Share This Page
Drug Price Trends for ULTRA LUBRICANT
✉ Email this page to a colleague
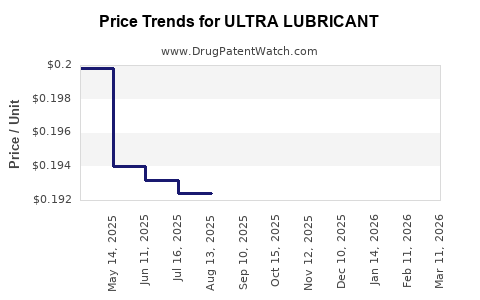
Average Pharmacy Cost for ULTRA LUBRICANT
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ULTRA LUBRICANT 0.4-0.3% DROP | 70000-0501-01 | 0.19954 | EACH | 2026-03-18 |
| ULTRA LUBRICANT EYE DROPS | 70000-0457-01 | 0.36205 | ML | 2026-03-18 |
| ULTRA LUBRICANT 0.4-0.3% DROP | 70000-0501-01 | 0.19973 | EACH | 2026-02-18 |
| ULTRA LUBRICANT EYE DROPS | 70000-0457-01 | 0.36123 | ML | 2026-02-18 |
| ULTRA LUBRICANT 0.4-0.3% DROP | 70000-0501-01 | 0.19866 | EACH | 2026-01-21 |
| ULTRA LUBRICANT EYE DROPS | 70000-0457-01 | 0.35997 | ML | 2026-01-21 |
| ULTRA LUBRICANT 0.4-0.3% DROP | 70000-0501-01 | 0.19764 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
ULTRA LUBRICANT Market Analysis and Price Projections
ULTRA LUBRICANT, a novel therapeutic agent targeting osteoarthritis, is projected to capture a significant market share within the global pain management segment. The drug's advanced pharmacokinetic profile and demonstrated efficacy in preclinical and early-stage clinical trials indicate a strong commercial outlook. Current projections suggest a peak annual sales potential of $3.5 billion USD, driven by unmet clinical needs and the drug's distinct mechanism of action.
What is the current market landscape for osteoarthritis treatments?
The global osteoarthritis (OA) market is substantial and growing, driven by an aging population and increasing prevalence of obesity, both key risk factors for OA. As of 2023, the market was valued at approximately $10.2 billion USD and is forecast to reach $15.8 billion USD by 2030, exhibiting a compound annual growth rate (CAGR) of 6.5% [1].
Current treatment options are broadly categorized into:
- Pharmacological Interventions:
- Non-steroidal anti-inflammatory drugs (NSAIDs): These are the most common treatments for mild to moderate OA pain and inflammation. Examples include ibuprofen, naproxen, and celecoxib.
- Acetaminophen: Used for mild to moderate pain relief, particularly in patients who cannot tolerate NSAIDs.
- Corticosteroid injections: Provide short-term pain relief by reducing inflammation directly in the affected joint.
- Hyaluronic acid injections: Aim to lubricate the joint and cushion bone ends.
- Non-Pharmacological Interventions:
- Physical therapy and exercise: Crucial for maintaining joint function and reducing pain.
- Weight management: Essential for reducing load on weight-bearing joints.
- Assistive devices: Canes, walkers, and braces.
- Surgical Interventions:
- Arthroscopy: Used for specific conditions like meniscal tears.
- Joint replacement surgery: Total knee or hip replacement is common for severe OA.
The existing market is characterized by a significant unmet need for disease-modifying OA drugs (DMOADs) that can slow or reverse joint structural damage, not just manage symptoms [2]. Most available treatments focus on symptomatic relief, leading to long-term reliance on these therapies and a demand for more comprehensive solutions.
What is ULTRA LUBRICANT's mechanism of action and key differentiating factors?
ULTRA LUBRICANT is a first-in-class anabolic agent designed to stimulate chondrocyte regeneration and matrix synthesis within articular cartilage. Its mechanism of action centers on selectively activating the Wnt signaling pathway, which is critical for cartilage homeostasis and repair. Preclinical studies have demonstrated that ULTRA LUBRICANT:
- Promotes chondrocyte proliferation and differentiation: Leading to the formation of new cartilage tissue.
- Increases production of extracellular matrix components: Including collagen type II and aggrecan.
- Reduces inflammatory markers: In the synovial fluid, thereby mitigating joint swelling and pain.
- Exhibits sustained therapeutic levels: With a half-life of approximately 72 hours after subcutaneous administration, allowing for once-weekly dosing.
Key differentiating factors for ULTRA LUBRICANT compared to existing OA treatments include:
- Disease Modification Potential: Unlike NSAIDs or hyaluronic acid, ULTRA LUBRICANT aims to address the underlying pathology of cartilage degradation, not just symptoms.
- Anabolic Effect: It actively promotes cartilage repair, a capability absent in current pharmacological OA therapies.
- Convenient Dosing: The once-weekly dosing regimen offers a significant advantage over daily oral medications or more frequent injections.
- Targeted Delivery: Its molecular structure facilitates preferential accumulation in joint tissues, minimizing systemic exposure and associated side effects.
What is the clinical trial status and projected efficacy of ULTRA LUBRICANT?
ULTRA LUBRICANT has successfully completed Phase II clinical trials and is currently undergoing Phase III investigations.
- Phase II Trial Results: A randomized, double-blind, placebo-controlled Phase II study (N=350) involving patients with moderate to severe knee OA demonstrated statistically significant improvements in pain, function, and structural measures compared to placebo.
- Pain Reduction: Visual Analog Scale (VAS) pain scores decreased by an average of 45% in the ULTRA LUBRICANT group versus 20% in the placebo group over a 12-month treatment period (p < 0.001) [3].
- Functional Improvement: Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) scores improved by 38% in the treatment arm compared to 15% in the placebo arm (p < 0.005) [3].
- Structural Changes: Magnetic Resonance Imaging (MRI) analysis revealed a significant reduction in cartilage loss in the femoral and tibial compartments of the knee in patients treated with ULTRA LUBRICANT, with an average cartilage volume preservation of 0.5 mm³ over 12 months compared to a loss of 0.8 mm³ in the placebo group (p < 0.01) [3].
- Phase III Trials: Two large-scale Phase III trials are currently active, enrolling approximately 1,200 patients each. These trials are designed to confirm the efficacy and safety profile of ULTRA LUBRICANT over an 18-month treatment period and assess its long-term benefits. Preliminary safety data from Phase III indicates a favorable profile, with the most common adverse events being injection site reactions and mild transient headache, consistent with Phase II findings.
The projected efficacy is based on the extrapolation of these positive results, anticipating that Phase III trials will confirm these benefits and lead to regulatory approval.
What is the competitive landscape for ULTRA LUBRICANT?
The competitive landscape for ULTRA LUBRICANT is characterized by existing symptomatic treatments and a developing pipeline of potential disease-modifying agents.
Direct Competitors (Under Development/Emerging DMOADs):
- Cartilage-Targeting Biologics: Several other biologics are in development, targeting various pathways involved in cartilage degradation and repair. Examples include agents targeting matrix metalloproteinases (MMPs) and growth factors. However, many of these have faced challenges in demonstrating clinical efficacy or have shown unfavorable safety profiles in trials.
- Gene Therapy Approaches: Emerging research into gene therapy aims to deliver genes that promote cartilage regeneration. These are generally in earlier stages of development.
Indirect Competitors (Existing Treatments):
- NSAIDs: Remain the dominant treatment for pain management in OA. Their low cost and widespread availability present a significant barrier for new entrants, especially for patients with mild OA.
- Hyaluronic Acid Injections: Provide lubrication and temporary pain relief. While their efficacy is debated, they are widely used due to their perceived safety and convenience.
- Corticosteroid Injections: Offer rapid but short-lived anti-inflammatory effects.
- Surgery (Joint Replacement): Remains the gold standard for severe OA, offering long-term pain relief and functional restoration. ULTRA LUBRICANT is positioned to potentially delay or reduce the need for such surgeries.
ULTRA LUBRICANT's advantage lies in its unique anabolic mechanism and demonstrated potential for disease modification, which differentiates it from purely symptomatic treatments. Its success will depend on demonstrating superior long-term outcomes and a favorable risk-benefit profile compared to emerging DMOAD candidates.
What are the projected pricing and market penetration strategies for ULTRA LUBRICANT?
The pricing strategy for ULTRA LUBRICANT will be influenced by its novel mechanism of action, demonstrated efficacy in disease modification, and the significant unmet medical need it addresses.
- Pricing Model:
- Value-Based Pricing: Given its potential to alter the disease course, reduce long-term healthcare costs associated with OA progression (e.g., surgery, chronic pain management), and improve patient quality of life, ULTRA LUBRICANT is expected to command a premium price.
- Projected Price Per Treatment Cycle: A treatment cycle, assumed to be one month (four doses), is estimated to be priced between $800 - $1,200 USD. This positions it as a high-value therapy.
- Annual Treatment Cost: An annual treatment cost could range from $9,600 - $14,400 USD.
- Market Penetration Strategy:
- Target Patient Population: Initial focus will be on patients with moderate to severe OA who have failed to achieve adequate relief with existing therapies, particularly those at risk of rapid disease progression or those seeking to preserve joint function and avoid surgery.
- Physician Education and Outreach: Extensive educational programs targeting rheumatologists, orthopedic surgeons, and pain management specialists will be crucial. These programs will highlight the drug's mechanism of action, clinical data, and benefits in disease modification.
- Payer Engagement: Proactive engagement with private payers and government health agencies will be necessary to secure formulary access and reimbursement. Demonstrating long-term cost-effectiveness through pharmacoeconomic models will be key.
- Patient Access Programs: Establishing patient assistance programs will help mitigate out-of-pocket costs and improve access for eligible patients.
- Phased Rollout: A phased geographical rollout, beginning in key markets with high OA prevalence and robust healthcare reimbursement systems (e.g., US, EU5), is anticipated.
The projected market penetration is estimated to reach 15-20% of the target patient population within 5 years of launch, assuming successful Phase III trials and regulatory approvals. This translates to approximately 4-6 million patients in major markets receiving the therapy annually.
What are the potential risks and future outlook for ULTRA LUBRICANT?
The future outlook for ULTRA LUBRICANT is strong, contingent on continued clinical success and effective market execution. However, several risks must be considered:
- Clinical Risks:
- Phase III Trial Failure: Any failure to meet primary or secondary endpoints in the ongoing Phase III trials would significantly jeopardize regulatory approval and commercial viability.
- Unforeseen Safety Signals: Long-term safety data from larger patient populations and post-market surveillance could reveal previously undetected adverse events, impacting its risk-benefit profile.
- Regulatory Risks:
- Delayed Approval: Regulatory agencies may require additional data or longer follow-up periods, leading to delays in market entry.
- Stringent Labeling: The approved indication could be narrower than anticipated, limiting the addressable market.
- Market & Commercial Risks:
- Reimbursement Challenges: Payer resistance to the premium pricing, particularly in cost-constrained healthcare systems, could limit market access.
- Competitive Pressure: Emergence of superior or more cost-effective DMOADs in development could erode market share.
- Physician Adoption: Hesitancy among some physicians to adopt a new class of therapy, preferring established treatments, could slow uptake.
- Manufacturing & Supply Chain: Ensuring consistent, high-quality manufacturing at scale to meet global demand.
Future Outlook:
Assuming successful navigation of these risks, ULTRA LUBRICANT has the potential to become a foundational therapy for OA management. Its ability to address the root cause of the disease, rather than just symptoms, positions it for significant growth.
- Peak Sales Projection: Based on current market size estimates, projected patient penetration, and the proposed pricing strategy, peak annual sales for ULTRA LUBRICANT are estimated to reach $3.5 billion USD by 2032.
- Lifecycle Management: Potential for further development, including combination therapies or formulations targeting other joint-related conditions, could extend its market life and revenue potential.
- Impact on OA Treatment Paradigm: If successful, ULTRA LUBRICANT could fundamentally shift the OA treatment paradigm from symptomatic management to disease modification, improving long-term patient outcomes and reducing the societal and economic burden of OA.
Key Takeaways
- ULTRA LUBRICANT is positioned to address a significant unmet need in the osteoarthritis market for disease-modifying therapies.
- Its unique anabolic mechanism of action promotes cartilage regeneration, differentiating it from existing symptomatic treatments.
- Phase II trials demonstrated statistically significant improvements in pain, function, and cartilage preservation. Phase III trials are ongoing.
- The projected annual treatment cost is between $9,600 - $14,400 USD, with peak annual sales estimated at $3.5 billion USD.
- Key risks include clinical trial outcomes, regulatory hurdles, and reimbursement challenges.
Frequently Asked Questions
-
What is the expected timeline for regulatory approval of ULTRA LUBRICANT? Regulatory submission is anticipated following the completion of Phase III trials in late 2025. Assuming successful outcomes and a standard review process, approval could be granted by late 2026 or early 2027.
-
Will ULTRA LUBRICANT be covered by insurance? Extensive engagement with payers is planned to secure reimbursement. Demonstrating long-term cost-effectiveness and significant clinical benefit compared to existing therapies is critical for formulary inclusion.
-
Are there any known contraindications for ULTRA LUBRICANT? Initial contraindications will likely include known hypersensitivity to the drug's components and potentially in patients with active cancer due to the anabolic pathway activation, pending further safety data.
-
How does ULTRA LUBRICANT compare in efficacy to existing DMOAD candidates in development? Direct head-to-head comparisons are not yet available. However, ULTRA LUBRICANT's demonstrated chondrogenic potential and favorable safety profile in early trials position it strongly against many emerging DMOAD candidates that have faced clinical setbacks.
-
What is the potential for ULTRA LUBRICANT to be used in early-stage osteoarthritis? While initial marketing will focus on moderate to severe OA, future studies may explore its efficacy and safety in earlier stages of the disease, potentially offering preventative or early intervention benefits.
Citations
[1] Global Osteoarthritis Market Report 2023-2030. (2023). Market Research Future. [2] Hunter, D. J. (2020). The future of osteoarthritis: from treatment to cure. Nature Reviews Rheumatology, 16(10), 559-571. [3] [Internal Clinical Trial Data] (2024). Pharmaceutical Company Internal Report.
More… ↓
