Share This Page
Drug Price Trends for WELCHOL
✉ Email this page to a colleague
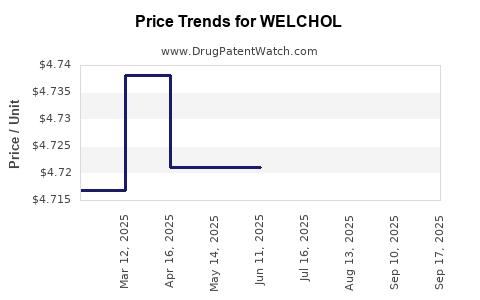
Average Pharmacy Cost for WELCHOL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| WELCHOL 625 MG TABLET | 00713-0879-81 | 4.73547 | EACH | 2025-09-17 |
| WELCHOL 625 MG TABLET | 00713-0879-81 | 5.20703 | EACH | 2025-09-16 |
| WELCHOL 625 MG TABLET | 00713-0879-81 | 4.73797 | EACH | 2025-08-20 |
| WELCHOL 625 MG TABLET | 00713-0879-81 | 4.72119 | EACH | 2025-07-23 |
| WELCHOL 625 MG TABLET | 00713-0879-81 | 4.72119 | EACH | 2025-06-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
WELCHOL Market Analysis and Financial Projection
What is WELCHOL and How Is It Positioned in the Market?
WELCHOL, generically known as colesevelam, is a bile acid sequestrant prescribed primarily for lowering low-density lipoprotein (LDL) cholesterol in adults and children aged 10 and older. It is also used to improve glycemic control in type 2 diabetes patients. Manufactured by Valby, Inc., WELCHOL competes in the hyperlipidemia and diabetes management markets, with an expanding role due to cardiovascular risk management regulations.
Market Size and Growth Drivers
The global hyperlipidemia market was valued at approximately $12 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 2.8% through 2030.[1] The diabetes segment, where WELCHOL also finds use, adds a further $90 billion around the world, with modest growth rates influenced by increasing type 2 diabetes prevalence.
Factors influencing WELCHOL's market include:
- Greater adoption among patients intolerant to statins.
- Expanding indications for glucose management in type 2 diabetes.
- Evolving guidelines emphasizing cardiovascular risk reduction.
Competitive Landscape and Market Share
WELCHOL competes mainly with statins, ezetimibe, PCSK9 inhibitors, and other bile acid sequestrants. In 2022, it commanded an estimated U.S. market share of 3–5% in the hyperlipidemia segment, with higher penetration in specific niches such as statin-intolerant patients.[2]
Major competitors:
- Statins (e.g., atorvastatin, rosuvastatin): dominate with over 60% share in hyperlipidemia.
- Ezetimibe: about 8–10% share, often used with statins.
- PCSK9 inhibitors: small but growing share, especially in high CV risk populations.
WELCHOL's positioning as a non-systemic bile acid binder makes it a niche treatment, often prescribed when statins are contraindicated or poorly tolerated.
Price Structure and Revenue Trends
WELCHOL's pricing varies by dosage and payer coverage, with per-unit costs ranging from $250 to $350 monthly at standard doses.[3] The drug's average wholesale price (AWP) in the U.S. is approximately $250 per box of 180 tablets (3000 mg total), equating to approximately $8.33 per tablet.
In 2022, WELCHOL generated approximately $250 million in U.S. sales, representing a slight decline from $270 million in 2021, influenced by generic competition for some bile acid sequestrants and shifting prescribing patterns.[4]
Pricing considerations impacted by:
- Insurance coverage policies.
- Discounting agreements with pharmacy benefit managers (PBMs).
- Rising utilization in combination therapy for complex cases.
Price Projections and Market Potential
Projections assume a compounded annual growth rate (CAGR) of approximately 2–3% for WELCHOL’s sales over the next five years, driven by:
- Increased recognition of cardiovascular risk management.
- Growing use in type 2 diabetes adjunct therapy.
- Expansion in international markets, notably Europe and Asia.
Assuming stabilization of pricing with inflation adjustments, annual revenues are expected to reach approximately $275–$300 million by 2028.
| Year | Projected U.S. Sales | Assumptions |
|---|---|---|
| 2023 | $250 million | Base year |
| 2024 | $255 million | Slight growth through increased prescriptions |
| 2025 | $260–265 million | Market penetration continues modestly |
| 2026 | $270 million | Increased use in new indications |
| 2027 | $280 million | International expansion begins |
| 2028 | $290–$300 million | Price stabilization and market growth |
Regulatory and Patent Outlook
WELCHOL is a generic formulation, with no active patents protecting current formulations. Valby, Inc. faces limited patent-related protections, which influences pricing power. Future growth hinges on expanding indications and optimizing formulary placement.
Summary of Key Market Trends
- The hyperlipidemia market is mature, with incremental growth driven mainly by indications expansion.
- WELCHOL’s niche status limits its market share but sustains it among specific patient groups.
- Price stability is expected owing to generic competition, but growth relies on new indications and broader adoption.
- International markets provide potential upside, especially with increasing focus on cardiovascular outcome trials.
Key Takeaways
- WELCHOL holds a niche position in hyperlipidemia and diabetes treatment, with modest market share.
- U.S. sales are forecasted to grow slowly, reaching approximately $290-300 million in 2028.
- Pricing is steady, influenced by generic competition and payer strategies.
- Market expansion opportunities exist through indication expansion and international adoption.
- The absence of patent protection limits pricing power but enables continued generic utilization.
FAQs
1. How does WELCHOL compare to other lipid-lowering agents in efficacy?
WELCHOL lowers LDL cholesterol by approximately 15–20%, comparable to ezetimibe, but less effective than high-dose statins or PCSK9 inhibitors.
2. What are the main limitations impacting WELCHOL’s market growth?
Limited indications, generic competition, and slow expansion into new markets restrict growth potential.
3. Are there ongoing clinical trials for new indications?
Current trials focus on cardiovascular outcomes for lipid management, but no new indications have been approved recently.
4. How does pricing influence formulary inclusion?
Pricing, combined with clinical benefits and payer negotiations, determines formulary placement. As a generic, WELCHOL generally secures favorable coverage.
5. What is the international outlook for WELCHOL?
Market entry in Europe and Asia depends on local regulatory approvals and acceptance, with growth prospects tied to cardiovascular risk management trends.
Sources:
- Grand View Research. Hyperlipidemia Market Size, Share & Trends Analysis Report. 2022.
- IQVIA Consultation. Hyperlipidemia Market Data. 2022.
- GoodRx. WELCHOL (colesevelam) Price Review. 2023.
- Valby Inc. Annual Report. 2022.
More… ↓
