Share This Page
Drug Price Trends for WARFARIN SODIUM
✉ Email this page to a colleague
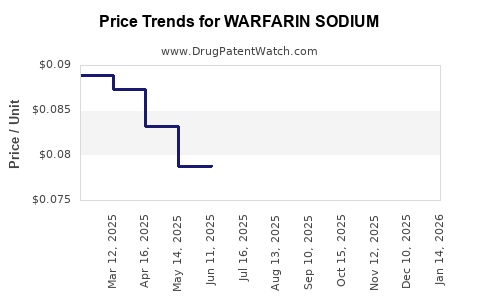
Average Pharmacy Cost for WARFARIN SODIUM
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| WARFARIN SODIUM 10 MG TABLET | 76282-0335-01 | 0.10397 | EACH | 2026-04-22 |
| WARFARIN SODIUM 7.5 MG TABLET | 76282-0334-01 | 0.08892 | EACH | 2026-04-22 |
| WARFARIN SODIUM 6 MG TABLET | 76282-0333-10 | 0.10107 | EACH | 2026-04-22 |
| WARFARIN SODIUM 6 MG TABLET | 76282-0333-01 | 0.10107 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Warfarin Sodium
What is the Current Market Situation for Warfarin Sodium?
Warfarin Sodium is an oral anticoagulant used to prevent and treat thromboembolic events, such as stroke and deep vein thrombosis. It is one of the oldest anticoagulants still in use, with a market presence spanning over 60 years. The drug remains widely prescribed globally, particularly in countries with established healthcare systems.
Global demand is driven by the increasing prevalence of cardiovascular diseases and venous thromboembolism. The drug's low cost and extensive clinical experience sustain its high prescribing rates, despite the emergence of newer anticoagulants.
In 2022, the global warfarin market was valued around USD 580 million, with annual growth estimated at roughly 2.5% due to increasing cardiovascular incidents worldwide [1].
How Does the Manufacturing Landscape Affect Price Structures?
Major manufacturers include Mylan, Pfizer, and Hexal. Patent expirations, notably in the U.S., have facilitated the entry of generic competitors, leading to decreased prices. As of 2023, more than 20 generic formulations are available globally, with significant price reductions.
Generic warfarin's manufacturing costs are low due to its long-established synthesis routes and generic production scale. This should keep prices relatively stable unless supply chain disruptions occur.
What are the Price Dynamics and Trends?
Current Pricing (2023)
| Region | Typical Wholesale Price per 30 mg Tablet | Market Notes |
|---|---|---|
| U.S. | USD 0.05 - 0.08 | Wide generic availability, price erosion |
| Europe | EUR 0.02 - 0.06 | Similar trends, regional variations |
| Asia | USD 0.01 - 0.03 | Lower cost, growing production capacity |
Historical Trends
Since patent expiration in 2007 in many markets, prices have declined sharply. The trend continues with annual average decreases of 2-3% per year, aligned with generic competition and manufacturing efficiencies.
Price Drivers
- Regulatory policies: Stringent quality standards can influence manufacturing costs.
- Supply chain stability: Disruptions can temporarily elevate prices.
- Market penetration: Increased adoption in emerging markets can influence regional pricing.
What are Future Price Projections?
Based on current trends, prices for generic warfarin are projected to remain stable or slightly decline over the next five years, with annual decreases around 1-2% driven by ongoing generic competition.
Innovative formulations or delivery methods are unlikely to significantly impact pricing due to the drug's longstanding generic status. Price fluctuations expected are primarily due to supply chain factors and regional policy changes.
Projected Price Range (2028)
| Region | Estimated Wholesale Price per 30 mg Tablet |
|---|---|
| U.S. | USD 0.03 - 0.05 |
| Europe | EUR 0.01 - 0.04 |
| Asia | USD 0.005 - 0.02 |
What are Market Barriers and Opportunities?
- Barriers: Stringent regulatory approval processes in some markets, supply chain vulnerabilities, and the emergence of direct oral anticoagulants (DOACs) reducing warfarin prescriptions.
- Opportunities: Cost-sensitive healthcare systems favor warfarin; increased use in developing countries; potential for biosimilar entry if regulatory pathways are optimized.
How do Competitive Dynamics Impact Pricing?
Market concentration remains relatively low, with multiple generic manufacturers lowering prices. Key players have cut prices aggressively post-patent expiry, ensuring low-cost options persist. The entry of biosimilars is not applicable to warfarin due to its small molecule status.
Conclusions
Warfarin Sodium maintains a low-price profile with competitive dynamics favoring further reductions. Market growth remains steady, supported by global aging populations and increased cardiovascular care. Price stability is expected barring supply disruptions or regulatory changes.
Key Takeaways
- The global warfarin market was valued at USD 580 million in 2022, with steady growth.
- Prices for generics have declined significantly post-patent expiry, averaging USD 0.05 - 0.08 per 30 mg tablet in the U.S.
- Future price declines are modest, around 1-2% annually, with regional variations.
- Supply chain and regulatory factors predominantly influence short-term pricing fluctuations.
- Market opportunities remain in emerging markets and cost-sensitive healthcare environments.
FAQs
1. What are the main factors influencing warfarin pricing?
Market competition, manufacturing costs, regulatory policies, and supply chain stability.
2. How does warfarin differ from newer anticoagulants in cost?
Warfarin is significantly cheaper due to its generic status and longstanding manufacturing, while newer agents (e.g., dabigatran, rivaroxaban) cost several times more.
3. Will warfarin prices increase with new formulations?
Unlikely, as existing formulations dominate, and innovation does not significantly affect pricing for a generic compound.
4. What regional differences impact warfarin prices?
Pricing is lower in regions with multiple generic options and less regulatory burden, such as Asia, compared to the U.S. and Europe.
5. Can supply chain issues raise warfarin prices?
Yes, disruptions in sourcing raw materials or manufacturing bottlenecks can cause temporary price hikes.
References
- Industry Reports. (2023). Global anticoagulant drug market report. MarketResearch.com.
More… ↓
