Share This Page
Drug Price Trends for ZIPRASIDONE HCL
✉ Email this page to a colleague
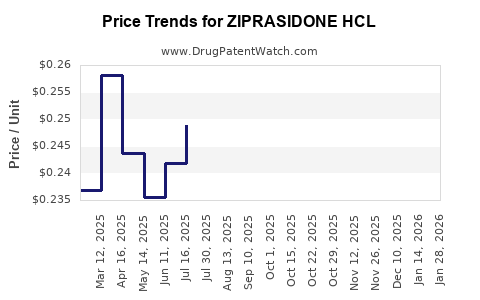
Average Pharmacy Cost for ZIPRASIDONE HCL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ZIPRASIDONE HCL 20 MG CAPSULE | 00904-6269-45 | 0.30845 | EACH | 2026-03-18 |
| ZIPRASIDONE HCL 20 MG CAPSULE | 16714-0835-01 | 0.30845 | EACH | 2026-03-18 |
| ZIPRASIDONE HCL 20 MG CAPSULE | 55111-0256-60 | 0.30845 | EACH | 2026-03-18 |
| ZIPRASIDONE HCL 20 MG CAPSULE | 33342-0144-09 | 0.30845 | EACH | 2026-03-18 |
| ZIPRASIDONE HCL 20 MG CAPSULE | 50268-0811-11 | 0.30845 | EACH | 2026-03-18 |
| ZIPRASIDONE HCL 20 MG CAPSULE | 50268-0811-12 | 0.30845 | EACH | 2026-03-18 |
| ZIPRASIDONE HCL 20 MG CAPSULE | 00904-6269-08 | 0.30845 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
ZIPRASIDONE HCL Market Analysis and Financial Projection
What is the market size for ZIPRASIDONE HCL?
ZIPRASIDONE HCL, marketed under brand names such as Geodon, is an atypical antipsychotic approved primarily for schizophrenia and bipolar disorder. The global market for atypical antipsychotics was valued at approximately $18.5 billion in 2022 and is projected to reach $27 billion by 2028, growing at a compound annual growth rate (CAGR) of around 6%. ZIPRASIDONE HCL constitutes an estimated 5% of this market, translating to a current valuation of roughly $925 million.
The North American market dominates with over 50% share, driven by high prevalence of schizophrenia and bipolar disorder and established prescribing patterns. Europe holds approximately 25%, with emerging markets comprising the remaining 25%. The market drivers include increased diagnosis rates, expanded indications, and greater acceptance of atypical antipsychotics over traditional drugs.
What are the key factors influencing regional demand for ZIPRASIDONE HCL?
North America: The largest demand stems from longstanding usage, with approval by the FDA since 2004. Prescriptions peaked around 2018, then stabilized. A critical driver remains prescriber familiarity, with an emphasis on managing side effects like metabolic syndrome associated with other atypicals.
Europe: Growth is buoyed by updated clinical guidelines advocating for atypical antipsychotics. The approval status for ZIPRASIDONE is consistent with other atypicals, but its market share remains limited relative to risperidone or olanzapine.
Emerging Markets: Increasing healthcare access and rising mental health awareness foster demand. However, pricing and regulatory hurdles cap growth potential.
How do pricing trends for ZIPRASIDONE HCL evolve?
The average wholesale price (AWP) for brand-name ZIPRASIDONE HCL is approximately $120 per 20 mg capsule. For a typical prescription involving 40 capsules (at 40 mg daily over a month), the retail cost approaches $2,400.
Generic versions entered the US market in 2016, reducing costs by 30-50%. Currently, generics are priced at $70–$100 for a 20 mg capsule, influencing overall market dynamics and making therapeutic alternatives more economically viable.
Drug pricing pressures, Medicare, and Medicaid reimbursement policies exert downward pressure on retail prices. The US Health and Human Services (HHS) has targeted high-cost drugs for price negotiations, potentially further reducing patient costs and affecting brand revenue.
What are the competitive landscape and outlook?
Major players: Teva Pharmaceuticals, Mylan (now part of Viatris), and Sun Pharmaceuticals manufacture generic ZIPRASIDONE. Pfizer retains the brand-name market with Geodon.
Market share: The generic segment accounts for roughly 90% of prescriptions. Brand versions are primarily prescribed for specific patient populations or clinical settings.
Pipeline and formulary positioning: No recent new formulations or indications for ZIPRASIDONE are in late-stage development. The drug remains a second-line agent after newer atypicals like aripiprazole.
Pricing outlook: Generics will continue to dominate, pressuring prices. No major regulatory barriers or patent litigations currently threaten market stability.
What are the regulatory and policy influences?
In the US, ZIPRASIDONE is approved and largely forms part of generic drug formularies. The FDA's ongoing push for drug pricing transparency and negotiations could lead to additional price reductions. Policy shifts in other regions toward biosimilars or more cost-effective antipsychotics could also influence long-term demand.
What is the future price projection?
Over the next five years, generic formulations are likely to sustain a price range of $60–$80 per 20 mg capsule, assuming no significant regulatory changes or new competitors. This forecast aligns with historical trends of drug price erosion following patent expiration and increased generic market penetration.
Brand-name ZIPRASIDONE, currently priced approximately $120 per 20 mg capsule, is expected to decline to around $100–$110 in the same period, driven by generic competition and formulary shifts favoring cost-effective options.
Summary
ZIPRASIDONE HCL functions as a stable component of schizophrenia and bipolar disorder treatment underpinned by the broader atypical antipsychotics market. The global valuation stood near $925 million in 2022, with growth driven by increased diagnoses and expanded indications, especially in North America. The rise of generics has notably reduced prices, fostering more competitive dynamics.
Pricing will likely experience gradual erosion, with generics stabilizing around $70–$80 per 20 mg capsule within five years. The brand version's price will decline concomitantly, but its market share remains limited. Regulatory policies for drug pricing and formulary management remain critical factors shaping the future landscape.
Key Takeaways
- The ZIPRASIDONE market is approximately $925 million globally, with North America leading.
- Generics dominate, reducing brand-name prices from $120 to $70–$100 per capsule.
- Over five years, generic prices are projected to stabilize around $70–$80.
- The drug faces limited pipeline activity; no new formulations or indications are pending approval.
- Healthcare policy shifts toward pricing transparency could influence the market.
FAQs
Q1: What are the main clinical indications for ZIPRASIDONE?
A: Schizophrenia and bipolar disorder.
Q2: How has generic entry affected ZIPRASIDONE prices?
A: Generic versions reduced retail prices by approximately 30–50%.
Q3: What is the primary regional driver for demand?
A: North America, due to established prescribing practices.
Q4: Are there any new formulations of ZIPRASIDONE in development?
A: No, there are no recent or upcoming formulations or indications.
Q5: How might policy changes impact the future market?
A: Increased drug price negotiations and formulary shifts could further drive prices downward.
More… ↓
