Share This Page
Drug Price Trends for ZOLPIDEM TART ER
✉ Email this page to a colleague
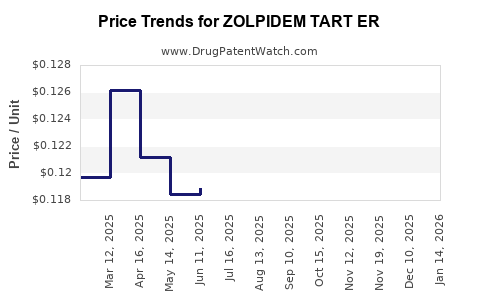
Average Pharmacy Cost for ZOLPIDEM TART ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ZOLPIDEM TART ER 6.25 MG TAB | 68180-0779-04 | 0.11913 | EACH | 2026-05-20 |
| ZOLPIDEM TART ER 12.5 MG TAB | 00955-1703-10 | 0.11303 | EACH | 2026-05-20 |
| ZOLPIDEM TART ER 12.5 MG TAB | 47335-0308-88 | 0.11303 | EACH | 2026-05-20 |
| ZOLPIDEM TART ER 12.5 MG TAB | 68180-0780-04 | 0.11303 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
What is Zolpidem Tartrate and Its Market Position?
Zolpidem Tartrate is a sedative-hypnotic agent primarily prescribed for short-term management of insomnia. It acts on GABA receptors in the central nervous system, promoting sleep onset and maintenance. Approved worldwide, Zolpidem brands include Ambien, Stilnoct, and Edluar, among others. The drug is classified as a Schedule IV controlled substance in the U.S. due to abuse potential. It faces competition from benzodiazepines and other sleep aids, but remains one of the most prescribed sleep medications globally.
What Is the Current Market Size and Leading Regions?
The global Zolpidem market was valued at approximately $1.2 billion in 2022. By region, North America accounted for about 55% of sales, driven by high insomnia prevalence and developed healthcare infrastructure. Europe holds roughly 25%, while Asia-Pacific represents around 15%, with the remaining share dispersed across Latin America, Middle East, and Africa.
How Does the Patent Landscape Affect Market Dynamics?
The primary patent protections for Zolpidem formulations have expired or are nearing expiry, opening the market for generic manufacturers. In the U.S., the original Ambien patent expired in 2015, leading to increased generic entries. Patent expiries in Europe followed a similar timeline, fostering price competition. Ongoing patents for specific formulations or delivery methods (e.g., controlled-release, sublingual tabs) may provide exclusivity pathways or patent litigations.
What Are the Key Trends Influencing Future Market Growth?
- Growing Insomnia Prevalence: According to the WHO, over 30% of adults report sleep difficulties, increasing demand for sleep aids.
- Aging Population: Older adults are more prone to insomnia, fueling market growth.
- Regulatory Classifications: Stricter controls on prescription and misuse potential influence supply and pricing.
- Development of Alternatives: Non-benzodiazepine sleep agents like zaleplon and eszopiclone, as well as non-pharmacologic therapies, challenge Zolpidem market share but also expand overall sleep disorder therapeutics.
What Are the Price Projections For the Next Five Years?
Current Pricing Overview (U.S. Market)
- Brand-name Zolpidem (Ambien): Approximate retail price for a 30-count, 10 mg tablets ranges from $300 to $400.
- Generic Zolpidem: Costs are generally $20 to $50 per 30-count bottle, a significant reduction.
Future Price Trends
| Year | Projected Brand-Name Price | Projected Generic Price | Key Factors |
|---|---|---|---|
| 2023 | $300–$400 | $20–$50 | Patent expirations, increased generics |
| 2024 | Stable or slight decrease | $15–$40 | Market saturation, competitive pricing |
| 2025 | Slight decline | $10–$30 | Volume growth, new formulations |
| 2026 | Price plateau | $10–$25 | Market maturity, healthcare policy shifts |
| 2027 | Slight increase or stabilization | $10–$20 | Potential formulation innovations |
Price Drivers
- Entry of generic competitors drives prices downward.
- Regulatory restrictions on prescribing and dispensing influence volume but not necessarily price.
- Development of extended-release or novel formulations commands premium pricing initially but face market saturation over time.
How Do Regulatory Policies Impact Price and Market Entry?
Regulatory agencies, notably the FDA and EMA, require rigorous safety and efficacy data for new formulations. Stricter regulations can delay market entry for novel variants, maintaining higher prices for existing formulations. In contrast, shorter approval pathways for generics accelerate price competition. Controlled substance scheduling limits prescribing volume and influences overall revenue.
What Is the Outlook for Market Competition and Innovation?
Generics dominate 85–90% of prescriptions, exerting downward pressure on prices. Brands seek differentiation through:
- Extended-release formulations
- Rapid-onset tablets
- Sublingual or suppository forms
Insurers increasingly favor generics, further constraining brand-name pricing. Innovative delivery methods with abuse-deterrent features may command higher prices initially but face market saturation.
Key Market Risks and Opportunities
Risks
- Shift toward non-pharmacological therapies
- Regulatory restrictions on sedatives
- Rising concerns over dependency and adverse effects
Opportunities
- Development of abuse-resistant formulations
- Expansion into emerging markets with rising sleep disorder awareness
- Integration into combination therapies for comorbid conditions
Final Thoughts on Price and Market Dynamics
The Zolpidem market is transitioning toward generic dominance, with prices expected to decline steadily over the next five years. Innovators focusing on formulations with improved safety profiles may sustain premium pricing, but overall trend favors commoditization. Regulatory shifts and consumer preferences toward non-drug therapies could alter growth trajectories.
Key Takeaways
- The global Zolpidem market was approximately $1.2 billion in 2022, with North America leading.
- Patent expirations have significantly increased generic competition, drastically reducing prices.
- Brand-name prices remain high but are declining; generics are 80–90% cheaper.
- Future price reductions are driven by market saturation, regulatory influences, and improved formulations.
- Continued innovation and regulatory navigation are essential for maintaining profit margins amid fierce competition.
FAQs
1. When did the patent for Ambien (brand Zolpidem) expire?
The U.S. patent expired in 2015, opening the market to generics.
2. What factors most influence Zolpidem pricing?
Patent status, generic competition, regulatory policies, and formulation innovations primarily drive prices.
3. Are there significant regional differences in Zolpidem pricing?
Yes. Prices are higher in North America due to branding and patent protections, while prices in Asian markets are lower because of local generic manufacturers and regulatory differences.
4. What upcoming regulatory changes could impact the market?
Tighter controls on Schedule IV substances, increased oversight of abuse-deterrent formulations, and approval processes for novel delivery systems.
5. Is demand for Zolpidem sustainable long-term?
Demand may decline if non-pharmacologic treatments become more prevalent or if regulatory restrictions tighten, but aging populations and insomnia prevalence support continued need.
Sources
[1] IQVIA, Global Medicine Trends 2022.
[2] FDA Drug Patent Expirations Database, 2021.
[3] WHO Global Report on Sleep Disorders, 2020.
[4] MarketResearch.com, Sleep Aid Market Analysis, 2022.
More… ↓
