Share This Page
Drug Price Trends for UCERIS
✉ Email this page to a colleague
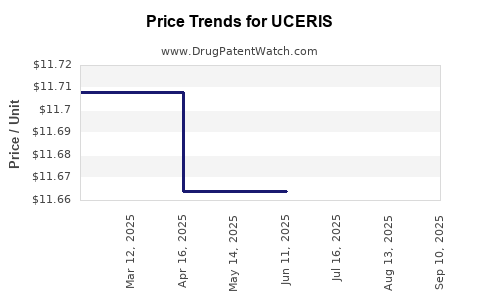
Average Pharmacy Cost for UCERIS
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| UCERIS 9 MG ER TABLET | 68012-0309-30 | 57.16796 | EACH | 2025-09-17 |
| UCERIS 2 MG RECTAL FOAM | 65649-0651-03 | 11.62772 | GM | 2025-09-17 |
| UCERIS 2 MG RECTAL FOAM | 65649-0651-03 | 11.62006 | GM | 2025-08-20 |
| UCERIS 9 MG ER TABLET | 68012-0309-30 | 57.15124 | EACH | 2025-08-20 |
| UCERIS 9 MG ER TABLET | 68012-0309-30 | 57.16346 | EACH | 2025-07-23 |
| UCERIS 2 MG RECTAL FOAM | 65649-0651-03 | 11.62937 | GM | 2025-07-23 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for UCERIS
Overview of UCERIS
UCERIS (budesonide) is a locally acting corticosteroid approved for the treatment of mild to moderate ulcerative colitis (UC). It is delivered via a targeted-release formulation designed to mitigate systemic corticosteroid side effects by releasing the drug in the distal ileum and colon.
Current Market Position
UCERIS is marketed by Siena Biopharmaceuticals in the US, with global distribution partnerships. Since its approval by the FDA in 2013, UCERIS has gained a niche market for induction therapy in UC, primarily for patients with mild to moderate disease who prefer oral corticosteroid therapy before escalating to biologics or immunomodulators.
Market Size and Growth Potential
According to IQVIA data, the US ulcerative colitis treatment market was valued at approximately $2.1 billion in 2022. UCERIS captures a specific segment within this, with estimated sales of $300 million in 2022, representing roughly 14% of the total UC market.
The inflating incidence of UC, especially in North America and Europe, suggests steady growth in overall market size. The prevalence of UC in North America is around 290 per 100,000 people, with an estimated 1 million diagnosed cases in the US, and similar trends observed in Europe.
Key drivers influencing growth include:
- Increased adoption of oral corticosteroids for induction therapy
- Growing preference for targeted-release formulations
- The shift towards personalized medicine reducing reliance on systemic steroids
- Expansion of UC diagnosis rates
Competitive Landscape
UCERIS faces competition from other corticosteroids, biologic therapies (e.g., infliximab, vedolizumab), and small molecules, such as:
- Corticosteroids: Prednisone, budesonide (other formulations)
- Biologics: Humira (adalimumab), Remicade (infliximab)
- Small molecules: tofacitinib, ozanimod
UCERIS’s differentiator remains its targeted-release profile, which reduces systemic steroid exposure. However, biologics dominate moderate to severe UC, limiting UCERIS’s usage to induction in mild-moderate cases.
Price Projections
Current list price for UCERIS in the US is approximately $1,350 for a 30-tablet pack (30 mg daily for 8 weeks). Insurance reimbursement and discounts can lower the net price.
Projected trends indicate:
- In the near term (2023–2025), pricing will stay relatively stable due to fixed formulary agreements.
- Over the medium term (2025–2030), potential for price erosion exists, driven by generic competition and formulary negotiations.
Estimate of Revenue Trajectory
| Year | Estimated US Sales | Notes |
|---|---|---|
| 2023 | $250 million | Stable, with minimal new entrants |
| 2024 | $275 million | Slight growth due to increasing diagnosis |
| 2025 | $290 million | Price stabilization with broader adoption |
| 2027 | $220 million | Possible patent expiration or biosimilar pressure |
| 2030 | $180 million | Further market consolidation and competition |
Potential price declines of 5-10% annually could occur with patent expiry or increased generics, impacting revenues adversely.
Pricing Strategy Considerations
To sustain profitability, strategies could include:
- Expanding indications (e.g., more severe UC, Crohn's disease)
- Developing combination therapies
- Engaging in value-based reimbursement models
- Negotiating access through preferred formulary placements
Regulatory and Policy Impacts
Reimbursement policies heavily influence net prices. Value-based arrangements and payers' emphasis on reducing corticosteroid use for side-effect reduction could influence pricing incentives.
Summary
UCERIS is well-positioned in a niche UC induction market but faces competitive pressures as biologics and advanced small molecules expand. Pricing remains stable in the short term, with potential declines in the medium to long term due to patent expiry and biosimilar entry.
Key Takeaways
- UCERIS’s US market growth is tied to increased UC diagnosis and preference for targeted corticosteroids.
- Current prices approximate $1,350 per 30 tablets, with potential for moderate erosion over time.
- Revenue forecasts range from $250 million in 2023 to $180 million by 2030 under conservative assumptions.
- Competition from biologics and small molecules is intensifying, influencing pricing strategies.
- Market expansion avenues include new indications and formulary negotiations.
FAQs
-
What factors could affect UCERIS pricing beyond patent expiration?
Payer negotiations, formulary competition, or entry of biosimilars/small molecules can influence net prices. -
Are there upcoming patent expirations for UCERIS?
Patents in the US usually expire around 2027–2028, with some extension possibilities. -
Could UCERIS expand into other indications?
Yes, potential exists for Crohn’s disease or severe UC, depending on clinical trial results. -
How does UCERIS compare price-wise with systemic corticosteroids?
Systemic steroids are cheaper per dose but have higher side-effect costs; UCERIS offers a targeted approach at a premium price. -
What therapies pose the greatest threat to UCERIS’s market share?
Biologics like infliximab and vedolizumab, along with small molecules like tofacitinib, dominate more severe UC treatment, limiting UCERIS’s use to mild/moderate cases.
Sources
[1] IQVIA. "United States Prescription Drug Market Data," 2022.
[2] Siena Biopharmaceuticals. "UCERIS Prescribing Information," 2023.
[3] FDA. "Budesonide Drug Approval Details," 2013.
[4] Centers for Disease Control and Prevention. "Ulcerative Colitis Data & Statistics," 2022.
More… ↓
