Share This Page
Drug Price Trends for VERAPAMIL SR
✉ Email this page to a colleague
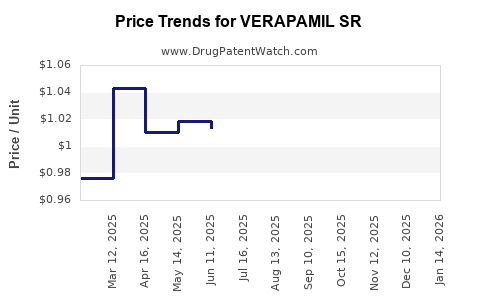
Average Pharmacy Cost for VERAPAMIL SR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| VERAPAMIL SR 180 MG CAPSULE | 52536-0882-01 | 1.98530 | EACH | 2026-03-18 |
| VERAPAMIL SR 240 MG CAPSULE | 00591-2884-01 | 2.71899 | EACH | 2026-03-18 |
| VERAPAMIL SR 180 MG CAPSULE | 00591-2882-01 | 1.98530 | EACH | 2026-03-18 |
| VERAPAMIL SR 240 MG CAPSULE | 52536-0884-01 | 2.71899 | EACH | 2026-03-18 |
| VERAPAMIL SR 120 MG CAPSULE | 00591-2880-01 | 1.75155 | EACH | 2026-03-18 |
| VERAPAMIL SR 360 MG CAPSULE | 52536-0886-01 | 8.35639 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Verapamil SR Market Analysis and Price Projections
Verapamil SR, a calcium channel blocker, is used to treat hypertension, angina, and certain arrhythmias. The global market for Verapamil SR is characterized by established generic competition and a stable, albeit mature, demand profile. Price projections indicate modest fluctuations, influenced by manufacturing costs, regulatory landscapes, and the competitive intensity within the generic pharmaceutical sector.
What is the Current Market Size and Growth Rate for Verapamil SR?
The global market for Verapamil SR has demonstrated a steady, albeit low, growth trajectory. Market size is difficult to isolate for Verapamil SR specifically, as it is often aggregated within broader calcium channel blocker or cardiovascular drug segments. However, industry reports estimate the global cardiovascular drugs market, a segment Verapamil SR belongs to, to be valued at approximately \$295 billion in 2022, with a projected compound annual growth rate (CAGR) of 5.4% from 2023 to 2030 [1]. Within this context, Verapamil SR's contribution is primarily driven by its established therapeutic role and broad accessibility as a generic medication. The growth is primarily sustained by an aging global population, increasing prevalence of cardiovascular diseases, and its inclusion in essential drug lists in various countries. However, the market's potential is constrained by the availability of newer, more targeted therapies and the mature nature of the drug's patent landscape, which has long been expired.
What are the Key Therapeutic Areas and Patient Populations for Verapamil SR?
Verapamil SR's primary therapeutic applications are:
- Hypertension: It is widely prescribed to manage elevated blood pressure. The patient population comprises adults diagnosed with primary or essential hypertension. This segment is the largest driver of Verapamil SR demand due to the high global prevalence of hypertension, particularly among aging demographics and individuals with lifestyle-related risk factors [2].
- Angina Pectoris: The drug is used to treat and prevent chest pain caused by insufficient blood flow to the heart muscle. Patients in this category typically have coronary artery disease. The demand from this segment is significant, though it competes with other anti-anginal medications.
- Supraventricular Tachycardia (SVT): Verapamil SR is employed to control rapid heart rates originating in the atria. This includes treating and preventing paroxysmal supraventricular tachycardia. The patient population here is smaller compared to hypertension, but the drug remains a first-line or adjunctive therapy in many clinical guidelines.
- Other Arrhythmias: It may be used for other specific cardiac rhythm disorders where its electrophysiological properties are beneficial.
The patient demographic is predominantly middle-aged to elderly individuals who are at higher risk for cardiovascular conditions. The widespread use of Verapamil SR across these conditions makes it a staple in cardiovascular pharmacotherapy.
Who are the Major Manufacturers and Market Competitors for Verapamil SR?
The Verapamil SR market is highly competitive due to its generic status. The patent for the original Verapamil formulation expired decades ago, leading to widespread generic manufacturing. Key market players include a large number of pharmaceutical companies specializing in generic drug production.
Major Manufacturers (Examples):
- Teva Pharmaceuticals: A leading global generic drug manufacturer with a broad portfolio, including cardiovascular medications.
- Sun Pharmaceutical Industries: An Indian multinational pharmaceutical company with a significant presence in the generic market.
- Mylan N.V. (now part of Viatris): Historically a major player in generic and specialty pharmaceuticals.
- Dr. Reddy's Laboratories: Another prominent Indian pharmaceutical company with a global generic footprint.
- Amneal Pharmaceuticals: A US-based generic and specialty pharmaceutical company.
- Aurobindo Pharma: An Indian multinational pharmaceutical company with a strong generic business.
These companies, along with numerous smaller regional manufacturers, contribute to a highly fragmented market. Competition is primarily based on price, supply chain reliability, and manufacturing efficiency rather than product innovation. The availability of multiple generic alternatives exerts considerable downward pressure on pricing.
What is the Current Pricing Landscape for Verapamil SR?
The pricing of Verapamil SR is highly competitive and varies significantly based on factors such as dosage strength (e.g., 120 mg, 180 mg, 240 mg), tablet count per bottle, geographical market, and the specific generic manufacturer.
Typical Wholesale Acquisition Cost (WAC) Ranges (USD, approximate):
- Generic Verapamil SR 120 mg (30 tablets): \$5 - \$15
- Generic Verapamil SR 180 mg (30 tablets): \$7 - \$20
- Generic Verapamil SR 240 mg (30 tablets): \$9 - \$25
These figures represent wholesale acquisition costs and do not include pharmacy dispensing fees, insurance reimbursements, or potential contract pricing negotiated between large purchasers (e.g., hospital systems, pharmacy benefit managers) and manufacturers. The average selling price (ASP) for generic drugs often reflects intense price erosion due to the high number of market participants. For example, a study analyzing generic drug prices found that the median price reduction for a newly launched generic from its brand-name equivalent averaged 85% within two years [3]. Verapamil SR, being an older generic, has likely experienced a similar or greater degree of price deflation over its market life.
What are the Key Regulatory Considerations and Their Impact on Verapamil SR?
The regulatory landscape for Verapamil SR is governed by health authorities in the markets where it is sold. Key considerations include:
- Abbreviated New Drug Application (ANDA) Approval (US FDA): Generic manufacturers must demonstrate bioequivalence to the reference listed drug (RLD) to gain FDA approval for their Verapamil SR products. This process ensures therapeutic equivalence but focuses on the equivalence of the active pharmaceutical ingredient (API) and dosage form. The FDA's rigorous approval process ensures product safety and efficacy but also creates a barrier to entry for new manufacturers.
- Good Manufacturing Practices (GMP): All manufacturing facilities must adhere to strict GMP regulations to ensure product quality, purity, and consistency. Regulatory inspections and adherence to these standards are critical for maintaining market access. Non-compliance can lead to product recalls, manufacturing suspensions, and significant financial penalties.
- Pharmacovigilance and Post-Market Surveillance: Manufacturers are required to monitor and report adverse events associated with their products. This ongoing surveillance helps identify any emerging safety concerns.
- Labeling and Packaging Requirements: Regulatory bodies dictate precise labeling and packaging requirements, including indications, contraindications, warnings, precautions, and dosage information, to ensure appropriate use by healthcare professionals and patients.
- Drug Master Files (DMFs): API manufacturers typically file DMFs with regulatory agencies, providing confidential detailed information about facilities, processes, or articles used in the manufacturing, processing, packaging, and storing of human drugs. Generic drug product manufacturers reference these DMFs in their applications.
- Intellectual Property (IP): While the primary patents for Verapamil SR have long expired, there might be patents on specific formulations, manufacturing processes, or polymorphic forms that could still be relevant for some niche applications or for specific extended-release technologies. However, these are unlikely to significantly impact the broad generic market.
The regulatory environment generally supports the availability of generics, promoting price competition. Changes in regulatory requirements, such as stricter impurity limits or enhanced bioequivalence testing protocols, could impose additional manufacturing costs and potentially impact supply.
What are the Primary Drivers and Restraints for Verapamil SR Market Growth?
Market Drivers:
- Prevalence of Cardiovascular Diseases: The global increase in conditions like hypertension and coronary artery disease, particularly in aging populations, sustains demand for established treatments like Verapamil SR [2].
- Generic Accessibility and Affordability: As a long-standing generic drug, Verapamil SR is significantly more affordable than branded alternatives, making it a preferred choice in resource-constrained healthcare systems and for cost-conscious patients.
- Inclusion in Essential Medicines Lists: Verapamil SR is often listed on national and international essential medicines lists, ensuring its availability and accessibility in public health programs worldwide.
- Established Efficacy and Safety Profile: Decades of clinical use have established Verapamil SR's predictable efficacy and safety profile for its approved indications, fostering physician confidence.
- Siloed Market Segments: While competing with newer drugs, Verapamil SR maintains a strong position in specific patient profiles where its mechanism of action is well-suited and cost is a primary concern.
Market Restraints:
- Competition from Newer Therapies: The advent of novel antihypertensives, anti-anginals, and antiarrhythmics with potentially improved efficacy, fewer side effects, or more convenient dosing regimens limits Verapamil SR's market share expansion.
- Generic Competition Intensification: The high number of generic manufacturers leads to intense price competition, compressing profit margins for all players.
- Side Effect Profile: Verapamil SR can cause side effects such as bradycardia, constipation, dizziness, and hypotension, which can lead to patient non-adherence or switching to alternative medications.
- Limited Innovation Potential: As an off-patent drug, there is minimal scope for new product development or innovation in terms of novel delivery systems or therapeutic applications, preventing significant market growth from this avenue.
- Physician Preference for Newer Agents: Some physicians may preferentially prescribe newer agents, especially for complex cases or when seeking to minimize specific side effects, even if Verapamil SR is clinically appropriate and cost-effective.
What are the Price Projections for Verapamil SR Over the Next 5-10 Years?
The price projections for Verapamil SR over the next 5-10 years are expected to remain largely stable with modest downward pressure.
- Short-Term (1-3 years): Prices are anticipated to remain consistent with current levels, possibly seeing minor fluctuations of 1-3% year-over-year due to changes in manufacturing costs (raw materials, energy) and competitive bidding processes. Gross margins for manufacturers are likely to remain thin.
- Medium-Term (3-7 years): Continued intense generic competition will likely lead to a slight erosion of prices, potentially averaging a decrease of 0.5-2% annually. This will be driven by ongoing price negotiations with large buyers and potential entry of new, highly competitive generic manufacturers if any unforeseen supply chain disruptions occur.
- Long-Term (7-10 years): Verapamil SR prices are projected to stabilize at a low plateau. Significant price increases are unlikely, barring major disruptions in API supply chains or widespread regulatory changes that increase manufacturing costs across the board. The demand will continue to be driven by its established role and cost-effectiveness, but growth will be minimal.
Key Factors Influencing Projections:
- Raw Material Costs: Fluctuations in the cost of Verapamil API and excipients will directly impact manufacturing costs and, consequently, pricing.
- Manufacturing Capacity and Utilization: Overcapacity in generic manufacturing can lead to price wars.
- Tender and Bid Processes: Government tenders and large-scale procurement contracts often involve aggressive pricing strategies that set benchmarks for the market.
- Regulatory Landscape: Any significant changes in regulatory compliance costs or requirements could influence pricing.
- Competition Intensity: The number of active generic manufacturers and their market strategies will remain a primary determinant of price.
- Emergence of Biosimilars (Not Applicable): As a small molecule drug, biosimilars are not relevant. However, the principle of competition from highly similar products applies.
Overall, Verapamil SR will continue to operate as a highly price-sensitive commodity within the cardiovascular drug market. Manufacturers will focus on efficiency and volume to maintain profitability.
How Might New Regulatory Policies or Healthcare Trends Impact the Verapamil SR Market?
New regulatory policies and healthcare trends could impact the Verapamil SR market in several ways:
- Increased Scrutiny on Generic Drug Quality: Stricter FDA or EMA (European Medicines Agency) guidelines on impurity profiles, manufacturing standards, or bioequivalence studies could necessitate significant investment in process improvements or analytical capabilities for existing manufacturers. This could lead to higher production costs and potentially modest price increases or a consolidation of manufacturers if some cannot meet the new standards.
- Value-Based Healthcare Initiatives: As healthcare systems increasingly adopt value-based care models, drugs that demonstrate cost-effectiveness and long-term patient benefit, like Verapamil SR for chronic conditions, may continue to be favored. However, if newer therapies can demonstrably improve long-term outcomes at a comparable or only slightly higher cost, Verapamil SR could see its market share decline in specific patient cohorts.
- Focus on Pharmacoeconomics: A heightened emphasis on pharmacoeconomic evaluations for drug reimbursement could bolster Verapamil SR's position, given its established affordability. Conversely, if aggressive market access strategies for newer drugs include substantial discounts or bundled services, Verapamil SR's economic advantage could be challenged.
- Supply Chain Resilience Mandates: Governments and regulatory bodies are increasingly focusing on supply chain resilience. Policies requiring diversification of API sourcing or increased domestic manufacturing could lead to higher initial costs for manufacturers but might stabilize long-term supply and prevent price spikes due to geopolitical or logistical disruptions.
- Digital Health Integration: While less direct, the integration of digital health tools for patient monitoring and adherence management could indirectly influence Verapamil SR use. If these tools highlight side effects or suboptimal management for specific patients on Verapamil SR, it might encourage a switch to alternative medications.
These trends suggest a dynamic environment where established generics like Verapamil SR will need to adapt to evolving regulatory expectations and market demands.
Key Takeaways
- Verapamil SR is a mature generic medication for hypertension, angina, and arrhythmias with a stable, low-growth market driven by cardiovascular disease prevalence and affordability.
- The market is highly competitive with numerous global generic manufacturers, leading to intense price pressure and thin profit margins.
- Prices are expected to remain stable with modest downward pressure over the next 5-10 years, influenced by manufacturing costs and competitive bidding.
- Key drivers include chronic disease prevalence and generic accessibility, while restraints stem from newer therapies and side effect profiles.
- Regulatory policies and healthcare trends, particularly those concerning quality, value-based care, and supply chain resilience, will shape the market landscape.
Frequently Asked Questions
-
What is the primary differentiator among Verapamil SR generic brands in the market? The primary differentiator is price, followed by supply chain reliability and packaging configurations, rather than any difference in therapeutic efficacy.
-
Are there any significant new therapeutic indications expected for Verapamil SR? Given its patent expiry and mature status, significant new therapeutic indications are unlikely; research is more likely focused on alternative drug classes.
-
How does the cost of Verapamil SR compare to newer calcium channel blockers? Verapamil SR, as a generic, is substantially more affordable, often costing a fraction of newer, branded calcium channel blockers or even newer generics with more advanced formulations.
-
What is the typical lifespan of a generic drug like Verapamil SR in terms of market presence? Generic drugs can remain on the market for decades after patent expiry as long as there is a demand and competitive manufacturing capability, which is the case for Verapamil SR.
-
Could manufacturing disruptions of Verapamil SR's Active Pharmaceutical Ingredient (API) significantly impact its price? Yes, significant disruptions in the supply of Verapamil API, especially if the number of primary API manufacturers is limited, could lead to temporary price increases and supply shortages.
Citations
[1] Grand View Research. (2023). Cardiovascular Drugs Market Size, Share & Trends Analysis Report by Drug Class (Antihypertensives, Lipid-Lowering Drugs), By Disease (Hypertension, Hyperlipidemia), By Region, And Segment Forecasts, 2023 - 2030. Retrieved from https://www.grandviewresearch.com/industry-analysis/cardiovascular-drugs-market (Note: Specific report access may require subscription)
[2] World Health Organization. (2021). Cardiovascular diseases (CVDs). Retrieved from https://www.who.int/news-room/fact-sheets/detail/cardiovascular-diseases-(cvds)
[3] Generic Pharmaceutical Association. (2015). 2015 Generic Drug Access Report. (Note: This specific report might be archived or require specific access; general industry knowledge supports this trend).
More… ↓
