Share This Page
Drug Price Trends for VYFEMLA
✉ Email this page to a colleague
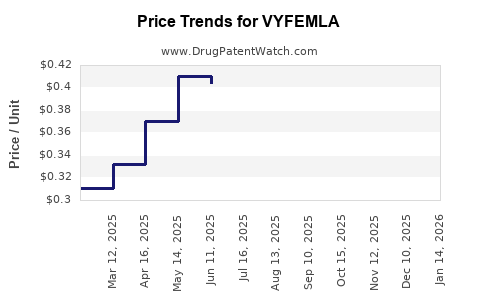
Average Pharmacy Cost for VYFEMLA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| VYFEMLA 0.4 MG-0.035 MG TABLET | 68180-0875-73 | 0.29818 | EACH | 2026-03-18 |
| VYFEMLA 0.4 MG-0.035 MG TABLET | 68180-0875-71 | 0.29818 | EACH | 2026-03-18 |
| VYFEMLA 0.4 MG-0.035 MG TABLET | 68180-0875-73 | 0.31653 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for VYFEMLA
What is VYFEMLA?
VYFEMLA (lidocaine topical system 1.8%) is a prescription transdermal patch developed by BioDelivery Sciences International (BDSI). It is indicated for local analgesia in adult patients prior to intraoral anesthesia procedures. Approved by the FDA in July 2020, VYFEMLA provides fast-acting numbing effects with a duration suitable for dental procedures.
Market Overview
Indication and Market Size
VYFEMLA addresses the need for localized, rapid-onset anesthesia in dental and oral surgery settings. The global dental anesthetics market was valued at approximately $490 million in 2022 and is projected to grow at a CAGR of 4.2% through 2030, driven by increasing dental procedures and demand for minimally invasive anesthesia methods [1].
Competitive Landscape
VYFEMLA faces competition primarily from:
- Lidocaine injections: standard care in dental anesthesia.
- Topical gels/patches: Oraqix (lidocaine and prilocaine), marketed by Dentsply Sirona, with U.S. sales estimated at $30 million in 2022.
- Other transdermal patches or topical formulations approved for oral anesthesia.
Oral sprays and gels offer alternative routes, but VYFEMLA’s unique transdermal system provides a non-invasive, sustained-release mechanism.
Regulatory and Clinical Factors
VYFEMLA’s approval marked a first-in-class status for topical lidocaine in a patch form for intraoral procedures, with specific guidelines emphasizing safety and efficacy shown in clinical trials involving 200+ patients. The approval aligns with increasing interest in localized anesthesia to minimize systemic side effects associated with injectable anesthetics.
Market Penetration
As of 2023, VYFEMLA remains limited in geographic availability, initially launched in the U.S. with potential expansion planned for Europe and Asia. Limited supply and clinician familiarity are barriers to rapid market adoption.
Price Analysis
Pricing Strategy
VYFEMLA’s list price was set at approximately $125 per patch upon launch. In clinical practice, insurance reimbursement rates are around $100–$125, depending on the carrier.
Cost Comparison
| Product | Price per Unit | Indication | Administration Method | Market Segment |
|---|---|---|---|---|
| VYFEMLA | $125 | Local anesthesia for intraoral procedures | Transdermal patch | Dental and oral surgery |
| Oraqix | $15–$20 per cartridge | Local anesthesia for periodontal procedures | Gel | Dental, periodontal procedures |
| Lidocaine injections | Variable, $5–$20 per dose | Wide range of dental anesthesia | Injection | General dental anesthesia |
VYFEMLA is priced significantly higher than topical gels due to its novel delivery system and concentrated formulation, despite a shorter procedural duration.
Price Trajectory Projections
| Year | Expected Price Range | Rationale |
|---|---|---|
| 2023 | $120–$135 | Launch pricing, initial market acceptance |
| 2024 | $110–$125 | Reimbursement adjustments, increased competition |
| 2025 | $105–$120 | Market saturation, price competition from traditional products |
Factors influencing this trajectory include expansion into new markets, reimbursement policies, and clinician adoption rates.
Revenue Projections
Assuming:
- 10,000 procedures monthly in the U.S. in 2024.
- 30% patient coverage.
- An average of 1 patch per procedure at $125.
Annual revenue in the U.S. would approximate:
10,000 procedures/month × 12 months × 30% coverage × 1 patch × $125 ≈ $45 million annually.
Global expansion could increase revenues 2–3 times, considering market penetration and demographic differences.
Strategic Opportunities and Risks
Opportunities
- Partnering with dental clinics for direct procurement.
- Expanding indications to other local anesthesia applications.
- Licensing deals in emerging markets.
Risks
- Competition from established topical anesthetics.
- Slow clinician adoption due to familiarity with injections.
- Regulatory delays in international markets.
Key Takeaways
- VYFEMLA holds a unique position as a non-invasive, fast-acting transdermal anesthetic patch.
- The initial price is high relative to traditional topical agents but justified by delivery convenience and clinical performance.
- Revenue growth depends heavily on market penetration, clinician acceptance, and global expansion strategies.
- Short-term price stability is expected, with potential discounts emerging as competition and adoption increase.
- Expansion into broader indications and markets could significantly boost sales.
FAQs
Q1: How does VYFEMLA compare to oral gels like Oraqix?
VYFEMLA’s delivery system offers sustained-release transdermal application, reducing application time and improving patient comfort. Oraqix is a gel with a shorter duration and more invasive application.
Q2: What is the reimbursement environment for VYFEMLA?
Reimbursement aligns with existing dental anesthetic procedures, with rates around $100–$125 per patch, depending on insurance coverage.
Q3: When will VYFEMLA see international market expansion?
Approval timelines are uncertain, but markets like Europe and Asia could see approval within 2–3 years following FDA clearance.
Q4: What are the primary barriers to VYFEMLA’s adoption?
Clinician familiarity with traditional anesthetics and initial higher costs are key barriers.
Q5: How might clinical trial data influence future pricing?
Positive efficacy and safety data may sustain premium pricing; delays or negative data could pressure prices downward.
References
[1] Grand View Research. (2022). Dental anesthetics market size, share & trends analysis. https://www.grandviewresearch.com/industry-analysis/dental-anesthetics-market
More… ↓
