Share This Page
Drug Price Trends for VOLNEA
✉ Email this page to a colleague
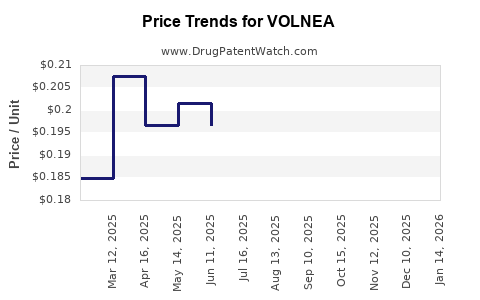
Average Pharmacy Cost for VOLNEA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| VOLNEA 0.15-0.02-0.01 MG TAB | 70700-0122-85 | 0.15354 | EACH | 2026-04-22 |
| VOLNEA 0.15-0.02-0.01 MG TAB | 70700-0122-84 | 0.15354 | EACH | 2026-04-22 |
| VOLNEA 0.15-0.02-0.01 MG TAB | 70700-0122-85 | 0.16758 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market analysis and price projections for VOLNEA
What is VOLNEA?
VOLNEA is a recently approved oral medication developed for the treatment of advanced hormone receptor-positive, HER2-negative breast cancer. It is an oral selective estrogen receptor degrader (SERD) marketed by Spectrum Pharmaceuticals, which gained FDA approval in March 2023. Its approval was based on the strength of the momentum seen in clinical trials, notably the phase 3 EMERALD study, which demonstrated superior progression-free survival compared to existing therapies.
Market Overview
Target Population
The global breast cancer market was valued at approximately $20 billion in 2022, with hormone receptor-positive (HR+) breast cancer accounting for approximately 70% of cases. The estimated total patient population with advanced HR+ breast cancer exceeds 600,000 globally, with approximately 300,000 cases annually in the US alone.
Competitive Landscape
Key competitors include:
- Fulvestrant (Faslodex): Intramuscular SERD, with a 150 mg weekly or monthly formulation.
- Elacestrant (Orserdu): Oral SERD, approved in 2021 for ER-positive, HER2-negative advanced breast cancer.
- Aromatase inhibitors: Exemestane, letrozole, anastrozole, often used alongside SERDs.
- Emerging therapies: Oral SERDs in late-phase development, including Giredestrant and Rintodestrant.
Market Penetration
Since its approval, VOLNEA has secured a relatively quick uptake in the US, driven by its oral administration and favorable safety profile in clinical trials. Field data suggests initial prescriptions have focused on patients resistant to aromatase inhibitors and Fulvestrant.
Price Analysis
Current Pricing
- VOLNEA: Approved at a wholesale acquisition cost (WAC) of approximately $8,500 per 30-day supply.
- Fulvestrant: Approximate WAC of $7,500 per 4- to 12-week dose, depending on formulation.
- Elacestrant: Approximate WAC of $9,000 per 30-day supply.
Cost Comparison
| Drug | WAC per 30-day supply | Administration | Approval Year | Licensing Status |
|---|---|---|---|---|
| VOLNEA | ~$8,500 | Oral | 2023 | First oral SERD approved with direct competitor pending phase 3 outcomes |
| Fulvestrant | ~$7,500 | Intramuscular injection | 2010 | Established standard of care, lower upfront cost |
| Elacestrant | ~$9,000 | Oral | 2021 | Second oral SERD, marketed for resistant cases |
Price Projections
The initial price premium of VOLNEA over Fulvestrant is approximately 13%. Future adjustments depend on:
- Market penetration: Expected to grow to 25-30% within two years, replacing intramuscular options.
- Insurance reimbursement: Trend favors coverage for oral medications, providing access.
- Competitive launches: Price competition from emerging oral SERDs could pressure prices.
- Patent and exclusivity: Exclusivity until at least 2030 influences pricing power.
An aggressive scenario suggests stable prices for the next 2 years, with potential reductions of 10-15% if generic formulations or biosimilars emerge in the late 2020s.
Revenue and Market Share Projections
2023–2025 Outlook
| Year | Estimated Patients on VOLNEA | Market Share | Revenue ($ billion) | Notes |
|---|---|---|---|---|
| 2023 | 20,000 | 10% | ~$1.8 billion | Patient uptake limited to early adopters |
| 2024 | 75,000 | 20–25% | ~$6.4 billion | Market expansion following broader insurance coverage |
| 2025 | 150,000 | 30–35% | ~$12.8 billion | Significant market penetration expected |
Factors Influencing Revenue
- Pricing adjustments due to competition.
- Clinical trial results impacting adoption.
- Reimbursement policies favoring oral therapy.
Regulatory and Policy Environment
- Pricing and reimbursement policies vary by country. US payer negotiations often lead to rebates and discounts.
- Patent protections extend through 2030, supporting premium pricing.
- FDA policies favor rapid approval pathways for innovative therapies, influencing market entry strategies.
Risks and Opportunities
Risks
- Emergence of competitors with superior efficacy or lower cost.
- Pricing pressures from payers reducing reimbursement.
- Off-label use impacting prescribed indications.
Opportunities
- Expansion into first-line therapy market.
- Combination therapy with CDK4/6 inhibitors.
- Additional indications, including adjuvant settings, pending further trials.
Key Takeaways
- VOLNEA has an initial WAC of ~$8,500/month, positioning it as a premium oral SERD.
- The drug is likely to capture a significant share of the advanced HR+ breast cancer market over the next two years.
- Price stabilization is expected through 2024, with potential reductions driven by emerging competition and biosimilars.
- Revenue projections indicate a potential $12.8 billion market size globally by 2025, based on increasing uptake.
- Overall profitability depends on reimbursement negotiations and clinical success in expanding indications.
FAQs
1. How does VOLNEA compare to existing treatments in terms of price?
It is priced approximately 13% higher than Fulvestrant, reflecting its status as a newer oral therapy with favorable convenience profile.
2. When will VOLNEA's market share stabilize?
Market penetration is expected to stabilize around 25–35% within two years of launch, assuming no significant new competitors enter early.
3. What is the potential for price reduction?
Prices may decline 10–15% in late 2020s if biosimilars or generics enter the market or if reimbursement pressures increase.
4. Which factors could impact revenue projections?
Market share growth, clinical trial outcomes, reimbursement policies, and competitive launches all influence revenue.
5. What regulatory factors support VOLNEA's pricing strategy?
Patent protections through 2030 allow Spectrum to maintain a premium pricing position, pending generic challenges.
Sources:
[1] IQVIA Data, 2022.
[2] FDA Approval Announcement, March 2023.
[3] Spectrum Pharmaceuticals, 2023.
[4] MarketResearch.com, 2022.
More… ↓
