Drug Price Trends for WINLEVI
✉ Email this page to a colleague
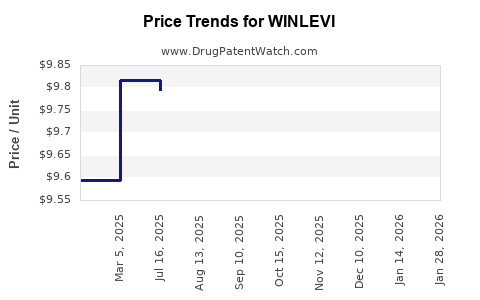
Average Pharmacy Cost for WINLEVI
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| WINLEVI 1% CREAM | 47335-0994-36 | 9.99316 | GM | 2026-01-23 |
| WINLEVI 1% CREAM | 47335-0994-36 | 9.79155 | GM | 2026-01-21 |
| WINLEVI 1% CREAM | 47335-0994-36 | 9.79422 | GM | 2025-12-17 |
| WINLEVI 1% CREAM | 47335-0994-36 | 9.79510 | GM | 2025-11-19 |
| WINLEVI 1% CREAM | 47335-0994-36 | 9.79556 | GM | 2025-10-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for WINLEVI
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| WINLEVI 1% CREAM,TOP | Sun Pharmaceutical Industries, Inc. | 47335-0994-36 | 60GM | 541.71 | 9.02850 | GM | 2022-01-17 - 2026-07-14 | FSS |
| WINLEVI 1% CREAM,TOP | Sun Pharmaceutical Industries, Inc. | 47335-0994-36 | 60GM | 395.48 | 6.59133 | GM | 2022-05-10 - 2026-07-14 | Big4 |
| WINLEVI 1% CREAM,TOP | Sun Pharmaceutical Industries, Inc. | 47335-0994-36 | 60GM | 541.71 | 9.02850 | GM | 2022-05-10 - 2026-07-14 | FSS |
| WINLEVI 1% CREAM,TOP | Sun Pharmaceutical Industries, Inc. | 47335-0994-36 | 60GM | 380.45 | 6.34083 | GM | 2023-01-01 - 2026-07-14 | Big4 |
| WINLEVI 1% CREAM,TOP | Sun Pharmaceutical Industries, Inc. | 47335-0994-36 | 60GM | 541.71 | 9.02850 | GM | 2023-01-01 - 2026-07-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
