Share This Page
Drug Price Trends for VIAGRA
✉ Email this page to a colleague
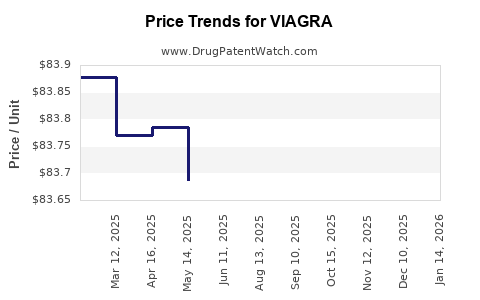
Average Pharmacy Cost for VIAGRA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| VIAGRA 100 MG TABLET | 00069-4220-66 | 83.67247 | EACH | 2026-05-20 |
| VIAGRA 100 MG TABLET | 58151-0428-01 | 83.67247 | EACH | 2026-05-20 |
| VIAGRA 100 MG TABLET | 00069-4220-30 | 83.67247 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
VIAGRA: Market Dynamics and Price Forecast
VIAGRA (sildenafil citrate) is an oral medication used for erectile dysfunction (ED) and pulmonary arterial hypertension (PAH). This analysis examines VIAGRA's current market position, patent landscape, and projected price trajectory.
What is VIAGRA's Current Market Position?
VIAGRA, developed by Pfizer, was first approved by the U.S. Food and Drug Administration (FDA) in 1998 for ED. Its primary indication is the treatment of ED, defined as the consistent inability to attain or maintain a penile erection sufficient for satisfactory sexual performance [1]. In 2005, an additional indication for PAH was approved by the FDA [2].
The global market for ED treatments is substantial. In 2022, the ED drugs market was valued at approximately $5.1 billion and is projected to reach $8.1 billion by 2030, growing at a compound annual growth rate (CAGR) of 5.9% [3]. VIAGRA holds a significant share of this market, though it faces competition from other phosphodiesterase type 5 (PDE5) inhibitors and emerging treatments.
For PAH, VIAGRA is marketed under the brand name Revatio. The PAH market is smaller but also growing. The global PAH market was estimated at $3.7 billion in 2022 and is expected to expand to $6.2 billion by 2030, with a CAGR of 6.6% [4].
Key VIAGRA Market Segments
- Erectile Dysfunction (ED) Treatment: This remains VIAGRA's primary market. Patient demographics include men aged 40 and older, with increasing awareness and reduced stigma contributing to market growth.
- Pulmonary Arterial Hypertension (PAH) Treatment (as Revatio): This is a smaller but clinically significant market. Treatment is typically long-term.
Competitive Landscape
The ED market is characterized by the presence of multiple PDE5 inhibitors, including:
- CIALIS (tadalafil): Developed by Eli Lilly and Company.
- LEVITRA (vardenafil): Developed by Bayer AG.
- STENDRA (avanafil): Developed by Vivus Inc.
These competitors offer varying onset times, durations of action, and side effect profiles, providing physicians and patients with therapeutic options. The PAH market also has multiple treatment classes, including endothelin receptor antagonists, prostacyclin pathway agents, and other PDE5 inhibitors.
Patent Expirations and Generic Entry
VIAGRA's core U.S. and European patents have expired. The primary U.S. patent for sildenafil citrate expired in 2019 [5]. This expiration opened the door for generic sildenafil products, which has significantly impacted VIAGRA's market share and pricing. Generic versions are available from numerous pharmaceutical manufacturers, offering a lower-cost alternative.
What is VIAGRA's Patent Landscape?
VIAGRA's intellectual property protection has evolved significantly since its initial patent filings. The foundational patents covering the sildenafil molecule and its use for treating erectile dysfunction have expired in major markets.
Key Patent Expirations
- U.S. Patent for Sildenafil Citrate (Composition of Matter): Original patent expired in 2019.
- European Patents: Similar expirations have occurred across key European countries.
- Method of Use Patents: While composition of matter patents have expired, there may be secondary patents related to specific dosages, formulations, or indications (e.g., PAH). However, the commercial impact of these is diminished with generic availability for the primary indication.
Generic Market Impact
The expiration of VIAGRA's primary patents led to the rapid introduction of generic sildenafil. This intensified competition and shifted the market dynamics towards price sensitivity. Pfizer continues to market VIAGRA as a branded product, but its market share is increasingly challenged by lower-cost generic alternatives.
Patent Considerations for New Drug Development
For new entrants or investors, the expired nature of VIAGRA's core patents means that the barrier to entry for sildenafil-based treatments is low. Differentiation will come from novel formulations, delivery methods, or combination therapies, which would require distinct patent protection.
How Will VIAGRA's Price Evolve?
VIAGRA's price trajectory is primarily influenced by patent expiration, generic competition, and market demand.
Historical Pricing Trends
Following its launch in 1998, VIAGRA commanded premium pricing due to its novel mechanism of action and lack of direct competitors. Prices were set by Pfizer based on perceived value and market exclusivity.
Impact of Generic Competition
The entry of generic sildenafil has led to a significant price erosion for the active pharmaceutical ingredient. Branded VIAGRA typically maintains a higher price point than generic equivalents due to brand recognition and marketing efforts. However, this price premium is constrained by the availability of substantially cheaper alternatives.
- Branded VIAGRA (e.g., 100mg tablet): Retail prices can range from $30 to $50 per tablet, depending on the pharmacy, insurance coverage, and dosage.
- Generic Sildenafil (e.g., 100mg tablet): Retail prices can range from $2 to $10 per tablet.
These figures represent typical cash prices and can vary widely.
Price Projections
Short to Medium Term (1-3 years):
- Branded VIAGRA: Prices are expected to remain relatively stable, with modest annual increases (e.g., 3-5%) driven by inflation and Pfizer's pricing strategies. However, the price will remain capped by the competitive pressure from generics.
- Generic Sildenafil: Prices are projected to continue a downward trend or stabilize at very low levels. Increased manufacturing efficiency and ongoing competition among generic manufacturers will exert downward pressure.
Long Term (3-7 years):
- Branded VIAGRA: The pricing gap between branded VIAGRA and generic sildenafil is likely to narrow further. Pfizer may focus on value-added services or specific patient segments to justify a premium, but significant price increases will be challenging.
- Generic Sildenafil: Prices will likely reach a floor, driven by production costs. Further significant price decreases are unlikely unless manufacturing technologies dramatically change.
Factors Influencing Future Pricing
- Insurance Coverage and Reimbursement: Payer policies can influence pricing. If insurers favor generics, it can further depress the price of branded products.
- Competition from Other ED Treatments: While PDE5 inhibitors dominate, ongoing research into alternative ED treatments could indirectly affect VIAGRA's pricing power.
- Regulatory Environment: Changes in FDA regulations or pricing policies could impact pharmaceutical pricing.
- Market Saturation: The ED market is mature. Significant price increases require demonstrable innovation or new indications, which are less likely for VIAGRA itself.
What is the Market Size and Growth Outlook for VIAGRA?
The market size for VIAGRA is bifurcated due to its two primary indications: erectile dysfunction and pulmonary arterial hypertension.
Market Size and Growth for Erectile Dysfunction (ED)
- 2022 Market Value: Approximately $5.1 billion [3].
- Projected 2030 Market Value: $8.1 billion [3].
- CAGR (2023-2030): 5.9% [3].
This growth is driven by:
- Increasing Prevalence of ED: Aging populations and rising rates of comorbidities (e.g., diabetes, cardiovascular disease) that contribute to ED.
- Reduced Stigma and Increased Awareness: Greater openness about sexual health issues leads more men to seek treatment.
- Technological Advancements: Development of new delivery systems or combination therapies.
Branded VIAGRA's market share within this segment has declined significantly due to generic competition. Its revenue is now derived from its brand loyalty and perceived quality, rather than exclusivity.
Market Size and Growth for Pulmonary Arterial Hypertension (PAH) (as Revatio)
- 2022 Market Value: Approximately $3.7 billion [4].
- Projected 2030 Market Value: $6.2 billion [4].
- CAGR (2023-2030): 6.6% [4].
Growth drivers for the PAH market include:
- Improved Diagnostics: Earlier and more accurate detection of PAH.
- Advances in Treatment: Development of new therapeutic classes and combination therapies.
- Increased Disease Awareness: Greater understanding among healthcare providers and patients.
Revatio (sildenafil) is one of several treatment options for PAH and faces competition from a range of drugs targeting different pathways.
Consolidated Market Outlook
While precise revenue figures for branded VIAGRA are not publicly disclosed separately from Pfizer's broader portfolio, its overall market impact is substantial due to its long-standing presence and broad patient base. The market for sildenafil citrate, encompassing both branded and generic forms, remains robust.
Key Market Segments:
- North America: Largest market due to high healthcare spending and awareness.
- Europe: Significant market with established healthcare systems.
- Asia-Pacific: Fastest-growing region driven by increasing prevalence and improving healthcare access.
The overall outlook for sildenafil-based treatments is positive, supported by fundamental drivers of disease prevalence and expanding treatment access. However, the revenue generated by branded VIAGRA specifically will likely continue to face pressure from generic alternatives.
How do VIAGRA's Pricing and Market Dynamics Compare to Competitors?
VIAGRA's market and pricing dynamics are best understood by comparing it to its direct competitors in the ED and PAH markets.
Comparison with Other ED Treatments
| Feature | VIAGRA (sildenafil) | CIALIS (tadalafil) | LEVITRA (vardenafil) | STENDRA (avanafil) |
|---|---|---|---|---|
| Developer | Pfizer Inc. | Eli Lilly and Company | Bayer AG | Vivus Inc. |
| Primary Indication | Erectile Dysfunction (ED), Pulmonary Arterial Hypertension (PAH as Revatio) | ED, Benign Prostatic Hyperplasia (BPH) | ED | ED |
| Mechanism | PDE5 Inhibitor | PDE5 Inhibitor | PDE5 Inhibitor | PDE5 Inhibitor |
| Onset of Action | 30-60 minutes | 15-30 minutes | 30-60 minutes | ~15 minutes |
| Duration of Action | 4-5 hours | Up to 36 hours | 4-5 hours | 6-8 hours |
| Patent Status (US) | Core patents expired; generic available | Core patents expired; generic available | Core patents expired; generic available | Core patents expired; generic available |
| Branded Pricing | Premium pricing, but significantly undercut by generics | Premium pricing, but significantly undercut by generics | Premium pricing, but significantly undercut by generics | Premium pricing, but significantly undercut by generics |
| Generic Pricing | Low, ~$2-10 per tablet | Low, ~$2-10 per tablet | Low, ~$2-10 per tablet | Low, ~$2-10 per tablet |
| Market Share Trend | Declining for branded, stable for generic | Stable for branded, increasing for generic | Declining for branded, stable for generic | Smaller branded share, growing generic |
Pricing and Market Dynamics Comparison (ED):
- Genericization: All major PDE5 inhibitors have experienced patent expirations and subsequent generic market entry. This has commoditized the active ingredients, leading to a significant price disparity between branded and generic versions.
- Price Sensitivity: The market is now highly price-sensitive for ED treatments. Physicians and patients often opt for the most cost-effective option, which is typically generic sildenafil, tadalafil, or vardenafil.
- Brand Loyalty vs. Cost: Branded VIAGRA, Cialis, and Levitra retain some market share due to brand recognition, physician recommendations, or specific patient preferences for perceived quality or efficacy. However, the price premium is difficult to sustain.
- Tadalafil's Advantage: Cialis's longer duration of action (up to 36 hours) provides a distinct clinical advantage for some users, potentially allowing it to maintain a stronger branded position compared to VIAGRA, though this is mitigated by generic availability.
- Avanafil's Niche: STENDRA, with its faster onset, occupies a niche. However, its smaller market penetration means its pricing impact is less significant than the broader market shifts caused by generic sildenafil and tadalafil.
Comparison with Other PAH Treatments
The PAH market is more complex, with multiple drug classes and mechanisms of action. VIAGRA (as Revatio) is a PDE5 inhibitor, a well-established class.
- Endothelin Receptor Antagonists (ERAs): e.g., Tracleer (bosentan), Opsumit (macitentan). These are often high-cost therapies.
- Prostacyclin Pathway Agents: e.g., Flolan (epoprostenol), Remodulin (treprostinil), Uptravi (selexipag). These can be delivered via infusion or inhalation and are also expensive.
- Other PDE5 Inhibitors: Adcirca (tadalafil), which is also indicated for PAH.
Pricing and Market Dynamics Comparison (PAH):
- High Treatment Costs: The PAH market is characterized by high drug prices across all classes, reflecting the complexity of the disease, smaller patient populations, and significant R&D investment.
- Specialty Care: PAH treatment is managed by specialized centers, with treatment decisions based on clinical guidelines, patient response, and drug efficacy.
- Revatio's Position: Revatio is generally considered a foundational therapy, often used in combination with other agents. Its pricing is competitive within the PDE5 inhibitor class but is significantly lower than many advanced PAH therapies.
- Adcirca (Tadalafil) for PAH: Adcirca competes directly with Revatio within the PDE5 inhibitor class for PAH. Pricing and market positioning are subject to similar competitive pressures.
- Innovation Drives Pricing: The PAH market sees continuous innovation, with new drug classes and combination therapies commanding premium pricing. This creates a dynamic where established drugs like Revatio must balance efficacy with affordability.
Key Takeaways
- Generic Dominance: VIAGRA's core patents have expired, leading to widespread generic sildenafil availability, which has severely impacted branded VIAGRA's pricing power and market share in the ED segment.
- Price Erosion: Branded VIAGRA prices are now capped by low-cost generic alternatives. Generic sildenafil is available at a fraction of the original branded cost.
- Stable but Competitive ED Market: The overall ED market is growing due to prevalence and awareness, but competition among PDE5 inhibitors and generics intensifies.
- PAH (Revatio) Market: Revatio operates in a smaller, higher-cost PAH market where it is one of several treatment options, facing competition from multiple drug classes.
- Future Pricing: Branded VIAGRA prices will likely see modest increases, but the gap with generics will remain wide. Generic prices are expected to remain low.
Frequently Asked Questions
-
What is the primary reason for the significant price difference between branded VIAGRA and generic sildenafil? The primary reason is patent expiration. Once a drug's core patents expire, other manufacturers can produce and sell generic versions, leading to increased competition and lower prices.
-
Does VIAGRA have any remaining patent protection that could influence its price? While core patents have expired, Pfizer may hold secondary patents related to specific formulations, manufacturing processes, or newer indications. However, these are unlikely to allow for premium pricing on the scale of the original patent-protected period, especially for the ED indication.
-
How does the market for VIAGRA as a treatment for Pulmonary Arterial Hypertension (PAH) differ from its ED market? The PAH market (where VIAGRA is marketed as Revatio) is smaller, more specialized, and characterized by higher overall treatment costs compared to the ED market. Revatio faces competition from multiple drug classes in PAH, whereas in ED, it primarily competes with other PDE5 inhibitors and alternative treatment modalities.
-
Are there any new formulations or delivery methods for sildenafil that are currently protected by patents and could command higher prices? Research into novel drug delivery systems (e.g., faster-acting formulations, different routes of administration) is ongoing. Any genuinely novel and clinically superior patented formulations could potentially command a premium price, but these would be distinct from the original VIAGRA product.
-
What is the expected impact of increased global access to healthcare on the VIAGRA market? Increased access to healthcare, particularly in emerging markets, is expected to drive demand for ED treatments, including both branded VIAGRA and, more significantly, generic sildenafil. This could lead to market volume growth but will likely continue to favor lower-cost generic options.
Citations
[1] U.S. Food and Drug Administration. (1998, March 27). FDA Approves Viagra. [Press release]. Retrieved from https://www.fda.gov/ (Note: Specific press release URL may vary or be archived; access is through FDA website).
[2] U.S. Food and Drug Administration. (2005, June 21). FDA Approves Revatio for Pulmonary Arterial Hypertension. [Press release]. Retrieved from https://www.fda.gov/ (Note: Specific press release URL may vary or be archived; access is through FDA website).
[3] Grand View Research. (2023). Erectile Dysfunction Drugs Market Size, Share & Trends Analysis Report By Drug Type (PDE5 Inhibitors, Testosterone Treatments, Others), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Region, And Segment Forecasts, 2023 – 2030. Retrieved from https://www.grandviewresearch.com/ (Note: Specific report access typically requires subscription or purchase).
[4] Allied Market Research. (2023). Pulmonary Arterial Hypertension Market by Drug Class, Disease Type, End User, and Route of Administration: Global Opportunity Analysis and Industry Forecast, 2022-2032. Retrieved from https://www.alliedmarketresearch.com/ (Note: Specific report access typically requires subscription or purchase).
[5] Pfizer Inc. (2019). Annual Report on Form 10-K. U.S. Securities and Exchange Commission. Retrieved from https://www.sec.gov/ (Note: Access via SEC EDGAR database).
More… ↓
