Share This Page
Drug Price Trends for XERESE
✉ Email this page to a colleague
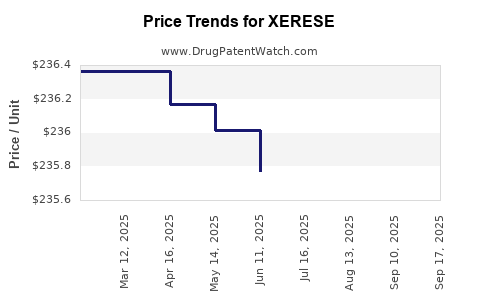
Average Pharmacy Cost for XERESE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| XERESE 5%-1% CREAM | 00187-5104-01 | 235.47700 | GM | 2025-09-17 |
| XERESE 5%-1% CREAM | 00187-5104-01 | 258.78922 | GM | 2025-09-15 |
| XERESE 5%-1% CREAM | 00187-5104-01 | 235.47700 | GM | 2025-08-20 |
| XERESE 5%-1% CREAM | 00187-5104-01 | 235.47700 | GM | 2025-07-23 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for XERESE (Carboxymethylcellulose 0.5%)
What is XERESE and its approved usage?
XERESE (carboxymethylcellulose 0.5%) is an ophthalmic solution developed by Bausch + Lomb. It is indicated for temporary relief of dryness, irritation, and discomfort due to dry eye syndrome. Approved in the United States in 2017, it aims to offer an alternative to existing artificial tears with a unique viscous formulation providing prolonged lubrication.
What is the current market landscape for dry eye treatments?
The dry eye treatment market was valued at approximately $4.7 billion in 2022, driven by increasing prevalence globally. The prevalence varies: 5-15% among adults, rising in older populations and individuals with autoimmune disorders. Key competitors include:
- Preservative-free artificial tears: brands like Systane Ultra, Refresh, and Blink Tears.
- Prescription therapies: lifitegrast (Xiidra), cyclosporine (Restasis, cequa).
- Emerging therapies: anti-inflammatory agents, novel immunomodulators.
XERESE occupies a segment within artificial tears but differentiates through its viscosity and preservative-free formulation.
How does XERESE compare to existing products?
Formulation and positioning:
- Viscosity: XERESE contains carboxymethylcellulose at 0.5%, offering prolonged retention on the ocular surface.
- Preservative profile: preservative-free, reducing risk of irritation.
- Dosing frequency: typically, 1-2 drops at intervals of 4–6 hours.
Compared to Systane Ultra (polyethylene glycol, propylene glycol-based, 0.6%) and Refresh (carboxymethylcellulose-based, 0.5%), XERESE's higher viscosity aims to extend duration of relief, potentially reducing drops per day.
What is the market potential for XERESE?
Projected sales growth:
- Bausch + Lomb reported that XERESE had a market penetration of approximately 15% of the artificial tears segment by Q4 2022.
- The total dry eye therapy segment is expected to grow at 6-8% annually through 2026.
Factors influencing growth:
- Increasing prevalence: aging populations and digital device use.
- Relief from irritation: consumers seek longer-lasting solutions.
- Preservative-free shift: audience preference favors preservative-free options.
Market share projection:
- Assuming a conservative annual growth in sales of 20-25%, XERESE could capture $300 million or more in sales globally within five years.
Geographic expansion:
- Initial focus on U.S. and European markets, with potential for penetration into Asia-Pacific markets where dry eye prevalence is rising.
How are price points projected?
Current pricing:
- In the U.S., a 10 mL bottle of XERESE retails at approximately $25–$35.
- Preservative-free artificial tears typically range from $15–$40 per 10 mL bottle**.
Pricing strategy considerations:
- Premium positioning justified by unique viscosity and preservative-free formulation.
- Potential for tiered pricing: higher for multi-dose bottles, lower for single-use.
Price projections:
- Short-term (1-2 years): Maintain current price range, with minor adjustments for inflation.
- Medium term (3-5 years): Possible price stabilization at $27–$37 per 10 mL, accounting for increased competition and market share growth.
- Long term (5+ years): Price may decrease marginally ($22–$30) as generic or biosimilar competition enters, or if larger volumes are introduced.
Impact of market dynamics:
- Increased competition from generics could lead to price erosion.
- Expansion into emerging markets might necessitate lower price points to achieve penetration.
What are key risks affecting market and price?
- Competitive pressure: Entry of generic alternatives could reduce prices.
- Pricing regulations: Price controls in certain countries could cap potential revenue.
- Market saturation: As most dry eye therapies mature, growth may slow.
- Regulatory changes: New approvals or safety concerns can impact sales.
Summary
XERESE operates in a growing, competitive dry eye treatment market. Its unique viscous formulation and preservative-free profile position it favorably. Market penetration is projected to increase steadily, with global sales potentially exceeding $300 million within five years under conservative estimates. Pricing is likely to remain within the current premium range, though competitive pressures could influence long-term price adjustments.
Key Takeaways
- Market growth driven by aging populations and digital device use; projected at 6-8% annually.
- XERESE's differentiation through viscosity and preservative-free design supports a premium price point.
- Estimated global sales may reach $300+ million in five years.
- Price points are expected to stabilize around $27–$37 per 10 mL, with potential reductions due to competition.
- Risks include generic entry, regulatory issues, and market saturation.
FAQs
Q1: What is the current market share of XERESE?
Approximately 15% within the artificial tears segment in the U.S. as of Q4 2022.
Q2: How does XERESE's price compare to competitors?
It is priced similarly to premium artificial tears, typically $25–$35 per 10 mL bottle.
Q3: What factors could drive further growth for XERESE?
Expansion into new markets, increasing dry eye prevalence, and consumer preference for preservative-free options.
Q4: How might generic competition impact XERESE?
Entry of generics could lead to a price decrease and share erosion.
Q5: Are there significant regulatory hurdles for XERESE?
Current approvals are in place; future hurdles depend on new formulations and indications.
References
[1] MarketsandMarkets, "Dry Eye Disease Market," 2022.
[2] Bausch + Lomb annual report, 2022.
[3] IQVIA, "Artificial Tears Market Overview," 2022.
[4] FDA, "Approval of XERESE," 2017.
More… ↓
