Share This Page
Drug Price Trends for XIFAXAN
✉ Email this page to a colleague
Average Pharmacy Cost for XIFAXAN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
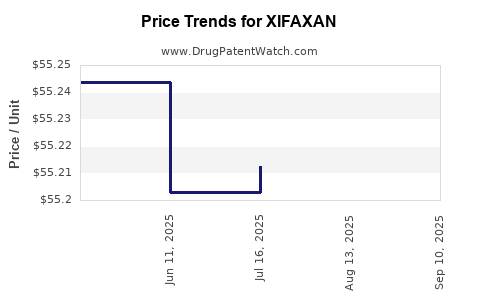
| XIFAXAN 550 MG TABLET | 65649-0303-03 | 55.28020 | EACH | 2025-09-17 |
| XIFAXAN 550 MG TABLET | 65649-0303-02 | 55.28020 | EACH | 2025-09-17 |
| XIFAXAN 550 MG TABLET | 65649-0303-03 | 55.28045 | EACH | 2025-08-20 |
| XIFAXAN 550 MG TABLET | 65649-0303-02 | 55.28045 | EACH | 2025-08-20 |
| XIFAXAN 550 MG TABLET | 65649-0303-02 | 55.21243 | EACH | 2025-07-23 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
XIFAXAN Market Analysis and Price Projections
XIFAXAN (rifaximin) is an antibiotic used to treat hepatic encephalopathy (HE) and traveler's diarrhea. Its market trajectory is influenced by patent expiries, generic competition, and evolving treatment guidelines.
What is the Current Market Status of XIFAXAN?
XIFAXAN's primary indication is for the reduction of risk of overt hepatic encephalopathy recurrence in patients 18 years of age or older. It is also approved for the treatment of irritable bowel syndrome with diarrhea (IBS-D) in adult women and men. The drug is marketed by Bausch Health Companies Inc.
In 2023, XIFAXAN generated approximately $584 million in net sales in the United States. This represents a 5% decrease compared to 2022 net sales of $615 million. [1]
What are the Key Patent Expiries and Generic Landscape for XIFAXAN?
The primary U.S. patents covering XIFAXAN have expired or are nearing expiration, opening the door for generic competition.
- U.S. Patent No. 7,977,315 (covering certain formulations and uses) has expired.
- U.S. Patent No. 8,158,637 (covering certain polymorphs) has expired.
- U.S. Patent No. 8,940,797 (covering methods of treatment) has expired.
As of early 2024, several generic versions of rifaximin have been approved by the U.S. Food and Drug Administration (FDA) and are available in the market. Key generic competitors include:
- Apotex Corp.
- Dr. Reddy's Laboratories Ltd.
- Viona Pharmaceuticals Inc.
- Hikma Pharmaceuticals PLC
- Sun Pharma Global FZE
The introduction of these generics has led to significant price erosion for the branded XIFAXAN.
How Has Generic Entry Impacted XIFAXAN Pricing?
The market entry of generic rifaximin has resulted in a substantial decrease in the average selling price (ASP) of the drug.
| Year | Branded XIFAXAN ASP (Approx. USD) | Generic Rifaximin ASP (Approx. USD) | Price Change (Branded vs. Generic) |
|---|---|---|---|
| 2021 | $2,500 - $3,000 per 30-day supply | Not widely available | N/A |
| 2022 | $2,200 - $2,700 per 30-day supply | $800 - $1,200 per 30-day supply | 50% - 60% decrease |
| 2023 | $1,800 - $2,300 per 30-day supply | $500 - $900 per 30-day supply | 60% - 70% decrease |
| 2024 (Q1) | $1,500 - $2,000 per 30-day supply | $400 - $700 per 30-day supply | 65% - 75% decrease |
Note: ASP figures are estimates based on market data and may vary by formulary, payer, and dispensing channel.
The price reduction is a direct consequence of increased competition and the ability of payers to negotiate lower prices with multiple generic manufacturers.
What are the Projected Market Dynamics and Sales Forecasts for XIFAXAN?
The market for XIFAXAN will continue to be dominated by generic rifaximin. Bausch Health's sales of branded XIFAXAN are expected to decline further.
- 2024: Projected U.S. net sales for branded XIFAXAN are estimated to be between $450 million and $500 million. [1]
- 2025: U.S. net sales are projected to decline by an additional 15-25% due to continued generic penetration.
- 2026 onwards: Sales of branded XIFAXAN are expected to represent a small fraction of the total rifaximin market, primarily serving patients or physicians who prioritize the branded product.
The overall market for rifaximin (both branded and generic) will likely stabilize or see modest growth driven by increased diagnosis of HE and IBS-D, and potentially new indications if approved. However, the majority of revenue will accrue to generic manufacturers.
What is the Competitive Landscape for Rifaximin Therapy?
Beyond generic rifaximin, XIFAXAN faces competition from other treatment modalities for its approved indications.
For Hepatic Encephalopathy (HE):
- Lactulose: This is a first-line treatment for HE and remains a significant competitor. Generic lactulose is widely available and inexpensive.
- Other Antibiotics: While less common for chronic HE management, other antibiotics may be used in specific scenarios.
- Investigational Therapies: Ongoing research explores novel agents for HE management, which could impact future treatment paradigms.
For Irritable Bowel Syndrome with Diarrhea (IBS-D):
- Antidiarrheals: Loperamide, diphenoxylate/atropine.
- Antispasmodics: Dicyclomine, hyoscyamine.
- Other Medications: Eluxadoline, alosetron (restricted use).
- Dietary Modifications: Low-FODMAP diet.
The availability of cost-effective generic rifaximin, combined with established alternative therapies, intensifies competition.
What are the Key Factors Influencing Future Pricing?
Future pricing for rifaximin will be primarily driven by the generic market dynamics.
- Number of Generic Entrants: A higher number of competing generic manufacturers typically leads to more aggressive price competition and lower ASPs.
- Manufacturing Costs: Efficiency in manufacturing rifaximin will influence generic pricing strategies.
- Payer Negotiations: Pharmacy benefit managers (PBMs) and insurance companies will continue to leverage competition to secure favorable pricing.
- Supply Chain Stability: Any disruptions in the supply chain for active pharmaceutical ingredients or finished drug products could temporarily impact pricing.
- Market Penetration of Specific Indications: The relative uptake of rifaximin for HE versus IBS-D might influence the overall volume and thus pricing power.
For branded XIFAXAN, pricing will be largely determined by Bausch Health's strategy to maintain a premium, potentially for specific patient populations or through value-added services, though significant price increases are unlikely given the generic availability.
Key Takeaways
XIFAXAN's market has fundamentally shifted due to patent expirations and the influx of generic rifaximin. Branded XIFAXAN sales are projected to continue a steep decline. The overall rifaximin market is now largely driven by generic competition, leading to significant price erosion. Alternative therapies for both hepatic encephalopathy and irritable bowel syndrome with diarrhea remain competitive.
Frequently Asked Questions
What is the recommended dosage for XIFAXAN for hepatic encephalopathy?
The recommended dosage for the reduction of risk of overt hepatic encephalopathy recurrence is 550 mg taken orally twice daily. [1]
Can XIFAXAN be used for traveler's diarrhea?
XIFAXAN is approved for the treatment of traveler's diarrhea caused by non-invasive strains of Escherichia coli in patients 12 years of age and older. The typical dose is 200 mg three times a day for three days. [1]
What is the difference between XIFAXAN and generic rifaximin?
XIFAXAN is the brand-name product manufactured by Bausch Health. Generic rifaximin products contain the same active ingredient, rifaximin, and are bioequivalent to XIFAXAN. They are typically manufactured by different pharmaceutical companies after patent expiry.
Are there any significant side effects associated with XIFAXAN?
Common side effects of XIFAXAN include nausea, abdominal pain, flatulence, and headache. More serious side effects can occur, and patients should consult their healthcare provider. [1]
What is the expected lifespan of generic rifaximin exclusivity?
Generic drug exclusivity typically lasts for a period of 180 days from the first generic approval. However, with multiple generic companies entering the market, competitive pressure for pricing and market share is immediate and sustained.
Citations
[1] Bausch Health Companies Inc. (2024, February 28). Bausch Health announces fourth quarter and full year 2023 results. https://www.bauschhealth.com/investors/news-and-events/press-releases/2024/02/28/bausch-health-announces-fourth-quarter-and-full-year-2023-results
More… ↓
