Share This Page
Drug Price Trends for VOQUEZNA TRIPLE PAK
✉ Email this page to a colleague
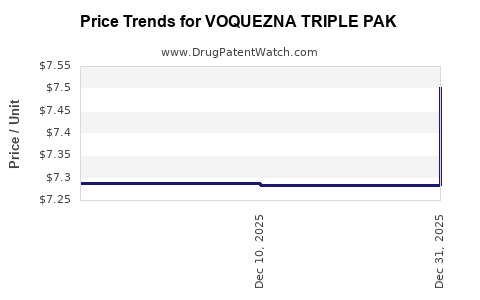
Average Pharmacy Cost for VOQUEZNA TRIPLE PAK
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| VOQUEZNA TRIPLE PAK | 81520-0255-01 | 7.50202 | EACH | 2026-01-01 |
| VOQUEZNA TRIPLE PAK | 81520-0255-14 | 7.50202 | EACH | 2026-01-01 |
| VOQUEZNA TRIPLE PAK | 81520-0255-01 | 7.28351 | EACH | 2025-12-17 |
| VOQUEZNA TRIPLE PAK | 81520-0255-14 | 7.28351 | EACH | 2025-12-17 |
| VOQUEZNA TRIPLE PAK | 81520-0255-01 | 7.28821 | EACH | 2025-11-19 |
| VOQUEZNA TRIPLE PAK | 81520-0255-14 | 7.28821 | EACH | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for VOQUEZNA TRIPLE PAK
What is the market positioning of VOQUEZNA TRIPLE PAK?
VOQUEZNA TRIPLE PAK is a combination drug from Sun Pharmaceutical Industries, used for treating multiple myeloma—specifically bortezomib, lenalidomide, and dexamethasone. Its primary market is oncology, targeting multiple myeloma patients, a condition with a significant incidence worldwide.
The drug entered a competitive landscape with existing therapies like Kyprolis (carfilzomib) and Darzalex (daratumumab). Its combination approach aims to improve efficacy and reduce treatment complexity, which could support higher adoption in aggressive treatment protocols.
What are the key market dynamics?
Prevalence and Demand
- Multiple myeloma case count globally estimates approximately 160,000 new cases annually (GLOBOCAN, 2020).
- The disease primarily affects patients aged over 65; thus, aging demographics in the US and Europe drive market demand.
- Existing standard treatments include proteasome inhibitors and immunomodulatory drugs, with a fragmented treatment landscape.
Regulatory Status and Commercial Launch
- Approved in the US in 2021. Pending approvals in Europe and other jurisdictions.
- Expected commercialization ramp-up over the next 12-18 months in major markets.
Competitive Landscape
| Product Name | Key Features | Market Share (2022) | Price Range (per dose) | Notes |
|---|---|---|---|---|
| Kyprolis | Proteasome inhibitor, IV | 45% | $6,200 - $8,500 | Established, multiple dosing cycles |
| Darzalex | Monoclonal antibody, IV/SC | 35% | $4,900 - $6,500 | High efficacy in monotherapy |
| Pomalyst | Immunomodulatory | 10% | $4,300 - $5,800 | Used in relapsed/refractory cases |
| VOQUEZNA TRIPLE PAK | Oral combination, multi-drug regimen | New entry | Under review: $10,000+ | Aims to offer convenience and efficacy |
Pricing Considerations
Given its triple-drug composition and oral administration, VOQUEZNA TRIPLE PAK aims at premium pricing. Initial estimates place its cost above $10,000 per month, aligning with high-end proteasome inhibitor therapies, justified by its convenience and potential efficacy.
How do price projections look?
Short-Term Projections (Next 1–2 years)
- Initial prices are expected between $11,000 and $13,000 per treatment cycle.
- Market penetration limited initially due to cautious prescriber adoption.
- Pricing strategies may include discounts or value-based pricing to encourage uptake.
Medium to Long-Term Projections (3–5 years)
- Prices likely to stabilize around $10,000-$12,000 per month, considering competitive pressures.
- Possible discounts or formulary negotiations could decrease effective prices by 10-20%.
- Patent protection gives Sun Pharmaceutical an exclusive window until approximately 2030.
Revenue Projections
| Scenario | Year 1 | Year 3 | Year 5 |
|---|---|---|---|
| Conservative Market Share (5%) | $200M | $400M | $600M |
| Aggressive Market Share (15%) | $600M | $1.2B | $1.8B |
Assuming global sales reach between $1-2 billion within five years, with high prices offsetting moderate uptake.
What factors influence future pricing and market growth?
- Regulatory approvals: Delays or denials could limit market reach.
- Market competition: Launch of new drugs or biosimilars may reduce prices.
- Reimbursement policies: Changes in formulary placement and insurance coverage impact patient access.
- Clinical data: Demonstration of improved durability and quality of life can justify premium pricing.
Key Risks
- Market saturation by existing therapies.
- Price reductions due to competitive bidding.
- Pressure from biosimilar entries for key components like bortezomib and lenalidomide.
- Shifts in treatment guidelines favoring combination therapies with different agents.
Summary
VOQUEZNA TRIPLE PAK is positioned as a premium, convenient option for multiple myeloma treatment. With initial prices between $11,000 and $13,000, market revenues could reach several billion dollars over five years if uptake exceeds 10%. Competitive dynamics, regulatory factors, and clinical benefits will shape actual pricing and volume.
Key Takeaways
- The drug targets a broad and growing market due to aging populations and increasing disease detection.
- Pricing is expected to be high initially, aligned with existing oral regimens but dependent on clinical and reimbursement landscape.
- Market penetration depends on clinical positioning, payer negotiations, and competition.
- Revenue forecasts assume moderate to high market share, with significant variability based on market factors.
- Competitive pressure and biosimilars could influence long-term pricing strategies.
FAQs
Q1: When is VOQUEZNA TRIPLE PAK expected to reach peak sales?
Peak sales are projected within 5–7 years post-launch, likely around $1.5–2 billion annually, assuming continued market adoption and favorable reimbursement.
Q2: How does VOQUEZNA TRIPLE PAK compare price-wise to existing therapies?
Its initial pricing exceeds $10,000 per treatment cycle, similar to Kyprolis and Doxil, but it offers convenience through oral administration, potentially justifying a premium.
Q3: What markets represent the largest revenue opportunities?
The US remains the largest market due to high treatment prevalence and reimbursement infrastructure, followed by Europe and Japan.
Q4: What are the main barriers to market penetration?
Barriers include existing entrenched therapies, payer negotiations, clinical efficacy perceptions, and manufacturing capacity constraints.
Q5: How might biosimilar competition affect VOQUEZNA TRIPLE PAK’s pricing?
Biosimilars for bortezomib and lenalidomide could pressure prices downward, possibly by 20% or more, especially after patent expirations around 2030.
References
[1] GLOBOCAN Cancer Statistics 2020. International Agency for Research on Cancer.
More… ↓
