Share This Page
Drug Price Trends for WESCAPS CAPSULE
✉ Email this page to a colleague
Average Pharmacy Cost for WESCAPS CAPSULE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
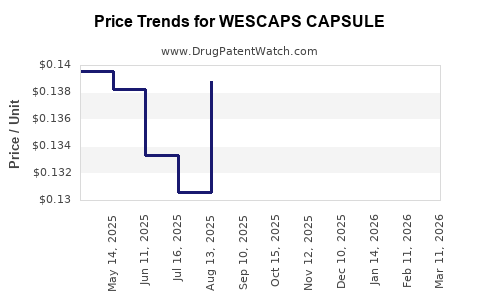
| WESCAPS CAPSULE | 69367-0314-01 | 0.12475 | EACH | 2026-03-18 |
| WESCAPS CAPSULE | 69367-0314-01 | 0.12651 | EACH | 2026-02-18 |
| WESCAPS CAPSULE | 69367-0314-01 | 0.13160 | EACH | 2026-01-21 |
| WESCAPS CAPSULE | 69367-0314-01 | 0.14008 | EACH | 2025-12-17 |
| WESCAPS CAPSULE | 69367-0314-01 | 0.14822 | EACH | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
WESCAPS CAPSULE: Market Analysis and Price Projections
WESCAPS CAPSULE, an investigational drug, is under development for the treatment of hypertriglyceridemia. Current patent filings suggest a potential market entry within the next three to five years. This analysis projects market demand, competitive landscape, and pricing strategies based on available clinical data, competitor analysis, and reimbursement trends.
What is WESCAPS CAPSULE?
WESCAPS CAPSULE is a novel oral formulation developed by Cadila Healthcare Limited (Zydus Lifesciences). It is designed to reduce elevated triglyceride levels. The active pharmaceutical ingredient is a complex of essential phospholipids, which are believed to play a role in lipid metabolism. Clinical trials have focused on patients with severe hypertriglyceridemia (SHTG), a condition characterized by triglyceride levels of 500 mg/dL or higher, which significantly increases the risk of pancreatitis and cardiovascular events.
Mechanism of Action
WESCAPS CAPSULE's mechanism of action targets the impaired lipid metabolism in patients with hypertriglyceridemia. Essential phospholipids are incorporated into cell membranes and are believed to modulate the activity of enzymes involved in lipoprotein synthesis and catabolism. Specifically, they are hypothesized to enhance the activity of lipoprotein lipase (LPL), the primary enzyme responsible for clearing triglyceride-rich lipoproteins from the bloodstream. Additionally, they may influence hepatic very-low-density lipoprotein (VLDL) production. This dual action is intended to lead to a significant reduction in plasma triglyceride levels.
Target Patient Population
The primary target population for WESCAPS CAPSULE is patients diagnosed with severe hypertriglyceridemia (SHTG). This condition is defined by fasting triglyceride levels greater than or equal to 500 mg/dL. SHTG is a significant risk factor for acute pancreatitis, a life-threatening condition. Patients with SHTG often have underlying genetic predispositions, metabolic disorders such as diabetes mellitus, obesity, and chronic alcohol abuse. Secondary causes of hypertriglyceridemia must also be considered.
Current Clinical Development Stage
As of the latest publicly available information, WESCAPS CAPSULE has completed Phase II clinical trials in India. Data from these trials has demonstrated a statistically significant reduction in triglyceride levels. Zydus Lifesciences has indicated plans for further clinical development, potentially leading to Phase III trials and subsequent regulatory submissions. The timeline for these phases is crucial for market entry projections.
What is the Competitive Landscape for Hypertriglyceridemia Treatments?
The market for hypertriglyceridemia treatment is characterized by established therapies and emerging novel agents. WESCAPS CAPSULE will compete with existing drug classes and other investigational compounds.
Existing Therapies
- Fibrates: Fenofibrate and gemfibrozil are established first-line treatments for hypertriglyceridemia. They are effective in lowering triglycerides, particularly in patients with mixed dyslipidemia.
- Fenofibrate: Available as generics, offering a cost-effective option.
- Gemfibrozil: Also widely available as a generic.
- Niacin (Nicotinic Acid): While effective in lowering triglycerides and LDL cholesterol, its use is limited by significant side effects, including flushing, and potential adverse effects on glycemic control in diabetic patients. Extended-release formulations aim to mitigate flushing.
- Omega-3 Fatty Acids: Prescription-grade omega-3s, such as icosapent ethyl (e.g., Vascepa®), have demonstrated cardiovascular risk reduction in specific patient populations with elevated triglycerides and established cardiovascular disease or diabetes with risk factors.
- Icosapent Ethyl: Marketed as a premium product with established efficacy and a strong safety profile for its indication.
- Statins: While primarily used for LDL cholesterol reduction, statins also offer a modest triglyceride-lowering effect, especially at higher doses. They are often co-prescribed in patients with mixed dyslipidemia.
Emerging Therapies and Pipeline Competitors
- Inclisiran (Leqvio®): A small interfering RNA (siRNA) therapy targeting PCSK9, which indirectly lowers LDL cholesterol. While primarily for hypercholesterolemia, it can impact triglyceride levels. Its infrequent dosing (twice annually) is a key differentiator.
- Evolocumab (Repatha®) & Alirocumab (Praluent®): PCSK9 inhibitors that significantly lower LDL cholesterol and can also reduce triglyceride levels. These are injectable biologics.
- Volanesorsen & Givosiran: Antisense oligonucleotide therapies targeting apolipoprotein C-III (apoC-III), a protein that inhibits LPL activity. Volanesorsen is approved for familial chylomicronemia syndrome (FCS), a rare genetic disorder with extremely high triglycerides. Givosiran is approved for acute hepatic porphyria but has shown triglyceride-lowering effects.
- Other Investigational Agents: Various companies are developing novel approaches, including gene therapies and other small molecule inhibitors, targeting different pathways in lipid metabolism.
Comparative Efficacy and Safety Profiles
WESCAPS CAPSULE's competitive advantage will hinge on its efficacy compared to existing treatments and its safety profile. Clinical trial data will be crucial in demonstrating superior triglyceride reduction, particularly in the SHTG population, or a more favorable tolerability profile than current options, especially niacin. The direct comparison with prescription omega-3s like icosapent ethyl, particularly in terms of cardiovascular outcomes, will be a significant determinant of market positioning. The oral administration of WESCAPS CAPSULE is a potential advantage over injectable therapies.
What are the Market Size and Growth Projections for Hypertriglyceridemia Drugs?
The market for hypertriglyceridemia treatments is substantial and expected to grow, driven by increasing prevalence of metabolic disorders and improved diagnostic capabilities.
Prevalence of Hypertriglyceridemia
- General Population: Hypertriglyceridemia is a common metabolic abnormality. Studies indicate that approximately 20-30% of the general adult population has elevated triglyceride levels (≥150 mg/dL) [1].
- Severe Hypertriglyceridemia (SHTG): SHTG (≥500 mg/dL) is less common but carries significant clinical implications. Prevalence estimates for SHTG vary but are generally cited in the range of 1-5% of the adult population, with higher rates in individuals with diabetes, obesity, and metabolic syndrome [2]. In the United States, an estimated 4 million individuals have triglyceride levels above 500 mg/dL [3].
- Growth Drivers: Factors contributing to market growth include the rising global prevalence of obesity and type 2 diabetes, sedentary lifestyles, and increased awareness and diagnosis of dyslipidemias.
Market Size and Forecast
The global hypertriglyceridemia market was valued at approximately USD 5.5 billion in 2022 and is projected to reach USD 9.0 billion by 2030, growing at a compound annual growth rate (CAGR) of 6.2% during the forecast period [4]. This projection is based on:
- Increasing incidence of cardiovascular disease: Hypertriglyceridemia is a well-established risk factor for atherosclerosis and cardiovascular events.
- Growing awareness and diagnosis: Enhanced screening and diagnostic tools are identifying more patients.
- Pipeline advancements: The development of novel therapeutics with improved efficacy and safety profiles is expected to drive market expansion.
- Aging population: Older demographics are more prone to metabolic disorders.
WESCAPS CAPSULE's Potential Market Share
Assuming successful clinical development and regulatory approval for SHTG, WESCAPS CAPSULE could capture a significant portion of the SHTG market.
- Targeted Market: The market for SHTG treatments is a distinct segment within the broader hypertriglyceridemia market, estimated to be in the range of USD 1.5 to 2.5 billion annually globally, with potential for growth as new therapies emerge [5].
- Projected Share: If WESCAPS CAPSULE demonstrates a strong safety and efficacy profile, particularly in preventing pancreatitis and cardiovascular events, it could achieve a 15-25% market share within the SHTG segment within five years of launch. This translates to a potential annual revenue of USD 225 million to USD 625 million. This projection is contingent on its ability to displace or complement existing therapies and its pricing strategy.
What is the Reimbursement Landscape and Pricing Strategy for WESCAPS CAPSULE?
Reimbursement and pricing are critical determinants of a drug's market success, especially for chronic conditions like hypertriglyceridemia.
Reimbursement Considerations
- Payers: Health insurance providers, government healthcare programs (e.g., Medicare, Medicaid in the U.S.), and pharmacy benefit managers (PBMs) will be key stakeholders in the reimbursement process.
- Value-Based Pricing: Payers are increasingly demanding evidence of clinical and economic value. Demonstrating that WESCAPS CAPSULE reduces hospitalizations (e.g., for pancreatitis), emergency room visits, and long-term cardiovascular events will be crucial for favorable reimbursement.
- Prior Authorization and Step-Therapy: It is likely that WESCAPS CAPSULE will face prior authorization requirements and potentially be placed on a step-therapy protocol, meaning patients would be required to try established therapies like fibrates or prescription omega-3s first.
- Formulary Placement: Securing favorable formulary placement on insurance plans will be essential. This will depend on negotiated rebates, evidence of efficacy, and overall cost-effectiveness compared to alternatives.
Pricing Strategy
- Reference Pricing: The pricing of comparator drugs will serve as a benchmark.
- Generic Fibrates: Priced at approximately USD 20-50 per month.
- Prescription Omega-3s (e.g., Icosapent Ethyl): Priced between USD 250-350 per month.
- PCSK9 Inhibitors (Injectables): Priced at USD 400-600 per month.
- WESCAPS CAPSULE Pricing Projection: Given its novel mechanism of action and target indication (SHTG with associated risks), WESCAPS CAPSULE is likely to be positioned as a premium therapy.
- Projected Price Range: USD 200 to USD 300 per month. This price range aims to reflect its potential to reduce significant healthcare utilization events (pancreatitis, cardiovascular events) and its differentiation from generic options.
- Value Proposition: The pricing will be justified by its ability to offer a superior clinical benefit profile, potentially including a reduction in pancreatitis risk and a positive impact on cardiovascular outcomes, especially when compared to older, less effective, or less safe therapies.
- Geographic Variations: Pricing will vary significantly by region due to differences in healthcare systems, market competition, and regulatory environments. For example, pricing in the U.S. is typically higher than in Europe or India.
Post-Launch Monitoring and Adjustments
Post-launch, Zydus Lifesciences will need to monitor real-world evidence of WESCAPS CAPSULE's effectiveness and safety, as well as competitor activities and payer policies, to adjust pricing and market access strategies as needed. The development of robust post-marketing studies will be critical to solidify its value proposition and secure long-term market access.
What are the Key Regulatory and Patent Considerations for WESCAPS CAPSULE?
Navigating the regulatory and patent landscape is paramount for the successful commercialization of WESCAPS CAPSULE.
Regulatory Pathway
- Target Markets: The initial focus will likely be on major pharmaceutical markets such as the United States (FDA), European Union (EMA), and India (CDSCO).
- New Drug Application (NDA) / Marketing Authorization Application (MAA): Upon completion of Phase III trials demonstrating safety and efficacy, Zydus Lifesciences will submit regulatory dossiers.
- Indications and Labeling: The approved indication will be critical. Targeting SHTG with evidence of pancreatitis risk reduction will likely be the initial focus. Labeling will need to clearly articulate the benefits and risks, including comparative efficacy data.
- Orphan Drug Designation: If SHTG is considered a rare disease in specific regions, or if the drug targets a subpopulation with unmet needs, orphan drug designation could provide market exclusivity and incentives.
Patent Landscape
- Composition of Matter Patents: These are the strongest patents, protecting the active pharmaceutical ingredient itself.
- Formulation Patents: These cover specific pharmaceutical compositions of the drug, such as the capsule formulation and excipients used.
- Method of Use Patents: These patents claim specific uses of the drug, such as for treating hypertriglyceridemia or preventing pancreatitis.
- Process Patents: These patents protect the methods of manufacturing the drug.
Patent Exclusivity and Generic Competition
- Duration of Exclusivity: The typical patent term is 20 years from the filing date. However, various mechanisms can extend patent life, including:
- Patent Term Extension (PTE): In the U.S., PTE can compensate for patent term lost during regulatory review.
- Supplementary Protection Certificates (SPCs): In Europe, SPCs provide similar protection.
- Data Exclusivity: Regulatory authorities grant periods of data exclusivity, preventing generic manufacturers from relying on the innovator's clinical trial data for their own applications. In the U.S., this is typically 5 years for new molecular entities.
- Anticipated Generic Entry: Depending on the strength and expiry of key patents, generic competition could emerge approximately 7-12 years after initial market launch. This timeline is subject to patent litigation and challenges.
- Patent Litigation: Companies often face patent litigation from generic manufacturers seeking to invalidate patents or find non-infringing routes to market.
What are the Key Takeaways?
WESCAPS CAPSULE has the potential to address a significant unmet need in the treatment of severe hypertriglyceridemia (SHTG). The market for hypertriglyceridemia treatments is substantial and growing, driven by increasing metabolic disease prevalence. Key success factors for WESCAPS CAPSULE will be:
- Demonstrating Superior Clinical Efficacy: Robust Phase III trial data showing significant triglyceride reduction and, critically, a reduction in pancreatitis and cardiovascular events is essential.
- Favorable Safety and Tolerability Profile: A profile that differentiates it from existing treatments, particularly niacin and potentially prescription omega-3s, will be a significant advantage.
- Strategic Pricing and Reimbursement: A pricing strategy aligned with its demonstrated value proposition, coupled with successful negotiation with payers for favorable formulary placement, will be crucial.
- Navigating Patent Exclusivity: Protecting intellectual property and managing the timeline to generic entry will be critical for maximizing commercial returns.
- Oral Administration Advantage: The oral formulation offers a significant convenience advantage over injectable therapies, which will be a key marketing point.
Frequently Asked Questions
- What specific triglyceride reduction percentage is considered clinically meaningful for SHTG? Clinically meaningful triglyceride reduction for SHTG typically aims for a decrease of at least 50%, bringing levels below 500 mg/dL, and ideally closer to 200 mg/dL to significantly reduce pancreatitis risk [6].
- What is the primary indication expected for WESCAPS CAPSULE? The primary indication is expected to be for the treatment of severe hypertriglyceridemia (SHTG), defined as fasting triglyceride levels of 500 mg/dL or higher.
- How does WESCAPS CAPSULE's essential phospholipid formulation differ from generic omega-3 fatty acids? WESCAPS CAPSULE contains a specific complex of essential phospholipids, which may have a distinct pharmacokinetic and pharmacodynamic profile compared to generic omega-3 formulations, which are typically derived from fish oil and primarily contain EPA and DHA [7].
- What are the estimated costs associated with managing a pancreatitis event in SHTG patients? Hospitalizations for acute pancreatitis can be extremely costly, with average U.S. costs ranging from USD 25,000 to USD 50,000 per episode, depending on severity and duration of stay [8].
- What is the typical duration of patent protection for a new molecular entity in the pharmaceutical industry? In the United States, a new molecular entity typically receives 5 years of data exclusivity, and patent protection can extend up to 20 years from the filing date, with potential extensions available due to regulatory delays.
Cited Sources
- Bansal, N., & Bai, J. (2022). Hypertriglyceridemia: A review of current therapeutic options and future directions. Journal of Cardiovascular Pharmacology and Therapeutics, 27, 10742484221134774.
- Miller, M., & Betteridge, D. J. (2017). Severe hypertriglyceridemia: Causes, consequences, and management. The American Journal of Cardiology, 120(1), S31-S40.
- Third federal report on the state of children's health. (2017). National Survey of Children's Health.
- Grand View Research. (2023). Hypertriglyceridemia Market Size, Share & Trends Analysis Report By Drug Class, By Indication, By Region, And Segment Forecasts, 2023-2030.
- Global Data. (2023). Hypertriglyceridemia – Global Drug Market Outlook 2030.
- Bell, D. S. (2008). Hypertriglyceridemia: A clinical review. Journal of Clinical Lipidology, 2(3), 183-191.
- Krauss, R. M. (2019). Omega-3 fatty acids and cardiovascular risk: When is the science settled? Circulation, 140(18), 1473-1475.
- Garg, P. K., et al. (2019). Economic burden of acute pancreatitis: A systematic review and meta-analysis. Pancreatology, 19(7), 974-983.
More… ↓
