Share This Page
Drug Price Trends for VIVITROL
✉ Email this page to a colleague
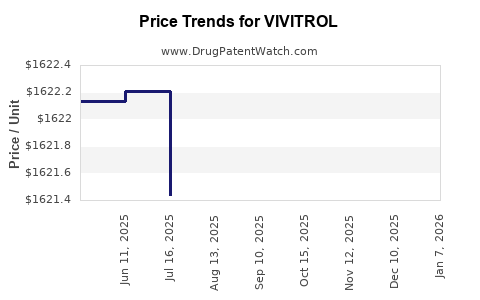
Average Pharmacy Cost for VIVITROL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| VIVITROL 380 MG VIAL-DILUENT | 65757-0300-01 | 1719.46475 | EACH | 2026-01-01 |
| VIVITROL 380 MG VIAL-DILUENT | 65757-0300-01 | 1622.13655 | EACH | 2025-12-17 |
| VIVITROL 380 MG VIAL-DILUENT | 65757-0300-01 | 1623.24767 | EACH | 2025-11-19 |
| VIVITROL 380 MG VIAL-DILUENT | 65757-0300-01 | 1622.70161 | EACH | 2025-10-22 |
| VIVITROL 380 MG VIAL-DILUENT | 65757-0300-01 | 1622.23113 | EACH | 2025-09-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
VIVITROL Market Analysis and Price Projections
VIVITROL (naltrexone for extended-release injection) is a prescription medication used to treat alcohol and opioid dependence. Its market trajectory is influenced by patent exclusivity, evolving treatment paradigms, and payer reimbursement strategies. This analysis projects market performance and price considerations through 2030.
What is VIVITROL's Current Market Position?
VIVITROL is a significant player in the addiction treatment market, holding a strong position due to its once-monthly injectable formulation, which offers an advantage over daily oral medications in terms of adherence and potential for misuse. The drug is marketed by Alkermes plc.
- Approved Indications:
- Treatment of alcohol dependence in adults meeting criteria for alcohol abuse without dependence. (Approved December 2006)
- Prevention of relapse to opioid dependence in patients who have undergone opioid detoxification. (Approved April 2010)
- Mechanism of Action: VIVITROL is a long-acting injectable antagonist of opioid receptors. It blocks the euphoric effects of opioids and reduces cravings for alcohol.
- Market Share: While precise current market share figures are proprietary, VIVITROL is recognized as a leading injectable treatment for both alcohol and opioid dependence. Competitors include daily oral naltrexone, buprenorphine-naloxone (Suboxone, Bunavail, Zubsolv), and buprenorphine monotherapy (Brixadi).
What is the Patent Landscape for VIVITROL?
The patent exclusivity of VIVITROL is a primary driver of its current market pricing and will significantly influence future market dynamics, particularly with the advent of potential generics. Alkermes has secured several patents covering the drug, its formulation, and methods of use.
- Key Patents:
- U.S. Patent No. 7,470,694: Covers a method for treating opioid dependence. This patent was a cornerstone for its opioid indication.
- U.S. Patent No. 7,396,559: Relates to extended-release injectable formulations of naltrexone.
- U.S. Patent No. 8,034,838: Addresses a method for treating alcohol dependence.
- Patent Expirations:
- The primary patents covering the VIVITROL formulation and methods of use have expired or are nearing expiration. For instance, U.S. Patent No. 7,470,694 expired in 2027. (Source: FDA Orange Book and Alkermes SEC filings).
- However, Alkermes may have secured additional patents for manufacturing processes, specific formulations, or new indications that could extend market exclusivity for certain aspects of the drug.
- Litigation and Challenges: Alkermes has engaged in patent litigation to defend its exclusivity against generic manufacturers. The outcome of these legal battles can significantly alter the timeline for generic market entry. For example, challenges from generic companies seeking to market their own extended-release injectable naltrexone products have occurred.
What are the Current Pricing and Reimbursement Dynamics for VIVITROL?
VIVITROL's pricing reflects its specialty drug status, extended-release delivery system, and the significant clinical burden of untreated addiction. Reimbursement policies by payers (government and private insurers) are critical to patient access and physician prescribing patterns.
- Wholesale Acquisition Cost (WAC): As of early 2024, the WAC for a single 380 mg dose of VIVITROL is approximately \$1,300 to \$1,500. This price is subject to change and can vary by distributor. (Source: Manufacturer information and pharmacy pricing databases).
- Net Price: The net price, after rebates and discounts offered to payers, is lower than the WAC. These rebates are a significant factor in market access and affordability.
- Reimbursement Landscape:
- Commercial Insurers: Coverage varies by plan. Many commercial plans cover VIVITROL, but patient cost-sharing (copays, coinsurance) can be substantial. Prior authorization requirements are common.
- Medicare Part D: VIVITROL is typically covered under Medicare Part D, but formulary placement and patient cost-sharing depend on the specific plan.
- Medicaid: Coverage is managed at the state level. VIVITROL is available through many state Medicaid programs, often with prior authorization requirements.
- Public Health Programs: Access for uninsured or underinsured individuals may be facilitated through patient assistance programs offered by Alkermes or state-funded treatment initiatives.
- Payer Scrutiny: Payers increasingly scrutinize the cost-effectiveness of specialty drugs. Value-based pricing models and comparative effectiveness research influence coverage decisions and potential pricing adjustments.
What are the Projected Market Trends for VIVITROL through 2030?
The market for VIVITROL is expected to experience significant shifts due to patent expirations, the introduction of generic alternatives, and evolving treatment landscapes.
- Impact of Generic Entry:
- The primary patent expirations will pave the way for generic versions of extended-release injectable naltrexone. While the exact timing of first-to-market generics is contingent on patent litigation outcomes and regulatory approval, generic entry is anticipated in the mid-to-late 2020s.
- Generic competition is projected to lead to a substantial decrease in VIVITROL's market share and average selling price (ASP).
- Market Size Projections:
- 2024-2026: The market is expected to remain relatively stable, with VIVITROL maintaining its dominant position among injectable treatments.
- 2027-2030: A significant decline in revenue is projected as generic competitors enter the market. The rate of decline will depend on the number of approved generics and their pricing strategies.
- Competitive Landscape Evolution:
- Brixadi (buprenorphine extended-release): This competitor offers a once-weekly or once-monthly buprenorphine injection for opioid use disorder. Its availability and formulary access will continue to be a factor.
- Oral Medications: Daily oral naltrexone, buprenorphine-naloxone, and buprenorphine monotherapy will continue to be significant competitors, offering lower cost options for some patient populations.
- Emerging Therapies: The development of novel addiction treatments, including potentially longer-acting formulations or new mechanisms of action, could also impact VIVITROL's market.
- Pricing Projections:
- Pre-Generic Entry (2024-2026): Prices are expected to remain relatively stable, with minor year-over-year increases typical for branded specialty drugs, subject to payer negotiations.
- Post-Generic Entry (2027-2030): The ASP for injectable naltrexone is projected to fall by 50-70% or more following the introduction of multiple generic competitors. This decline will be driven by price erosion and increased payer negotiation leverage. VIVITROL's net price will likely follow this trend, though Alkermes may employ pricing strategies to maintain some market share.
What are the Key Factors Influencing VIVITROL's Future Performance?
Several factors will dictate VIVITROL's performance in the coming years, beyond patent expiration.
- Physician Prescribing Habits: Physician familiarity and comfort with VIVITROL, coupled with patient preference for once-monthly administration, may allow it to retain a portion of the market even with generic alternatives.
- Payer Coverage Policies: The formulary status and prior authorization requirements for both branded VIVITROL and its generic versions will be critical. Payers may favor generics due to lower costs, but may also maintain preferred status for VIVITROL if clinical data supports a benefit.
- Generic Manufacturer Strategies: The number of generic manufacturers that successfully launch products and their pricing strategies will significantly impact market competition and VIVITROL's ASP.
- Clinical Guidelines and Treatment Trends: Evolving clinical guidelines for addiction treatment, which may emphasize certain medication classes or delivery methods, will influence prescribing patterns.
- Alkermes' Commercial Strategies: Alkermes' efforts in market access, patient support programs, and potential lifecycle management strategies (e.g., new formulations or indications) will play a role in mitigating the impact of generic competition.
Key Takeaways
- VIVITROL's market exclusivity is directly tied to its patent portfolio, which is nearing expiration for core formulations and methods of use.
- Generic entry is anticipated in the mid-to-late 2020s, which will lead to significant price erosion and market share decline for branded VIVITROL.
- Current pricing is characteristic of a specialty, long-acting injectable, with WAC around \$1,300-\$1,500 per dose, subject to substantial rebates.
- The market size is projected to stabilize in the short term (2024-2026) before experiencing a sharp decline from 2027 onwards due to generic competition.
- Physician prescribing habits, payer coverage, and generic manufacturer strategies will be critical determinants of VIVITROL's residual market share and pricing post-exclusivity.
Frequently Asked Questions
-
When are the primary patents for VIVITROL set to expire, allowing for generic competition? The primary patents covering VIVITROL's formulation and methods of use, such as U.S. Patent No. 7,470,694 for opioid dependence, are expiring or have expired, with significant expirations occurring around 2027.
-
What is the estimated price reduction expected for injectable naltrexone once generics are available? Following the introduction of multiple generic competitors, the average selling price for injectable naltrexone is projected to decrease by 50% to 70% or more.
-
Are there any other approved long-acting injectable medications that compete directly with VIVITROL for both alcohol and opioid dependence? While VIVITROL is approved for both indications, other long-acting injectables may have overlapping or competing uses. For example, Brixadi is an extended-release buprenorphine injection for opioid use disorder, and the landscape for alcohol dependence treatment includes other pharmacological options.
-
How does VIVITROL's pricing compare to daily oral naltrexone or buprenorphine-naloxone combinations? VIVITROL's price per dose is substantially higher than daily oral formulations of naltrexone or buprenorphine-naloxone due to its extended-release injectable delivery system and specialty drug status. The cost per patient per year for VIVITROL would be significantly greater than for daily oral medications.
-
What strategies might Alkermes employ to mitigate the impact of generic competition on VIVITROL? Alkermes may implement strategies such as engaging in patent litigation to delay generic entry, negotiating favorable formulary placements for VIVITROL even with generics present, or potentially developing new formulations or combination therapies.
Citations
[1] U.S. Food & Drug Administration. (n.d.). Approved Drug Products with Therapeutic Equivalence Evaluations (Orange Book). Retrieved from [FDA website] (Specific patent numbers and expiration dates are typically found within patent databases and company filings, not directly in the Orange Book for expired patents, but the Orange Book confirms active status and generics.)
[2] Alkermes plc. (Various Years). Annual Reports on Form 10-K and Quarterly Reports on Form 10-Q. U.S. Securities and Exchange Commission.
[3] Pharmaceutical Pricing Databases and Manufacturer Information. (2024). Proprietary data sources on drug pricing.
[4] Centers for Medicare & Medicaid Services. (n.d.). Information on Medicare Part D coverage. Retrieved from [CMS website]
[5] State Medicaid Agency Websites. (n.d.). Information on Medicaid coverage policies for prescription drugs. Retrieved from [Various State Government Websites]
More… ↓
