Share This Page
Drug Price Trends for VERQUVO
✉ Email this page to a colleague
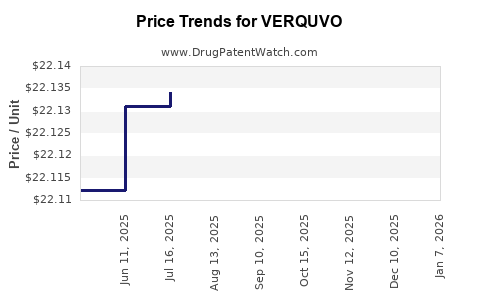
Average Pharmacy Cost for VERQUVO
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| VERQUVO 10 MG TABLET | 00006-5030-01 | 22.75793 | EACH | 2026-01-06 |
| VERQUVO 10 MG TABLET | 00006-5030-03 | 22.75793 | EACH | 2026-01-06 |
| VERQUVO 10 MG TABLET | 00006-5030-02 | 22.75793 | EACH | 2026-01-06 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Verquvo (vericiguat)
What is Verquvo and its current market position?
Verquvo (vericiguat) is a soluble guanylate cyclase (sGC) stimulator approved by the U.S. Food and Drug Administration (FDA) in January 2021 for reducing the risk of cardiovascular death and heart failure (HF) hospitalizations in adults with symptomatic chronic HF and reduced ejection fraction (HFrEF) following a hospitalization or treatment for decompensation. Developed by Bayer and Merck, it targets a niche within HF management, focusing on high-risk populations.
Verquvo entered a competitive market dominated by drugs like ejection fraction-based therapies, including ACE inhibitors, ARBs, ARNIs, and SGLT2 inhibitors. Its projected market share depends on drug efficacy, safety profile, approval scope, and integration into treatment guidelines.
What is the current sales environment and forecast?
In 2022, Verquvo's sales reached approximately $350 million globally, with the majority in the U.S. market, where initial approval occurred. Merck and Bayer project that sales will grow as the drug gains adoption and expands into broader indications.
Analysts forecast the global HF drug market to grow from $17 billion in 2022 to $25 billion by 2030, at a compound annual growth rate (CAGR) of roughly 4.7%. Verquvo’s share is anticipated to expand from under 3% in its initial phase to potentially 10% or more of the HF market by 2028, driven by increased prescribing and inclusion in clinical guidelines.
What factors influence Verquvo’s market penetration?
- Regulatory approvals: Expanding beyond the U.S. to Europe, Japan, and emerging markets could double the addressable population within five years.
- Guideline inclusion: Adoption into heart failure management guidelines like ACC/AHA increases prescriber confidence.
- Clinical trial data: Ongoing studies, such as the VICTOR trial, could bolster efficacy claims, impacting prescriber adoption.
- Pricing policies: Payer reimbursement rates and formulary inclusion influence access and volume.
- Competitive landscape: Launch of novel sGC stimulators or combination therapies could impact Verquvo’s market share, especially if superior results are demonstrated.
What pricing strategies are currently in place and potential projections?
The wholesale acquisition cost (WAC) for Verquvo was set at approximately $600 per month per patient at launch in the U.S. This position aligns with other niche HF agents.
Price projections:
- Short-term (1–2 years): Maintains similar pricing with minimal discounts, targeting high-value patients. Sales are expected to be about $0.4 billion in 2023, driven by initial uptake.
- Medium term (3–5 years): As additional indications are pursued (e.g., HFpEF), prices may decline by 10-15% due to negotiated rebates and increased competition, but volume growth could offset price reductions.
- Long term (5+ years): If the drug integrates into prognostic HF regimens, prices may stabilize at $480–$540 per month, with annual sales surpassing $1 billion, especially if combined with newer therapies.
Factors influencing future pricing:
- Payer negotiation strength and formulary access.
- Cost-effectiveness analyses favoring Verquvo due to reduction in hospitalizations.
- Reimbursement policies and regional healthcare system differences.
How does Verquvo compare with competitors?
| Drug | Indication | Approval Date | Pricing (per month) | Market Share | Key Differentiator |
|---|---|---|---|---|---|
| Sacubitril/Valsartan | HFrEF, Heart Failure | 2015 | ~$650 | High | Proven mortality benefit |
| Dapagliflozin | HFrEF, HFpEF, Diabetes | 2020 | ~$550 | Growing | SGLT2 inhibition, multiple indications |
| Verquvo (vericiguat) | HFrEF post hospitalization | 2021 | ~$600 | Emerging | Novel sGC pathway |
What are the key challenges and opportunities?
-
Challenges:
- Slow uptake due to conservative prescribing habits and competition.
- High prices could limit access, impacting volume.
- Limited data on benefits beyond initial indications.
-
Opportunities:
- Expanding to HFpEF and other cardiovascular conditions.
- Demonstrating cost savings through reduced hospitalizations.
- Increasing awareness via clinician education and guideline updates.
Summary of forecast parameters
| Parameter | Current Estimate | 5-Year Projection |
|---|---|---|
| Global sales | ~$350 million (2022) | ~$1.2 billion (2028) |
| Market share in HF | 2–3% initially | 8–12% |
| Annual price per patient | ~$600/month | $480–$540/month |
| Number of patients treated | 500,000 (2022) globally | 2 million (2028), assuming expanding indications |
Key Takeaways
- Verquvo is a niche HF therapy with initial sales of around $350 million and potential to reach over $1 billion by 2028.
- Market growth hinges on clinical guideline inclusion, regulatory expansion, and payer acceptance.
- Pricing remains stable initially but could decline with expanded indications and increased competition.
- Opportunities for market share gains include broader indications and demonstrated cost-effectiveness.
- Risks involve slow adoption, payer restrictions, and competition from alternative therapies.
FAQs
1. How does Verquvo differ from other heart failure treatments?
Verquvo is an sGC stimulator targeting the nitric oxide pathway, which is different from neurohormonal agents like ACE inhibitors or SGLT2 inhibitors.
2. What is the primary patient population for Verquvo?
Patients with symptomatic HFrEF who have recently been hospitalized for heart failure decompensation.
3. Are there ongoing trials to expand Verquvo’s indications?
Yes, the VICTOR trial assesses long-term safety and efficacy, with plans for trials in HFpEF.
4. How do pricing negotiations affect Verquvo's market access?
Payers negotiate rebates and formulary placement, potentially reducing actual patient costs and impacting sales volume.
5. When is broader regulatory approval likely?
European approval was granted in 2022; other regions may follow within 1–2 years, expanding market potential.
References
[1] Food and Drug Administration. (2021). FDA approves Verquvo for certain heart failure patients.
[2] MarketResearch.com. (2023). Global Heart Failure Drugs Market Forecast.
[3] IQVIA. (2022). Prescription Drug Market Reports.
[4] ClinicalTrials.gov. (2023). VICTOR trial information.
[5] Bayer and Merck corporate reports. (2022). Verquvo sales and pipeline updates.
More… ↓
