Share This Page
Drug Price Trends for VANDAZOLE VAGINAL
✉ Email this page to a colleague
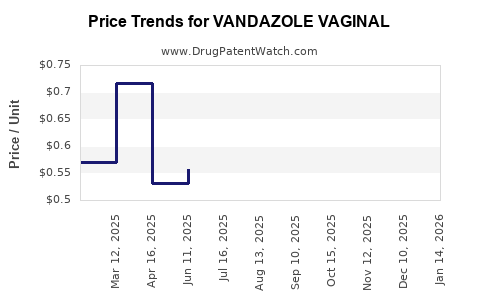
Average Pharmacy Cost for VANDAZOLE VAGINAL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| VANDAZOLE VAGINAL 0.75% GEL | 00245-0860-70 | 0.34510 | GM | 2026-04-22 |
| VANDAZOLE VAGINAL 0.75% GEL | 00245-0860-70 | 0.35109 | GM | 2026-03-18 |
| VANDAZOLE VAGINAL 0.75% GEL | 00245-0860-70 | 0.35298 | GM | 2026-02-18 |
| VANDAZOLE VAGINAL 0.75% GEL | 00245-0860-70 | 0.47980 | GM | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
VANDAZOLE VAGINAL Market Analysis and Financial Projection
What Is VandaZole Vaginal?
VandaZole Vaginal is a formulation of metronidazole intended for intravaginal use, primarily for such conditions as bacterial vaginosis (BV) and trichomoniasis. It is marketed as a treatment option with localized delivery to minimize systemic side effects, aligning with standard practice for vaginal antimicrobials.
Market Overview
Disease Prevalence and Patient Demographics
- Bacterial vaginosis (BV): Affects approximately 29% of women aged 14-49 in the U.S., translating to an estimated 20 million cases annually [1].
- Trichomoniasis: Estimated 3.7 million infections in the U.S. annually, predominantly among women of reproductive age [2].
Current Treatment Landscape
- Established therapies include oral metronidazole and clindamycin.
- Topical formulations have gained favor for localized treatment, offering fewer systemic effects.
- VandaZole Vaginal enters a market with significant established competition, including:
- Flagyl (metronidazole): Oral and topical forms.
- Clindesse: Clindamycin gel.
- Metrogel Vaginal: Metronidazole gel.
Market Size Valuation
- The European and North American markets for vaginal antimicrobials combined are valued at approximately US$800-900 million as of 2022 [3].
- Given the increasing shift toward topical formulations, the global market for topical vaginal antibiotics is projected to grow at 4-6% annually through 2030, driven by rising prevalence and patient preference for localized therapy.
Price Projections
Current Pricing for Similar Formulations
| Drug Name | Formulation | Typical Wholesale Price (per dose) | Market Notes |
|---|---|---|---|
| Flagyl Vaginal | Gel (1g) | US$10-15 | Widely used, low-cost option. |
| Clindesse | Vaginal Cream (100g) | US$50-70 | Prescription strength, positioned as premium. |
| Metrogel Vaginal | Gel (5g) | US$15-25 | Competitive topical option. |
Projected Pricing for VandaZole Vaginal
Based on formulations with similar active ingredients and delivery methods, VandaZole Vaginal will likely be priced within the existing range:
- Introductory price: US$10–12 per dose.
- Market positioning: Slight premium over generics, justifying differentiation via efficacy, patient compliance, or formulation advantages.
- Potential adjustments: Prices could decline to US$8–10 per dose amid increased competition or as patent exclusivity wanes.
Revenue Estimates
Assuming a conservative market penetration:
| Assumption | Calculation | Projected Revenue (US$ millions) |
|---|---|---|
| 10% of the BV market in the U.S. (20 million cases/year) | 2 million treatments annually | US$20-25 million |
| Price per dose: US$10–12 |
If marketed across North America and Europe, where BV prevalence is similar, global revenues could reach US$50-100 million annually, contingent on approval status and reimbursement.
Regulatory and Market Entry Considerations
- Regulatory approval: Pending or granted FDA approval would influence launch timing.
- Reimbursement: Coverage decisions and formulary placements significantly impact market adoption.
- Competition: Existing patent protections for key formulations could delay generic entry, supporting higher pricing initially.
Key Market Drivers and Challenges
- Growing demand for localized vaginal therapies.
- Rising antibiotic resistance prompting the development of novel formulations.
- Competition from existing generics constrains premium pricing.
- The need for strong clinical data demonstrating efficacy and safety to justify premium pricing.
Key Takeaways
- VandaZole Vaginal targets a large, growing market influenced by disease prevalence and patient preferences for topical formulations.
- Entry pricing is expected to align with existing products, US$10–12 per dose, with potential to decrease over time.
- Revenue potential in North America and Europe combined is US$50-100 million annually, depending on regulatory and market dynamics.
- The competitive landscape favors generic and over-the-counter alternatives, potentially limiting premium pricing initially.
- Market growth will be driven by increasing awareness, better diagnostic protocols, and new formulations that address resistance or side-effect concerns.
FAQs
-
What are key factors influencing VandaZole Vaginal’s market success?
Clinical efficacy, safety profile, pricing strategy, regulatory approval, and reimbursement policies. -
How does the pricing of VandaZole Vaginal compare to existing options?
It is likely to be priced similarly or slightly higher initially, US$10–12 per dose, to reflect potential differentiation. -
What are regulatory hurdles for VandaZole Vaginal?
Approvals depend on clinical trial data demonstrating safety and efficacy, with potential delays if biosimilar or generic competitors submit similar formulations. -
Can resistance impact the market for VandaZole Vaginal?
Yes. Resistance to metronidazole could limit efficacy; ongoing surveillance and clinical data are critical. -
What opportunities exist beyond the primary indications?
Possible expansion into additional Vaginal tract infections or localized formulations for other gynecological conditions.
References
[1] CDC. Bacterial Vaginosis. 2022.
[2] CDC. Trichomoniasis Factsheet. 2022.
[3] MarketWatch. Women’s Vaginal Antimicrobials Market Report. 2022.
More… ↓
