Share This Page
Drug Price Trends for XALATAN
✉ Email this page to a colleague
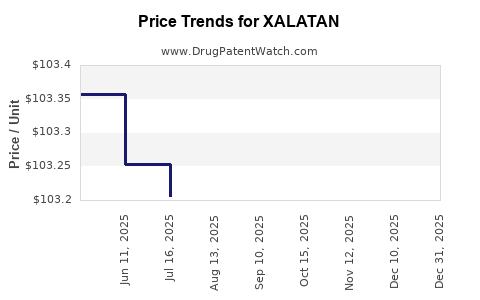
Average Pharmacy Cost for XALATAN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| XALATAN 0.005% EYE DROPS | 00013-8303-04 | 108.44342 | ML | 2026-01-01 |
| XALATAN 0.005% EYE DROPS | 58151-0419-35 | 108.44342 | ML | 2026-01-01 |
| XALATAN 0.005% EYE DROPS | 58151-0419-35 | 103.27944 | ML | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for XALATAN (Latanoprost)
What is the Market Size for XALATAN?
XALATAN (latanoprost) is a mainstay in the treatment of open-angle glaucoma and ocular hypertension. The global glaucoma medication market was valued at approximately USD 3.2 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 4.5% from 2023 to 2030. XALATAN holds a significant market share owing to its efficacy and tolerability.
Key Market Segments
- Geography: North America accounts for nearly 45% of the market, driven by high prevalence rates and healthcare spending. Europe accounts for around 25%. Asia-Pacific shows rapid growth, estimated at a CAGR of 6%, due to increasing glaucoma prevalence and healthcare infrastructure improvements.
- Demand Drivers: Rising glaucoma prevalence (approx. 80 million cases globally in 2022), aging populations, and increased awareness drive demand.
- Competitive Landscape: XALATAN’s main competitors include Travatan Z (travoprost), Lumigan (bimatoprost), and generic latanoprost products.
Market Penetration and Usage
- High-presence in ophthalmology clinics and retail pharmacies.
- Chronic nature necessitates long-term medication adherence, promoting repeated prescription cycles.
What are Price Trends and Projections for XALATAN?
Current Pricing Landscape
- Brand Name (XALATAN): Wholesale acquisition cost (WAC) ranges between USD 200 and USD 300 per bottle (2.5 mL).
- Generics: Competitors offer prices from USD 50 to USD 150 per bottle, impacting brand market share.
- rebates and discounts: Insurance coverage limits out-of-pocket costs, but premiums vary.
Price Drivers
- Patent expiration in 2018 opened the market to generics, leading to price erosion.
- Supply chain disruptions, manufacturing costs, and regulatory approvals influence prices.
- Market consolidation and payer negotiations affect actual transaction prices.
Projections (2023-2030)
| Year | Brand Price Range (USD per bottle) | Generic Price Range (USD per bottle) | Note |
|---|---|---|---|
| 2023 | 200 – 300 | 50 – 150 | Patent loss in 2018; competition risen |
| 2025 | 180 – 250 | 45 – 130 | Generic market share expands |
| 2030 | 150 – 220 | 40 – 110 | Increased generic penetration |
Future Pricing Influences
- Innovator companies may introduce improved formulations or fixed-dose combinations, potentially maintaining higher prices.
- Entry of biosimilars or new delivery mechanisms could alter price dynamics.
- Healthcare policy reforms favoring cost containment may pressure prices downward.
Implications for Stakeholders
- Pharmaceutical companies: Investments in next-gen formulations or combination therapies could sustain premium pricing.
- Payers and insurers: Cost management initiatives may shift utilization toward generic products.
- Investors: The patent cliff's impact has stabilized prices; future revenue depends on market share retention and potential innovation.
Key Takeaways
- The global glaucoma drug market is growing, with XALATAN as a leading product.
- Price erosion occurred post-patent expiration, but brand loyalty and formulation improvements support higher-than-average prices.
- Market competition, regulatory factors, and healthcare policies will influence future pricing strategies.
- Generic penetration will likely continue, driving prices downward, especially in cost-sensitive regions.
- Long-term growth hinges on demographic trends and development of new treatment modalities.
FAQs
1. When did the patent for XALATAN expire?
The patent for XALATAN expired in 2018, leading to increased generic competition.
2. How does XALATAN compare in price to its generic counterparts?
Brand XALATAN costs between USD 200 and USD 300 per bottle, whereas generics range from USD 50 to USD 150.
3. What factors can influence XALATAN’s future price?
Development of new formulations, patent protections on next-generation products, healthcare policy changes, and generic market share expansion.
4. Is there potential for new formulations or delivery methods for XALATAN?
Yes, research into sustained-release formulations and fixed-dose combinations is ongoing to improve adherence and patient outcomes.
5. How does geographic variation affect XALATAN pricing?
In North America, prices tend to be higher due to higher healthcare spending and insurance coverage; in emerging markets, lower prices prevail due to cost sensitivity and regulatory environments.
References
- MarketsandMarkets. (2023). Glaucoma drugs market. Retrieved from https://www.marketsandmarkets.com/
- IQVIA. (2022). Global ophthalmology drug market report.
- U.S. Food and Drug Administration. (2018). Patent information for ophthalmic drugs.
- Statista. (2023). Prevalence of glaucoma worldwide.
- FDA. (2022). Approved ophthalmic products and biosimilars.
More… ↓
