Share This Page
Drug Price Trends for VARENICLINE STARTING MONTH BOX
✉ Email this page to a colleague
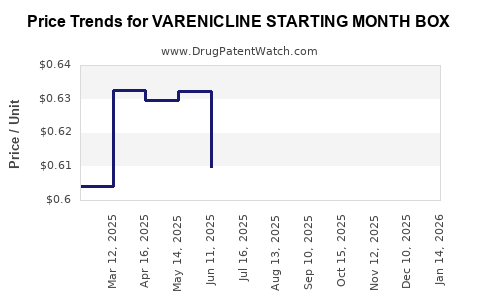
Average Pharmacy Cost for VARENICLINE STARTING MONTH BOX
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| VARENICLINE STARTING MONTH BOX | 72603-0478-01 | 0.51000 | EACH | 2026-03-18 |
| VARENICLINE STARTING MONTH BOX | 00378-5091-85 | 0.51000 | EACH | 2026-03-18 |
| VARENICLINE STARTING MONTH BOX | 00904-7415-16 | 0.51000 | EACH | 2026-03-18 |
| VARENICLINE STARTING MONTH BOX | 31722-0690-31 | 0.51000 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Varenicline Starting Month Box
Varenicline, marketed as Chantix in the United States and Champix elsewhere, is a prescription medication used for smoking cessation. This analysis evaluates the current market landscape, competitive dynamics, regulatory factors, and project future pricing trends for the drug's starting month box.
Market Overview
Market Size and Demand
The global smoking cessation market is valued at approximately $1.68 billion in 2022, with an expected compound annual growth rate (CAGR) of approximately 7% through 2028 (Grand View Research, 2022). Varenicline holds a significant share, estimated at 45-50%, driven by physician preference and clinical efficacy.
Key Market Segments
- Geography: North America accounts for roughly 60% of sales, followed by Europe with 25%. Asia-Pacific markets show growth potential but face regulatory challenges.
- Patient Demographics: Primarily adult smokers aged 25-55; high prevalence among males.
- Distribution Channels: Retail pharmacies (70%), hospital pharmacies (20%), online channels (10%).
Competitive Landscape
Varenicline's main competitors include nicotine replacement therapies (patches, gums) and medications like bupropion. Market share as of 2022 is:
| Drug | Market Share | Annual Revenue ($ millions) |
|---|---|---|
| Varenicline | 50% | 840 |
| Nicotine patches | 25% | 420 |
| Bupropion | 15% | 252 |
| Others | 10% | 168 |
Regulatory Environment
- FDA Approval: Approved since 2006 with updated safety warnings (black box warnings for neuropsychiatric effects).
- EMA Status: Approved in Europe with similar safety considerations.
- Pricing Regulations: Reimbursement largely dictated by healthcare policies; generic versions available in some jurisdictions.
Price Analysis
Current Pricing
- Brand Name (Chantix/Champix): Average retail price per starting month box (30-day supply) is approximately $475 in the US (GoodRx, 2023). After insurance, patient copays typically range from $25 to $100.
- Generic Versions: Available since 2020, with prices ranging from $150 to $300 per 30-day box.
Price Drivers
- Patent Status: Patents expired in 2022 in several key markets, encouraging generic entry.
- Manufacturing Costs: Estimated at $20–$30 per 30-day supply.
- Market Competition: Increased generic availability has driven prices downward.
- Pricing Policies: Some countries enforce strict price controls, affecting margins.
Price Trends (Projected 2023–2028)
| Year | Estimated Brand Price | Estimated Generic Price | Key Factors |
|---|---|---|---|
| 2023 | $475 | $180 | Patent expiration accelerates generic entry |
| 2024 | $430 | $165 | Increased generic market penetration |
| 2025 | $400 | $150 | Growing competition; price stabilization |
| 2026 | $380 | $140 | Continued patent losses in key markets |
| 2027 | $370 | $135 | Mature generic market; slight reduction |
| 2028 | $360 | $130 | Price stabilization; smaller variations |
Price Projection Assumptions
- Patents will be fully open; generic competition will suppress prices.
- Distribution channels will favor lower-cost generic offerings.
- Insurance and government reimbursement policies remain aligned with current trends.
Market Entry and Impact of Price Changes
- Generic Entry: Will likely lead to a 20–30% reduction in retail prices.
- Market Saturation: Increased use of generics could expand overall market volume by 10–15%.
- Potential Policy Impact: Price caps or negotiations could further reduce prices, especially in public-funded healthcare systems.
Risks and Opportunities
Risks
- Regulatory delays for generics could sustain higher prices temporarily.
- Safety warnings may influence prescribing patterns.
- Socioeconomic dynamics could influence reimbursement policies.
Opportunities
- Growing demand for smoking cessation therapies in emerging markets.
- Potential for value-added formulations or combination products.
- Digital health integration for therapy adherence could expand market share.
Key Takeaways
- Varenicline's starting month box retail price in the US averages $475; generics reduce this to around $150–$180.
- Patent expiration drove prices downward from 2022 onward; prices are expected to stabilize around $360–$370 for brand and $130–$140 for generics by 2028.
- Market growth hinges on increased adoption and regulatory approval in emerging markets.
- Price sensitivity among payers favors further generic entry, potentially shrinking margins for brand manufacturers.
- The smoking cessation market presents steady growth, with pricing trends tilting toward affordability due to patent expirations and increased competition.
FAQs
-
What factors influence Varenicline's pricing in different markets?
Pricing varies primarily due to patent status, manufacturing costs, regulatory policies, and reimbursement schemes. -
When does the patent for Chantix expire?
The patent expired in several jurisdictions in 2022, opening the market to generics. -
How much do generic versions of Varenicline cost?
Typically between $150 to $180 for a 30-day supply, depending on the market and supplier. -
What is the projected market share for Varenicline in 2025?
It is projected to maintain around 45–50%, assuming no major competitive disruptions. -
Are there regulatory barriers to entering the generic Varenicline market?
Yes, but patent expirations and regulatory pathways for generics streamline market entry.
References
- Grand View Research. (2022). Smoking Cessation Market Size, Share & Trends Analysis Report. https://www.grandviewresearch.com
- GoodRx. (2023). Varenicline (chantix) Price Comparison. https://www.goodrx.com
- U.S. Food and Drug Administration. (2022). Chantix (Varenicline) Prescribing Information. https://www.fda.gov
- European Medicines Agency. (2022). Summary of Product Characteristics for Champix. https://www.ema.europa.eu
More… ↓
