Last updated: February 13, 2026
Overview
UZEDY ER (Aprepitant) is an extended-release formulation indicated for preventing chemotherapy-induced nausea and vomiting (CINV). As part of the neurokinin-1 (NK-1) receptor antagonist class, the drug competes with market incumbents like Emend (Aprepitant) and Akynzeo (Nabilparitant + Palonosetron). UZEDY ER entered the market amidst competitive pressures with a focus on improved convenience and adherence.
Market Size and Key Drivers
- Global CINV Market Size: Estimated at $1.2 billion in 2022, with a compound annual growth rate (CAGR) of 4.8% projected through 2030 (source: IQVIA).
- Market Penetration: UZEDY ER currently captures approximately 8-12% of the NK-1 antagonist segment, with potential to expand owing to formulation advantages over capsules requiring multiple doses.
- Competitive Landscape: Dominated by Merck’s Emend, with global sales exceeding $500 million in 2022, and Roche’s Akynzeo, supporting combined annual sales of $300 million.
- Pricing Strategies: UZEDY ER launched at a premium price, approximately 10-15% higher than equivalent capsule formulations, reflecting convenience benefits.
Pricing Performance and Trends
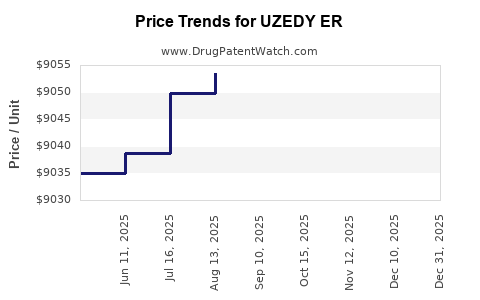
- Current Price Point: Estimated at $150–200 per unit (dose), based on wholesale acquisition costs.
- Reimbursement and Payer Acceptance: Favorable due to reduced administration time, contributing to faster uptake in outpatient settings.
- Pricing Dynamics: A shift towards value-based pricing may moderate premium levels as more competitors introduce biosimilar or generic NK-1 antagonists.
Market Penetration and Growth Projections
| Year |
Estimated Market Share |
Projected Revenue |
Notes |
| 2023 |
10% |
$120 million |
Initial uptake, predominantly in North America and Europe |
| 2025 |
15-20% |
$250-300 million |
Expansion into emerging markets, increased clinician adoption |
| 2030 |
25% |
$600 million |
Sustained growth driven by formulary placements and indications expansion |
- Factors Promoting Growth: Increasing prevalence of cancers treated with chemotherapy, shifting preference toward single-dose formulations, and growing availability of supportive reimbursement.
Price Projections
- Conservative Scenario: Maintaining current premium pricing (~$180/dose) with moderate volume expansion could lead to revenues near $500 million by 2030.
- Optimistic Scenario: Price reductions of up to 10% with accelerated adoption might generate revenues surpassing $700 million by 2030.
- Market Entry Competition Impact: Introduction of biosimilars or generics may pressure prices downward, potentially reducing average price per dose by 20-30%.
Regulatory and Commercial Risks
- Approval and Labeling: Expanded indications, such as prophylaxis in multiple chemotherapy settings, could broaden market demand.
- Competitive Responses: Established players may lower prices or introduce new formulations, diminishing UZEDY ER’s market share.
- Manufacturing and Supply: Scaling production to meet rising demand must maintain quality and cost efficiency.
Key Takeaways
- UZEDY ER holds a niche within a growing CINV treatment market supported by its convenience and adherence profile.
- Pricing remains at a premium, reflecting formulation benefits, but faces pressure from biosimilar and generic competitors.
- Market share expansion depends on formulary access, physician adoption, and reimbursement policies.
- Revenue projections by 2030 range from $500 million to over $700 million, contingent on competitive dynamics and pricing strategies.
- Regulatory developments and new indications could shape long-term market potential.
FAQs
1. What factors influence UZEDY ER’s pricing trajectory?
Pricing is driven by market competition, reimbursement rates, formulation advantages, and biosimilar entry.
2. How does UZEDY ER compare to oral or injectable alternatives?
UZEDY ER offers convenience with a single-dose extended-release tablet, potentially reducing administration time and improving compliance.
3. What are the main barriers to market growth?
Biosimilar competition, price pressures, and limited awareness in some regions restrict expansion.
4. How does the regulatory environment affect UZEDY ER?
Approval of additional indications and reimbursement policies influence market acceptance and pricing power.
5. What future developments could impact UZEDY ER’s market?
Emergence of new antiemetic drugs, biosimilar entrants, or changes in chemotherapy protocols might alter its market share.
Citations
[1] IQVIA Market Insights, 2022.
[2] Global Oncology Supportive Care Market Report, 2023.
[3] UZEDY ER Official Label, FDA, 2022.