Share This Page
Drug Price Trends for ZADITOR
✉ Email this page to a colleague
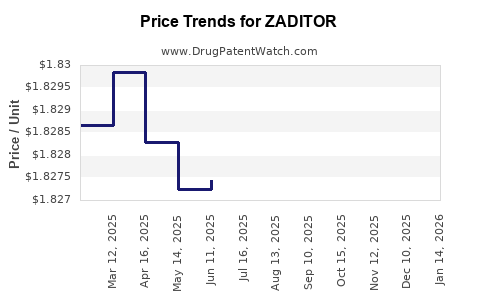
Average Pharmacy Cost for ZADITOR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ZADITOR 0.025% (0.035%) DROPS | 00065-4011-05 | 2.21835 | ML | 2026-05-20 |
| ZADITOR 0.025% (0.035%) DROPS | 00065-4011-06 | 1.85612 | ML | 2026-05-20 |
| ZADITOR 0.025% (0.035%) DROPS | 00065-4011-05 | 2.21851 | ML | 2026-04-22 |
| ZADITOR 0.025% (0.035%) DROPS | 00065-4011-06 | 1.85579 | ML | 2026-04-22 |
| ZADITOR 0.025% (0.035%) DROPS | 00065-4011-06 | 1.85010 | ML | 2026-03-18 |
| ZADITOR 0.025% (0.035%) DROPS | 00065-4011-05 | 2.22041 | ML | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for ZADITOR
Overview of ZADITOR
ZADITOR (ketotifene ophthalmic solution 0.025%) is an antihistamine used in the treatment of allergic conjunctivitis. Approved by the Food and Drug Administration (FDA) in 1986, it is marketed primarily as a prescription medication. Its primary competitors include over-the-counter antihistamines and other ophthalmic allergy treatments.
Market Size and Key Drivers
Global Allergic Conjunctivitis Market
The global allergy market, which includes treatments for allergic conjunctivitis, was valued at approximately $18.8 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 5% over the next five years, reaching $24 billion by 2027[1].
ZADITOR's Niche and Competitive Position
ZADITOR holds a niche as a topical antihistamine with a long market presence. Its advantages include a well-established safety profile, fixed-dose formulation, and familiarity among clinicians. It competes with:
- Over-the-counter oral antihistamines (e.g., cetirizine, loratadine)
- Other ophthalmic allergy medications (e.g., Olopatadine, Ketotifen fumarate brands like Alaway and Zaditor OTC)
Prescription vs. Over-the-Counter (OTC) Sales
A significant portion of ZADITOR’s sales derive from prescription channels, though some formulations have transitioned to OTC status in various markets. OTC availability has increased competition, exerting downward pressure on prices and margins.
Pricing Analysis
Current Pricing Landscape
- Prescription ZADITOR (U.S.): Average wholesale price (AWP) approximates $100 for a 5-mL bottle (30-day supply). Actual pharmacy cash prices vary based on insurance, discount programs, and pharmacy benefit managers.
- OTC Versions (Zaditor OTC): Retail prices range from $10 to $15 per bottle (10 mL), roughly equivalent to $80 to $120 per 30-day supply.
Price Trends
- Historical trends: The pricing of prescription ZADITOR has remained relatively stable over the past five years, with slight decreases driven by increased OTC availability and generic competition.
- Impact of generics: Though ZADITOR's patent expired decades ago, limited generic competition exists due to manufacturing complexities and market dynamics. This has kept prices relatively stable, with minor reductions.
Regulatory and Market Dynamics Impacting Pricing
- OTC Transition: The switch of some formulations to OTC enhances volume but exerts pressure on prices.
- Reimbursement policies: Insurance coverage favors OTC formulations, increasing access but reducing reimbursement for prescription versions.
- Competition: Newer antihistamine eye drops (e.g., Olopatadine 0.1%) offer enhanced efficacy, competitive pricing, and higher market share, impacting ZADITOR's sales growth.
Future Price Projections and Market Outlook
Short-term (Next 1-2 Years)
- Prices for prescription ZADITOR are likely to stabilize around current levels, owing to limited direct competition.
- OTC prices may decline slightly due to generic competition and market saturation.
- Overall market share remains steady, with modest growth driven by increased allergy prevalence.
Mid to Long-term (3-5 Years)
- Introduction of new antihistamine formulations could further erode ZADITOR’s market share.
- Price erosion is plausible, with retail prices potentially decreasing 10-15% as newer, more effective drugs gain prominence.
- Market expansion may occur in emerging markets, but pricing strategies will differ based on local regulatory and reimbursement landscapes.
Investment and Business Implications
- For industry stakeholders, maintaining competitive pricing will require innovation, such as combination products or improved formulations.
- Price sensitivity exists in markets where OTC options dominate, reducing profit margins.
- Strategic partnerships with pharmacy benefit managers could influence pricing dynamics favorably.
Summary
ZADITOR sustains a niche within the allergy ophthalmic treatment landscape. Price stability persists due to limited generic competition, but growing OTC availability and newer treatment options restrict growth and exert downward pricing pressure. Short-term prospects suggest stable pricing, whereas mid-term outlook indicates potential declines driven by market competition and changing regulatory environments.
Key Takeaways
- ZADITOR's global market is constrained by competition from OTC products and newer antihistamines.
- Current wholesale and retail prices are stable, with minor reductions attributable to market dynamics.
- The transition of formulations to OTC influences pricing and sales volume.
- Future pricing will depend on drug efficacy, market competition, and regulatory shifts, especially in emerging markets.
- Strategic positioning and product differentiation are necessary to sustain margins.
FAQs
1. How does ZADITOR compare price-wise with its competitors?
ZADITOR's prescription prices hover around $100 per 30-day supply, similar to other prescription antihistamines but higher than OTC brands like Zaditor OTC, which retail for about $10-$15.
2. Is ZADITOR expected to lose market share to newer medications?
Yes, newer antihistamine eye drops with improved efficacy and safety profiles are capturing market share, especially in the OTC segment.
3. What are the regulatory prospects affecting ZADITOR's pricing?
OTC transitions increase accessibility but can lead to price reductions. Regulatory barriers for new formulations or patents impact future pricing strategies.
4. How do reimbursement policies influence ZADITOR's pricing?
Reimbursement favors OTC options, decreasing pharmacy benefit coverage for prescription products and pressuring pricing and margins.
5. What is the outlook for ZADITOR’s market growth?
Growth will mainly stem from increased allergy prevalence and geographic expansion, but market share stagnation or decline is expected amid rising competition.
References
[1] Grand View Research, "Allergic Conjunctivitis Market Size, Share & Trends," 2022.
More… ↓
