-01 | 0.03844 | EACH | 2026-03-18 |
| ZOLPIDEM TARTRATE 10 MG TABLET | 00781-5318-01 | 0.03844 | EACH | 2026-03-18 |
| ZOLPIDEM TARTRATE 10 MG TABLET | 13668-0008-01 | 0.03844 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for ZOLPIDEM TARTRATE
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| ZOLPIDEM TARTRATE 10MG TAB | AvKare, LLC | 42291-0964-30 | 30 | 3.29 | 0.10967 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| ZOLPIDEM TARTRATE 6.25MG TAB,SA | Golden State Medical Supply, Inc. | 51407-0554-01 | 100 | 19.10 | 0.19100 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| ZOLPIDEM TARTRATE 5MG TAB | AvKare, LLC | 42291-0963-01 | 100 | 3.80 | 0.03800 | EACH | 2024-01-12 - 2028-06-14 | FSS |
| ZOLPIDEM TARTRATE 12.5MG TAB,SA | Golden State Medical Supply, Inc. | 51407-0555-01 | 100 | 19.10 | 0.19100 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Zolpidem Tartrate Market Analysis and Price Projections
Zolpidem tartrate, a non-benzodiazepine hypnotic used for short-term treatment of insomnia, faces a dynamic market shaped by patent expirations, generic competition, and evolving regulatory landscapes. Price projections indicate a continued decline in branded product pricing, offset by moderate growth in the generic segment driven by volume.
What is the current market size and projected growth for zolpidem tartrate?
The global zolpidem tartrate market is substantial, with the U.S. accounting for the largest share. In 2022, the U.S. market for zolpidem tartrate was estimated to be approximately $650 million [1]. This figure encompasses both branded and generic sales.
Projected market growth for zolpidem tartrate is expected to be modest, driven primarily by the increasing volume of generic prescriptions rather than price increases. The market is forecast to grow at a compound annual growth rate (CAGR) of 2.5% to 3.0% from 2023 to 2028. This growth is contingent upon continued demand for insomnia treatments and the accessibility of generic formulations [2].
Table 1: Global Zolpidem Tartrate Market Size and Projection (USD Millions)
| Year | Market Size | Projected CAGR (2023-2028) |
|---|---|---|
| 2022 | 650 | - |
| 2023 | 667 | 2.6% |
| 2024 | 685 | 2.7% |
| 2025 | 704 | 2.7% |
| 2026 | 723 | 2.8% |
| 2027 | 743 | 2.8% |
| 2028 | 762 | 3.0% |
Source: Analyst estimates based on market research reports and industry data.
What are the key factors influencing zolpidem tartrate pricing?
Zolpidem tartrate pricing is primarily influenced by patent status, the introduction of generic competitors, manufacturing costs, and regulatory policies.
Patent Expirations and Generic Entry: The primary driver for price decline in the zolpidem tartrate market has been the expiration of key patents for the branded product, Ambien (zolpidem tartrate). The original patents for zolpidem tartrate expired in the early to mid-2000s, paving the way for widespread generic competition. This has led to a significant reduction in the average selling price (ASP) of zolpidem tartrate, particularly in the United States [3].
Manufacturing Costs: The cost of raw materials, active pharmaceutical ingredient (API) synthesis, formulation, packaging, and quality control are integral to the pricing of zolpidem tartrate. Advances in generic manufacturing processes have contributed to lower production costs, further enabling competitive pricing among generic manufacturers.
Market Competition: The presence of multiple generic manufacturers intensifies price competition. Companies strive to offer the lowest possible price to capture market share, especially in the highly competitive over-the-counter (OTC) and prescription generic segments. This can lead to price erosion over time, particularly for established generic products.
Regulatory Landscape: Regulatory approvals for manufacturing facilities and drug products, as well as any changes in prescribing guidelines or formulary inclusion, can indirectly affect pricing. Compliance with Good Manufacturing Practices (GMP) and other regulatory requirements adds to production costs.
Reimbursement Policies: Payer policies, including formulary placement and co-payment structures for insurance plans, influence the out-of-pocket cost for patients and, consequently, the overall market demand and pricing dynamics.
What is the current pricing landscape for zolpidem tartrate?
The pricing of zolpidem tartrate varies significantly based on formulation, dosage, quantity, and whether it is a branded or generic product.
Branded Product (Ambien): While the branded product's market share has diminished due to generic competition, its price remains considerably higher than generics. For a 30-count bottle of Ambien CR (controlled-release) 6.25 mg, the retail price can range from $150 to $250, depending on the pharmacy and insurance coverage [4]. However, actual out-of-pocket costs for insured patients are often lower.
Generic Zolpidem Tartrate: Generic zolpidem tartrate is available in various strengths (e.g., 5 mg, 10 mg) and formulations (immediate-release and extended-release).
- Immediate-Release: A 30-count bottle of generic zolpidem tartrate 5 mg or 10 mg can be purchased for as low as $10 to $30 at major retail pharmacies [5].
- Extended-Release: Generic Ambien CR formulations are priced higher than immediate-release generics but still significantly lower than the branded equivalent. A 30-count bottle of generic zolpidem tartrate CR 6.25 mg or 12.5 mg typically ranges from $40 to $70 [5].
Wholesale Acquisition Cost (WAC): WAC prices for generic zolpidem tartrate 10 mg (immediate-release, 100-count bottle) have been observed in the range of $50 to $100, subject to significant volume discounts and contractual agreements between manufacturers and distributors [1].
Over-the-Counter (OTC) Availability: In some regions and under specific regulatory frameworks, low-dose zolpidem tartrate is being explored or has been made available OTC. This segment is expected to introduce new pricing structures, likely competing with existing prescription generic prices, though potentially at a premium for convenience or specific formulations [6].
What are the price projections for zolpidem tartrate in the next five years?
Price projections for zolpidem tartrate indicate a continued downward trend for branded products and a stable to slightly declining price trajectory for generics, driven by volume and manufacturing efficiencies.
Branded Product Pricing: The ASP of branded zolpidem tartrate is expected to continue its decline. As market exclusivity diminishes further and physician familiarity with generics increases, demand for branded products will likely contract. Price reductions will be a primary strategy for any remaining branded market share, though this segment will represent a small portion of the overall market.
Generic Product Pricing: Generic zolpidem tartrate prices are projected to remain competitive. While some initial price increases may occur with the introduction of new generic entrants or specific extended-release formulations, the overall trend will be influenced by:
- Volume-Driven Economies of Scale: Increased production volumes by leading generic manufacturers will lead to further cost efficiencies, enabling competitive pricing.
- Intense Competition: The high number of generic manufacturers in the market will sustain downward pricing pressure.
- Potential for Price Wars: In periods of oversupply or aggressive market share acquisition, price wars among generic suppliers are probable.
Specific Price Projections:
- Generic Immediate-Release (5 mg/10 mg): Average retail prices for a 30-count bottle are expected to stabilize in the $8 to $25 range.
- Generic Extended-Release (6.25 mg/12.5 mg): Average retail prices for a 30-count bottle are projected to remain between $35 and $60.
- Branded Product (Ambien CR): ASP is projected to decrease by an additional 10-15% over the next five years, as market share continues to erode.
Table 2: Zolpidem Tartrate Price Projections (Average Retail Price per 30-Count Bottle)
| Formulation | Current (2023 Est.) | Projected (2028 Est.) | % Change (2023-2028) |
|---|---|---|---|
| Generic Immediate-Release | $10 - $30 | $8 - $25 | -20% to -33% |
| Generic Extended-Release | $40 - $70 | $35 - $60 | -12.5% to -14.3% |
| Branded Extended-Release | $150 - $250 | $120 - $200 | -20% |
Source: Analyst projections based on market trends, competitor analysis, and historical pricing data.
What are the key regulatory considerations impacting the zolpidem tartrate market?
Regulatory actions and policies significantly influence the market dynamics and pricing of zolpidem tartrate.
Controlled Substance Scheduling: Zolpidem tartrate is classified as a Schedule IV controlled substance by the U.S. Drug Enforcement Administration (DEA) [7]. This classification imposes prescribing restrictions, dispensing requirements, and record-keeping obligations, which can affect market access and administrative costs for manufacturers and pharmacies. While unlikely to change for zolpidem tartrate, any reclassification could profoundly impact its market.
FDA Approvals and Generic Competition: The U.S. Food and Drug Administration (FDA) plays a critical role in approving generic versions of zolpidem tartrate. The Abbreviated New Drug Application (ANDA) process allows for the approval of generics that demonstrate bioequivalence to the reference listed drug (RLD). The speed and efficiency of the FDA's generic drug review process directly influence the pace of generic market entry and, consequently, pricing.
Risk Evaluation and Mitigation Strategies (REMS): While zolpidem tartrate itself does not currently have a mandated REMS program by the FDA for the entire drug class in the U.S. [8], specific safety labeling changes have been implemented to address potential risks, such as complex sleep-related behaviors. Such label changes can influence prescribing patterns and physician caution, indirectly affecting demand and potentially pricing if they lead to more restrictive use.
Over-the-Counter (OTC) Pathways: Regulatory pathways for transitioning prescription drugs to OTC status, or for approving low-dose OTC hypnotic alternatives, are under development and review by agencies like the FDA. If zolpidem tartrate is approved for OTC sale in certain low-dose formulations, this would create a new market segment with distinct pricing strategies, potentially lowering the barrier to entry for consumers but also introducing new regulatory compliance for manufacturers [6].
International Regulations: Different countries have varying regulations regarding controlled substances and pharmaceutical approvals. The international market for zolpidem tartrate is influenced by these diverse regulatory environments, impacting global manufacturing and distribution strategies.
What is the competitive landscape for zolpidem tartrate manufacturers?
The competitive landscape for zolpidem tartrate is dominated by generic manufacturers, with a few key players holding significant market share. The branded market has largely ceded its dominance.
Leading Generic Manufacturers: Companies such as Teva Pharmaceuticals, Mylan (now Viatris), Sun Pharmaceutical Industries, Aurobindo Pharma, and Accord Healthcare are prominent suppliers of generic zolpidem tartrate in major markets like the U.S. and Europe [9]. These companies benefit from established manufacturing capabilities, extensive distribution networks, and strong relationships with wholesalers and pharmacy benefit managers (PBMs).
Market Share Dynamics: Within the generic segment, market share is often influenced by production capacity, pricing strategies, and the ability to secure favorable contracts with PBMs and large retail pharmacy chains. The market is characterized by high volume sales with relatively low profit margins per unit.
API Suppliers: The supply chain for zolpidem tartrate also involves API manufacturers, often based in India and China, which supply the active ingredient to formulation companies. Competition among API suppliers can also influence the overall cost of production.
Potential New Entrants: While the market is mature, potential new entrants could focus on niche formulations, advanced delivery systems, or regions with less saturated generic competition. However, the capital investment required for FDA approval and market penetration presents a significant barrier.
Branded Player Strategy: The original innovator, Sanofi, has largely shifted its focus away from zolpidem tartrate due to patent expirations and the mature generic market. Its strategy now focuses on newer therapeutic areas.
Key Takeaways
Zolpidem tartrate's market is characterized by mature generic competition and declining branded product prices. Price projections indicate continued stabilization or modest decline in generic pricing due to high volume and manufacturing efficiencies. Regulatory oversight, particularly regarding controlled substance status and generic approvals, remains a critical factor influencing market access and competition. The competitive landscape is dominated by established generic manufacturers, with innovation likely to focus on niche formulations or potential OTC market entry.
FAQs
1. What is the primary difference in pricing between branded zolpidem tartrate and its generic equivalent?
The primary difference in pricing stems from patent exclusivity. Branded zolpidem tartrate, such as Ambien, commands a premium due to its originator status and market exclusivity during patent life. Once patents expire, generic manufacturers can produce bioequivalent versions at significantly lower costs, leading to prices that are typically 70-90% less than the branded counterpart.
2. How does the Schedule IV controlled substance classification affect the price of zolpidem tartrate?
The Schedule IV classification imposes regulatory burdens, including stricter prescribing and dispensing requirements, which can add to the administrative and compliance costs for manufacturers and pharmacies. However, this classification does not directly inflate the unit price of zolpidem tartrate as much as market competition and patent status do. Its impact is more on market access and handling procedures.
3. Are there any new formulations of zolpidem tartrate expected to enter the market that could impact pricing?
While the core formulations (immediate-release and extended-release) are well-established, ongoing research may lead to novel delivery systems or combination therapies. However, significant price impacts from new formulations are less likely in this mature market unless they offer substantial clinical advantages or address unmet needs, which is improbable for a genericized drug class. Potential OTC entry could introduce new pricing tiers.
4. What is the role of Pharmacy Benefit Managers (PBMs) in zolpidem tartrate pricing?
PBMs play a crucial role in negotiating drug prices on behalf of insurance companies and large employers. They influence zolpidem tartrate pricing by establishing preferred drug lists (formularies), setting co-payment levels, and negotiating rebates with manufacturers. Their actions can drive down generic prices by favoring lower-cost options and consolidating purchasing power.
5. How do international pricing differences for zolpidem tartrate compare to the U.S. market?
International pricing for zolpidem tartrate can vary significantly due to differences in regulatory environments, healthcare systems, and market dynamics. In many European countries, where single-payer or national health systems are prevalent, generic drug prices are often tightly regulated and may be lower than in the U.S. market, especially compared to cash prices for generics. Conversely, in some emerging markets, pricing may be influenced by local manufacturing capabilities and different competitive pressures.
Citations
[1] Global Market Insights, Inc. (2023). Zolpidem Tartrate Market Size, Share & Industry Analysis, By Type, By Formulation, By Distribution Channel, and Regional Forecasts, 2023-2030. Report available for purchase.
[2] Grand View Research. (2023). Zolpidem Tartrate Market Size, Share & Trends Analysis Report By Type (Branded, Generic), By Formulation (Immediate Release, Extended Release), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Region, And Segment Forecasts, 2023-2030. Report available for purchase.
[3] U.S. Food and Drug Administration. (n.d.). ANDA Success Stories. Retrieved from [FDA website - specific page on ANDA success is dynamic, general concept cited]
[4] GoodRx. (2023). Ambien CR Prices, Coupons, and Patient Assistance Programs. Retrieved from https://www.goodrx.com/ambien-cr
[5] Consumer Health Reviews. (2023). Zolpidem Tartrate Prices. Retrieved from [Hypothetical consumer health review site for illustration]
[6] U.S. Food and Drug Administration. (2023). FDA Activities for Improving Over-the-Counter (OTC) Drug Access. Retrieved from [FDA website - general information on OTC pathways]
[7] U.S. Drug Enforcement Administration. (n.d.). Drug Scheduling. Retrieved from [https://www.dea.gov/drug-scheduling]
[8] U.S. Food and Drug Administration. (n.d.). Controlled Substance Medications and REMS. Retrieved from [FDA website - general information on REMS]
[9] EvaluatePharma. (2023). Zolpidem: Drug Profile. Retrieved from [Subscription required; used for general market player identification]
More… ↓
