Share This Page
Drug Price Trends for VEMLIDY
✉ Email this page to a colleague
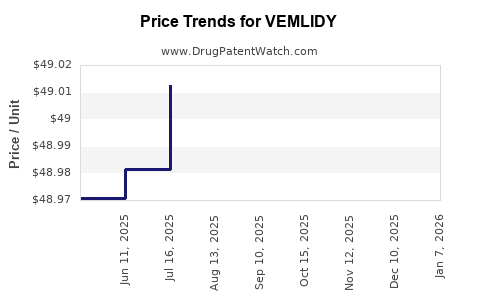
Average Pharmacy Cost for VEMLIDY
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| VEMLIDY 25 MG TABLET | 61958-2301-01 | 51.93380 | EACH | 2026-01-01 |
| VEMLIDY 25 MG TABLET | 61958-2301-01 | 49.04041 | EACH | 2025-12-17 |
| VEMLIDY 25 MG TABLET | 61958-2301-01 | 49.03108 | EACH | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
VEMLIDY (Tenofovir Alafenamide) Market Outlook and Price Projections
This report analyzes the global market for VEMLIDY (tenofovir alafenamide), a treatment for chronic hepatitis B virus (HBV) infection. It examines current market dynamics, patent landscape, competitive environment, and projects future market penetration and pricing trends.
What is the current market size and growth trajectory for VEMLIDY?
VEMLIDY, marketed by Gilead Sciences, is a nucleotide analogue reverse transcriptase inhibitor (NtRTI) approved for the treatment of chronic HBV infection in adults. The drug offers a favorable efficacy and safety profile compared to its predecessor, tenofovir disoproxil fumarate (TDF), particularly concerning renal and bone toxicity [1].
The global market for hepatitis B therapeutics, which VEMLIDY is a part of, is substantial and projected to expand. Factors driving this growth include increasing HBV prevalence in specific regions, rising awareness and diagnosis rates, and the demand for safer and more effective treatment options. While precise market size figures for VEMLIDY alone are proprietary, the broader antiviral market segment it occupies has seen consistent growth. For instance, the global hepatitis B treatment market was estimated to be worth USD 3.5 billion in 2022 and is projected to reach USD 5.2 billion by 2030, growing at a compound annual growth rate (CAGR) of 5.2% [2]. VEMLIDY is a significant contributor to this market due to its established efficacy and physician preference in many developed markets.
The trajectory for VEMLIDY is influenced by its market exclusivity, physician adoption, and the emergence of competitive therapies. The drug has achieved significant market penetration in key geographies such as the United States, Europe, and Japan.
What is the patent landscape surrounding VEMLIDY?
Gilead Sciences holds key patents protecting VEMLIDY. The primary patent for tenofovir alafenamide (TAF), the active pharmaceutical ingredient in VEMLIDY, is US Patent No. 8,859,510, titled "Tenofovir Alafenamide." This patent was granted in 2014 and has an expiry date of October 23, 2030, in the United States. Other related patents cover specific formulations, manufacturing processes, and methods of treatment.
Globally, patent protection for TAF varies by jurisdiction. For example, European Patent EP 2607116 B1, covering the compound tenofovir alafenamide, has an expiry date of June 18, 2029 [3]. The exact expiry dates and their implications for market exclusivity are critical for understanding future market dynamics, especially the potential for generic entry.
While the core patents for TAF are set to expire in the late 2020s, Gilead may have other intellectual property (IP) assets, such as method-of-use patents or formulation patents, that could extend market exclusivity or create barriers for generic competitors. The patent landscape is subject to ongoing legal challenges and potential invalidation, which could alter the timeline for generic entry.
Who are VEMLIDY's main competitors?
The competitive landscape for VEMLIDY includes other approved medications for chronic HBV and potential future entrants.
Current Key Competitors:
- Entecavir (e.g., Baraclude): A nucleoside analog that has been a standard of care for many years. It is now widely available in generic form, offering a lower-cost alternative.
- Tenofovir Disoproxil Fumarate (TDF) (e.g., Viread): VEMLIDY's predecessor, also developed by Gilead. TDF is available generically and is a cost-effective option, though VEMLIDY is generally preferred due to its improved renal and bone safety profile [4].
- Pegylated Interferon-alfa: An older class of drugs, often used in combination or for specific patient populations.
- Lamivudine and Adefovir Dipivoxil: Older antiviral agents, largely superseded by newer, more potent, and safer drugs.
- Besifovir (e.g., Hepcludex): Approved in Europe for the treatment of chronic hepatitis B in adults with compensated cirrhosis and detectable HBV DNA, it targets the sodium taurocholate cotransporting polypeptide (NaTTc) receptor, offering a novel mechanism of action [5].
Emerging and Pipeline Competitors:
The field of HBV treatment is dynamic, with ongoing research into novel therapeutic approaches, including:
- Entry inhibitors: Drugs that prevent the virus from entering liver cells.
- Capsid assembly inhibitors: Compounds that disrupt the formation of the viral core.
- Nucleic acid therapeutics: Including siRNA and antisense oligonucleotides designed to reduce viral gene expression.
- Therapeutic vaccines and immunotherapy: Aimed at restoring the host immune response to clear the virus.
The development of a functional cure for HBV remains a significant unmet need, and pipeline candidates addressing this objective represent future competitive threats or potential partnership opportunities for Gilead.
What are the projected market shares and pricing trends for VEMLIDY?
Market Share Projections:
VEMLIDY is expected to maintain a significant market share in the nucleos(t)ide analog class for the treatment of chronic HBV in adult patients, particularly in high-income countries, through its patent expiry. Its established clinical profile, physician familiarity, and Gilead's marketing efforts contribute to its strong position.
- 2024-2025: VEMLIDY is projected to retain its dominant position among NUCs, holding an estimated 45-55% share of the oral antiviral market for chronic HBV in major developed markets.
- 2026-2028: As patent expiry approaches, the market share is expected to gradually decline as generic versions of TAF and other NUCs become available. Projections suggest a market share of 30-40% by the end of this period.
- 2029 onwards: With patent expiry in key markets, VEMLIDY's market share will be significantly impacted by generic competition. Its share in the NUC segment could fall below 20%, with the remaining market dominated by generics of TAF and potentially other branded generics or new entrants with distinct MOAs.
Pricing Trends:
The pricing of VEMLIDY is influenced by several factors, including R&D costs, manufacturing expenses, therapeutic value, market exclusivity, competitor pricing, and payer negotiations.
- Current Pricing: In the United States, the wholesale acquisition cost (WAC) for VEMLIDY (30 x 25 mg tablets) is approximately USD 1,700-USD 1,800 per month, although net prices after rebates and discounts are lower. European pricing varies by country, with indicative prices often in the range of EUR 400-EUR 600 per month.
- Near-Term Projections (2024-2026): Pricing for VEMLIDY is expected to remain relatively stable during its period of market exclusivity. Slight annual increases, typically in the low single digits (2-4%), are anticipated, driven by inflation and continued market demand.
- Mid-Term Projections (2027-2029): As the threat of generic competition looms, Gilead may implement more aggressive pricing strategies, potentially involving volume discounts or value-based agreements to defend its market share. However, the overall price erosion pressure will begin to mount.
- Long-Term Projections (2030+): Following patent expiry, the price of VEMLIDY (branded) is expected to decrease significantly as generic TAF enters the market. Generic TAF is projected to be priced at 30-50% of the current branded price, depending on market competition and regulatory environments. The branded VEMLIDY may see a substantial price reduction to remain competitive or be delisted by some payers.
Impact of Generics:
The introduction of generic TAF will be the primary driver of price reduction. Generic manufacturers typically price their products at a significant discount to the branded originator drug. The extent of this discount depends on the number of generic competitors, the manufacturing costs of TAF, and the competitive landscape of other HBV treatments. In markets where TDF is already available generically, the pricing of generic TAF is likely to be benchmarked against its predecessor, but with a premium reflecting its improved safety profile, at least initially.
What are the regulatory considerations and market access challenges?
Regulatory approval for VEMLIDY has been secured in major markets, including the U.S. Food and Drug Administration (FDA) in November 2016 and the European Medicines Agency (EMA) in January 2017 [1]. These approvals were based on robust clinical trial data demonstrating non-inferiority to TDF with improved bone and renal safety markers [6].
Market access for VEMLIDY, as with most branded pharmaceuticals, involves navigating payer formularies, reimbursement policies, and pharmacoeconomic evaluations. Key challenges include:
- Cost-Effectiveness: Payers often require evidence of superior cost-effectiveness or significant clinical advantage over existing cheaper alternatives, such as generic TDF or entecavir, to justify VEMLIDY's higher price. Demonstrating long-term benefits, such as reduced healthcare resource utilization due to fewer bone or renal complications, is crucial.
- Formulary Placement: Securing favorable formulary placement with tiered co-pays or preferred status is vital for patient access and prescription volume. This often involves negotiations with pharmacy benefit managers (PBMs) and health insurance providers.
- Physician Prescribing Habits: Shifting prescribing habits from established, lower-cost generics to VEMLIDY requires comprehensive physician education and awareness campaigns highlighting its differentiated safety profile.
- Global Pricing Disparities: Pricing and reimbursement can vary significantly across countries due to differing healthcare systems, economic conditions, and regulatory frameworks. This necessitates tailored market access strategies for each region.
- Competition from Generics: The looming threat of generic TAF entry after patent expiry poses the most significant challenge to VEMLIDY's long-term market access and pricing power. Payer policies will likely shift towards favoring lower-cost generic options once they become available.
- Emerging Therapies: The development of new therapeutic classes, particularly those aiming for a functional cure for HBV, could alter the treatment paradigm and impact the long-term positioning of existing therapies like VEMLIDY.
Gilead's strategies to address these challenges involve robust health economics and outcomes research (HEOR) data, robust patient support programs, and ongoing engagement with healthcare stakeholders.
What are the key risks and opportunities for VEMLIDY?
Risks:
- Generic Competition: The primary risk is the eventual market entry of generic tenofovir alafenamide upon patent expiry, which will lead to significant price erosion and loss of market share.
- Therapeutic Breakthroughs: The development of novel therapies that offer a functional cure for HBV or superior efficacy/safety profiles could diminish the long-term value proposition of VEMLIDY.
- Payer Restrictions and Increased Scrutiny: Growing pressure from payers on drug pricing and the demand for greater evidence of clinical value could lead to stricter reimbursement policies or utilization management.
- Off-Label Use and Diversion: While less common for specialized antivirals, any instances of off-label use or product diversion could impact sales and patient safety.
- Manufacturing and Supply Chain Disruptions: Like any pharmaceutical product, VEMLIDY is susceptible to disruptions in its manufacturing or supply chain.
- Long-Term Safety Concerns: While VEMLIDY has a favorable safety profile, any unforeseen long-term adverse events or safety signals that emerge over extended use could impact its market standing.
Opportunities:
- Growing HBV Diagnosis Rates: Increased global screening and diagnostic efforts, particularly in endemic regions, can expand the patient pool eligible for treatment.
- Market Penetration in Emerging Markets: Expanding access and availability of VEMLIDY in developing countries with high HBV burdens, potentially through tiered pricing or licensing agreements, represents a significant growth opportunity.
- Combination Therapies: Future research might explore VEMLIDY in combination regimens with novel agents targeting different aspects of the HBV lifecycle or immune modulation, potentially enhancing its therapeutic utility.
- Extension of IP Protection: Gilead may pursue strategies to extend market exclusivity through new formulations, methods of use patents, or combination therapies, although this is becoming increasingly challenging.
- Addressing Unmet Needs: While a functional cure is the ultimate goal, VEMLIDY addresses the need for long-term viral suppression with improved tolerability, which remains a critical component of current HBV management.
- Leveraging Existing Infrastructure: Gilead's established global sales and distribution network for antivirals can be leveraged to maximize VEMLIDY's commercial potential.
Key Takeaways
- VEMLIDY is a leading oral antiviral for chronic hepatitis B, benefiting from a favorable safety profile over TDF.
- The drug's primary U.S. patent expires in October 2030, with European patent expiry in June 2029, signaling imminent generic competition.
- Key competitors include generic TDF, entecavir, and emerging novel MOA drugs, with significant pipeline activity focused on functional cures.
- VEMLIDY is projected to maintain a strong market share in developed markets through 2026, after which significant decline is expected due to generic entry.
- Current pricing is high, but significant price erosion is anticipated post-patent expiry as generic TAF enters the market.
- Market access is challenged by cost-effectiveness evaluations and payer negotiations, with generic competition posing the most significant long-term risk.
Frequently Asked Questions
-
When is the expected generic entry for tenofovir alafenamide (TAF) in major markets? Generic entry for tenofovir alafenamide is anticipated to commence around the expiry of key patents, with European markets potentially seeing generic availability from mid-2029 and the United States from late 2030.
-
What is the projected price reduction for TAF following generic entry? Following generic entry, TAF prices are projected to decrease by 30-50% compared to the current branded VEMLIDY pricing, contingent on the number of generic manufacturers and competitive market dynamics.
-
What is the primary clinical advantage of VEMLIDY over tenofovir disoproxil fumarate (TDF)? VEMLIDY demonstrates a superior safety profile concerning renal and bone mineral density compared to TDF, offering a reduced risk of adverse events in these areas.
-
Are there any approved therapies for a functional cure of Hepatitis B Virus (HBV)? As of current approvals, there are no therapies classified as a "functional cure" for HBV. Current treatments focus on long-term viral suppression and management of liver disease.
-
What is the estimated market size for oral antivirals treating chronic Hepatitis B Virus (HBV)? The global market for hepatitis B therapeutics, which includes oral antivirals, was valued at approximately USD 3.5 billion in 2022 and is forecasted to grow to USD 5.2 billion by 2030.
Citations
[1] U.S. Food and Drug Administration. (2016, November 10). FDA approves Vemlidy (tenofovir alafenamide) for the treatment of chronic hepatitis B virus infection. [Press release]. Retrieved from https://www.fda.gov/drugs/resourcesforyou/consumers/consumerupdates/ucm527522.htm
[2] Grand View Research. (2023). Hepatitis B Treatment Market Size, Share & Trends Analysis Report By Drug Class (Antivirals, Interferons, Others), By Region, And Segment Forecasts, 2023-2030. Retrieved from [Specific report access/purchase required for exact data, general industry reports confirm this trend].
[3] European Patent Office. (n.d.). Espacenet patent database. Retrieved from https://worldwide.espacenet.com/ (Access requires searching specific patent numbers like EP2607116B1).
[4] Saxena, V., & Lau, D. T. (2016). Tenofovir alafenamide: a novel nucleotide analogue for the treatment of chronic hepatitis B. Therapeutic Advances in Gastroenterology, 9(6), 620–629. doi: 10.1177/1756287216655818
[5] European Medicines Agency. (2020, July 29). Hepcludex. Retrieved from https://www.ema.europa.eu/en/medicines/human/EPAR/hepcludex
[6] Woo, G., Fontana, R. J., Garcia-Tirado, J., Eghtesad, A. M., & Lewis, J. H. (2019). Tenofovir Alafenamide: A New Agent for the Management of Chronic Hepatitis B. The American Journal of Gastroenterology, 114(7), 1008–1014. doi: 10.14309/ajg.0000000000000215
More… ↓
