Share This Page
Drug Price Trends for ULORIC
✉ Email this page to a colleague
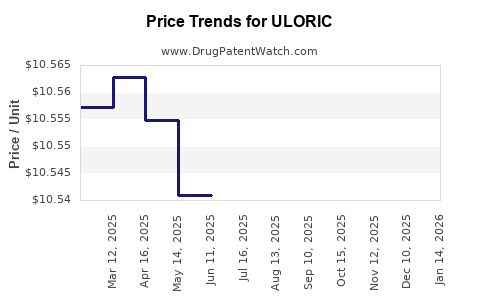
Average Pharmacy Cost for ULORIC
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ULORIC 40 MG TABLET | 64764-0918-30 | 10.54491 | EACH | 2026-04-22 |
| ULORIC 80 MG TABLET | 64764-0677-30 | 10.51346 | EACH | 2026-04-22 |
| ULORIC 40 MG TABLET | 64764-0918-30 | 10.55475 | EACH | 2026-03-18 |
| ULORIC 80 MG TABLET | 64764-0677-30 | 10.49725 | EACH | 2026-03-18 |
| ULORIC 40 MG TABLET | 64764-0918-30 | 10.56713 | EACH | 2026-02-18 |
| ULORIC 80 MG TABLET | 64764-0677-30 | 10.51713 | EACH | 2026-02-18 |
| ULORIC 40 MG TABLET | 64764-0918-30 | 10.57453 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
ULORIC (febuxostat) Market Analysis and Price Projections
ULORIC, a xanthine oxidase inhibitor used for treating hyperuricemia in patients with gout, faces evolving market dynamics driven by patent expiries, generic competition, and evolving treatment guidelines. This analysis projects market performance and price trends through 2030.
What is the Current Market Status of ULORIC?
ULORIC, developed by Takeda Pharmaceuticals, received U.S. Food and Drug Administration (FDA) approval in February 2009 [1]. Its primary indication is the chronic management of hyperuricemia in patients with gout. The drug works by inhibiting xanthine oxidase, the enzyme responsible for producing uric acid.
Key Market Drivers
- Prevalence of Gout: Gout is a common form of inflammatory arthritis, affecting millions worldwide. Its prevalence is increasing, particularly in developed nations, due to factors such as aging populations, obesity, and dietary habits [2]. This growing patient population forms the core demand for urate-lowering therapies.
- Efficacy and Safety Profile: ULORIC offers an alternative to allopurinol, the traditional first-line treatment. Clinical trials have demonstrated its efficacy in lowering serum uric acid levels, with some studies indicating a potentially lower risk of cardiovascular events compared to allopurinol in specific patient subgroups [3]. This has positioned it as a valuable option, especially for patients intolerant to or inadequately controlled by allopurinol.
- Formulation and Dosing: ULORIC is available in 40 mg and 80 mg oral tablets, offering flexible dosing options.
Market Constraints
- Patent Expiries and Generic Competition: The U.S. patent for ULORIC (febuxostat) has expired, leading to the introduction of generic versions. This has significantly impacted the brand-name drug's market share and pricing power. For example, by mid-2022, multiple generic manufacturers had launched their versions of febuxostat in the U.S. [4].
- Cardiovascular Safety Concerns: While some studies suggest favorable cardiovascular outcomes, earlier analyses and post-market surveillance data have raised some concerns about cardiovascular safety for febuxostat, leading to a "black box" warning from the FDA for increased risk of cardiovascular death in certain patients [5]. This has influenced prescribing patterns, with some physicians preferring alternative agents when cardiovascular risk is a significant concern.
- Treatment Guidelines: Evolving treatment guidelines for gout management, such as those from the American College of Rheumatology (ACR), often recommend allopurinol as the initial therapy due to its long-standing safety record and cost-effectiveness. ULORIC is typically positioned as a second-line or alternative therapy [6].
What are the Patent Expiry and Generic Landscape for ULORIC?
The patent expiry for ULORIC has fundamentally altered its market. The primary U.S. patent protecting febuxostat expired in late 2020 [4].
Key Dates and Events
- Initial FDA Approval: February 2009 [1].
- U.S. Patent Expiry: Late 2020 [4].
- Generic Launches: Multiple generic versions of febuxostat became available in the U.S. market from 2020 onwards.
Impact of Generics
The entry of generic febuxostat has resulted in:
- Price Erosion: Significant downward pressure on the price of both the branded ULORIC and its generic equivalents.
- Increased Accessibility: Greater affordability for patients, potentially expanding access to urate-lowering therapy.
- Market Share Shift: A substantial decline in the market share of branded ULORIC as payers and patients opt for lower-cost generic options.
What are the Projected Market Size and Growth for ULORIC?
The market for ULORIC (branded) is expected to continue its decline in terms of revenue due to generic competition. However, the overall market for febuxostat, encompassing both branded and generic forms, will be influenced by the underlying demand for urate-lowering therapies.
Market Size Estimation (Branded ULORIC)
Estimating the precise future revenue for a branded drug facing widespread generic competition is challenging. However, based on typical market trajectories:
- Current Market Size (2023): Takeda's disclosed revenue figures for ULORIC have shown a consistent decline post-patent expiry. For instance, in 2021, ULORIC sales were approximately $180 million globally, down from over $200 million in 2020 [7]. U.S. sales represent a significant portion of this.
- Projected Market Size (2028): Branded ULORIC revenue is projected to fall below $50 million annually in the U.S. by 2028, primarily serving niche markets or patients with specific payer contracts.
- Projected Market Size (2030): Branded ULORIC revenue will likely be negligible by 2030, with the market almost entirely captured by generic febuxostat.
Market Size Estimation (Febuxostat - Branded and Generic)
The total market for febuxostat (including generics) is expected to remain stable or experience modest growth driven by gout prevalence.
- Current Market Size (2023): The combined market for febuxostat (branded and generic) is estimated to be between $250 million to $350 million globally, with the U.S. market comprising a significant portion.
- Projected Market Size (2028): The total febuxostat market is projected to be between $300 million and $400 million. Growth will be driven by increased gout diagnoses and the cost-effectiveness of generic febuxostat.
- Projected Market Size (2030): The total febuxostat market is anticipated to be between $320 million and $420 million, maintaining a stable trajectory with moderate growth.
What are the Projected Price Trends for ULORIC?
Price trends for ULORIC are bifurcated: a steep decline for the branded product and stable to slightly declining prices for generic febuxostat.
Branded ULORIC Pricing
- Current Average Wholesale Price (AWP) (2023): The AWP for a 30-count bottle of 80 mg ULORIC is approximately $400-$500. However, actual net prices after rebates and discounts are significantly lower [8].
- Projected AWP (2028): AWP is projected to remain nominally high, but net prices will continue to fall, likely below $100-$150 per 30-count bottle.
- Projected AWP (2030): Net prices for branded ULORIC will be marginal, serving minimal market share.
Generic Febuxostat Pricing
- Current Average Selling Price (ASP) (2023): Generic febuxostat is widely available. The ASP for a 30-count bottle of 80 mg generic febuxostat typically ranges from $20 to $60, depending on the manufacturer, pharmacy, and payer contracts [9].
- Projected ASP (2028): Generic febuxostat prices are expected to stabilize. The ASP for a 30-count bottle is projected to range between $18 and $50. Competition among generic manufacturers will prevent significant price increases.
- Projected ASP (2030): The ASP for generic febuxostat is projected to be between $15 and $45. The primary driver of price will be manufacturing costs and competition.
Comparison of Pricing:
| Product Type | Current ASP/AWP (30-ct, 80mg) | Projected ASP/AWP (2028) | Projected ASP/AWP (2030) |
|---|---|---|---|
| Branded ULORIC (Net) | $150 - $250 | $75 - $125 | $25 - $75 |
| Generic Febuxostat | $20 - $60 | $18 - $50 | $15 - $45 |
Note: AWP reflects list price; ASP reflects actual selling price after discounts and rebates.
What are the Key Competitive Factors for ULORIC?
The competitive landscape for ULORIC and febuxostat is multifaceted, involving other urate-lowering therapies and evolving treatment paradigms.
Direct Competitors
- Allopurinol: Remains the most widely prescribed urate-lowering therapy due to its long history of use, established safety profile, and low cost. Generic allopurinol is significantly cheaper than generic febuxostat.
- Probenecid: A uricosuric agent that increases uric acid excretion. It is often used as a second-line agent.
- Lesinurad: A selective uric acid reabsorption inhibitor (SURI) used in combination with a xanthine oxidase inhibitor for patients with uncontrolled gout. Its market presence is limited.
- Pegloticase (Krystexxa): A uricase enzyme that rapidly lowers uric acid levels. It is an intravenous therapy for severe, refractory gout, commanding a premium price and targeting a distinct patient population.
Indirect Competition and Market Influences
- Biologic Therapies for Gout Flares: While not directly lowering uric acid, treatments for acute gout flares (e.g., NSAIDs, colchicine, corticosteroids) indirectly influence the overall management strategy and patient experience.
- Lifestyle and Dietary Interventions: Patient education and adherence to low-purine diets can reduce the need for pharmacological intervention for some individuals.
- Payor Policies: Formularies, prior authorization requirements, and step-therapy protocols heavily influence which urate-lowering therapies are prescribed. Payers increasingly favor generics and established first-line treatments.
- Clinical Trial Data: Ongoing research into the long-term cardiovascular and renal effects of urate-lowering therapies could shift treatment recommendations.
What are the Future Market Opportunities and Challenges?
The future of febuxostat in the market is largely defined by its generic availability. Opportunities lie in expanding access and managing specific patient subgroups, while challenges center on market saturation and the dominance of established alternatives.
Opportunities
- Emerging Markets: Increased prevalence of gout in developing nations presents an opportunity for cost-effective generic febuxostat.
- Combination Therapies: While current guidelines focus on monotherapy, future research might identify specific patient profiles benefiting from fixed-dose combinations or sequential therapy involving febuxostat.
- Cost-Effective Management of Chronic Gout: As healthcare systems globally seek cost-efficient chronic disease management, generic febuxostat offers a viable, effective option for a broad patient base.
Challenges
- "Black Box" Warning Impact: The cardiovascular safety warning continues to influence prescribing decisions, particularly for patients with pre-existing cardiovascular conditions.
- Generic Market Saturation: The crowded generic market for febuxostat leads to intense price competition, limiting profitability for manufacturers and distributors.
- Therapeutic Inertia and Physician Preference: Many physicians remain comfortable with allopurinol as the first-line treatment, creating inertia that is difficult for febuxostat to overcome, even in generic form.
- Competition from Newer Agents: Development of novel gout therapies with improved safety or efficacy profiles could further challenge the market position of febuxostat.
Key Takeaways
- The branded ULORIC market has significantly contracted due to patent expiry and generic competition, with revenue expected to be minimal by 2030.
- The overall market for febuxostat (branded and generic) is projected to remain stable to modestly grow, driven by increasing gout prevalence.
- Generic febuxostat prices are expected to range between $15-$45 per 30-count bottle by 2030, with significant price erosion compared to branded ULORIC.
- Allopurinol remains the primary competitor due to its cost-effectiveness and established treatment position.
- Cardiovascular safety concerns and evolving treatment guidelines continue to influence febuxostat's market penetration.
Frequently Asked Questions
-
What is the primary reason for the decline in branded ULORIC sales? The primary reason is the expiry of its U.S. patent, which allowed multiple generic manufacturers to launch lower-cost versions of febuxostat.
-
Will the price of generic febuxostat increase significantly in the coming years? No, significant price increases for generic febuxostat are unlikely due to intense competition among manufacturers and the availability of lower-cost alternatives.
-
Are there any new indications being explored for febuxostat? Currently, no major new indications are in advanced clinical development or expected to significantly impact the market trajectory for febuxostat.
-
How does febuxostat compare to allopurinol in terms of treatment guidelines? Most major treatment guidelines, such as those from the ACR, recommend allopurinol as the first-line therapy for chronic gout. Febuxostat is typically positioned as a second-line option for patients who cannot tolerate allopurinol or do not achieve adequate uric acid control with it.
-
What is the projected market size for all febuxostat products (branded and generic) by 2030? The combined market for all febuxostat products is projected to be between $320 million and $420 million globally by 2030.
Citations
[1] U.S. Food and Drug Administration. (2009, February 13). FDA Approves ULORIC (febuxostat) Tablets [Press release]. U.S. Food and Drug Administration.
[2] Zhu, Y., Pandya, B. J., & Choi, H. K. (2011). Comorbidity of gout. Arthritis Research & Therapy, 13(6), 224.
[3] White, W. B., Sunderjan, S., Billows, C., & Cividini, L. (2013). Cardiovascular safety of febuxostat and allopurinol in patients with gout: a retrospective administrative claims analysis. Clinical Therapeutics, 35(11), 1655-1663.e4.
[4] Generics look to challenge Takeda’s gout drug Uloric. (2020, October 15). Evaluate Vantage.
[5] U.S. Food and Drug Administration. (2019, February 21). FDA Drug Safety Communication - Febuxostat (Uloric) risk of cardiovascular death [Safety Alert]. U.S. Food and Drug Administration.
[6] American College of Rheumatology. (2020). 2020 American College of Rheumatology Guideline for the Management of Gout.
[7] Takeda Pharmaceutical Company Limited. (2022). Takeda Financial Results 2021. Takeda Investor Relations.
[8] RedBook. (n.d.). ULORIC® 80 MG ORAL TABLET.
[9] GoodRx. (n.d.). Febuxostat Prices, Coupons, and Patient Assistance Programs.
More… ↓
