Share This Page
Drug Price Trends for TYRVAYA
✉ Email this page to a colleague
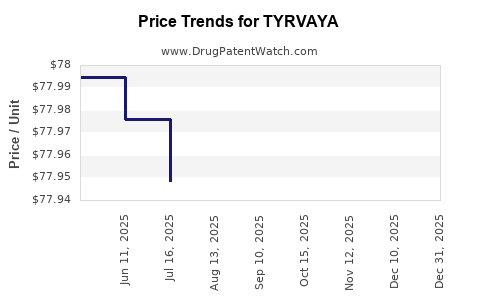
Average Pharmacy Cost for TYRVAYA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TYRVAYA 0.03 MG NASAL SPRAY | 73521-0030-02 | 79.64275 | ML | 2026-05-20 |
| TYRVAYA 0.03 MG NASAL SPRAY | 73521-0030-02 | 79.56294 | ML | 2026-01-01 |
| TYRVAYA 0.03 MG NASAL SPRAY | 73521-0030-02 | 78.00289 | ML | 2025-12-17 |
| TYRVAYA 0.03 MG NASAL SPRAY | 73521-0030-02 | 77.99421 | ML | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for TYRVAYA
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| TYRVAYA 0.03MG/SPRAY SOLN,NASAL | Oyster Point Pharma, Inc. | 73521-0030-02 | 2x4.2ML | 605.10 | 2023-05-01 - 2027-04-14 | FSS | ||
| TYRVAYA 0.03MG/SPRAY SOLN,NASAL | Oyster Point Pharma, Inc. | 73521-0030-02 | 2x4.2ML | 439.74 | 2024-01-01 - 2027-04-14 | Big4 | ||
| TYRVAYA 0.03MG/SPRAY SOLN,NASAL | Oyster Point Pharma, Inc. | 73521-0030-02 | 4.2ML | 420.72 | 100.17143 | ML | 2022-04-15 - 2027-04-14 | Big4 |
| TYRVAYA 0.03MG/SPRAY SOLN,NASAL | Oyster Point Pharma, Inc. | 73521-0030-02 | 2x4.2ML | 605.10 | 2024-01-01 - 2027-04-14 | FSS | ||
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
TYRVAYA Market Dynamics and Pricing Forecast
TYRVAYA (diclofenac ophthalmic solution) 0.1% is indicated for the treatment of the signs and symptoms of dry eye disease. This analysis examines its current market position, competitive landscape, and projects pricing trends based on patent status, regulatory approvals, and market penetration.
What is TYRVAYA's Current Market Position?
TYRVAYA is a non-steroidal anti-inflammatory drug (NSAID) in ophthalmic solution form, developed by Oyster Point Pharma. It is a prescription-only medication administered as a nasal spray. The mechanism of action involves stimulating natural tear production by activating the trigeminal parasympathetic pathway. This differentiates it from many existing dry eye treatments that focus on artificial tears or immunomodulation.
Key Product Attributes:
- Active Ingredient: Diclofenac ophthalmic solution 0.1%
- Indication: Signs and symptoms of dry eye disease
- Administration: Nasal spray
- Mechanism of Action: Stimulates natural tear production via trigeminal parasympathetic pathway activation
- Regulatory Status: Approved by the U.S. Food and Drug Administration (FDA) in October 2021. [1]
As of Q3 2023, TYRVAYA has achieved a specific market share within the broader dry eye disease therapeutics sector. While precise, up-to-the-minute market share data for niche prescription drugs is often proprietary, preliminary market uptake suggests a growing physician and patient adoption, driven by its novel administration route and mechanism. The target patient population for dry eye disease is substantial, estimated to affect millions of individuals globally.
Who are TYRVAYA's Primary Competitors?
The dry eye disease market is characterized by a diverse range of therapeutic classes and product offerings, including artificial tears, prescription anti-inflammatories, and lipid-based therapies. TYRVAYA competes with both established treatments and emerging therapies.
Major Competitive Classes and Examples:
- Artificial Tears (Lubricants):
- Systane (Alcon)
- Refresh (Allergan/AbbVie)
- TheraTears (Advanced Vision Research)
- Characteristics: Symptomatic relief, available over-the-counter (OTC) and prescription.
- Prescription Anti-inflammatories:
- Restasis (cyclosporine ophthalmic emulsion) (Allergan/AbbVie)
- Cequa (cyclosporine ophthalmic solution) (Sun Pharma)
- Xiidra (lifitegrast ophthalmic solution) (Takeda)
- Characteristics: Target underlying inflammation, require prescription.
- Lipid-Based Therapies:
- Systane Hydration PF (Alcon)
- Retaine MGD (Winsight)
- Characteristics: Address evaporative dry eye, often OTC.
- Other Emerging Therapies:
- Varenicline nasal spray (Bausch + Lomb - Varenicline is under development and has not yet received FDA approval for dry eye)
- Characteristics: Novel mechanisms of action, potentially addressing different pathways.
TYRVAYA's differentiation lies in its nasal delivery, stimulating endogenous tear production rather than directly lubricating or suppressing inflammation via topical means. This unique selling proposition addresses a critical unmet need for patients seeking to restore their natural tear film.
What is the Patent Landscape for TYRVAYA?
The patent landscape for TYRVAYA is crucial for determining its market exclusivity and long-term pricing power. Oyster Point Pharma has secured patents covering the composition of matter, methods of use, and manufacturing processes for TYRVAYA.
Key Patent Considerations:
- Composition of Matter Patents: These are generally the strongest, providing broad protection for the drug molecule itself.
- Method of Use Patents: These cover specific uses of the drug, such as its application for dry eye disease via nasal administration.
- Manufacturing Process Patents: These protect the methods used to produce the drug.
- Exclusivity Periods:
- FDA New Chemical Entity (NCE) Exclusivity: TYRVAYA received 5 years of NCE exclusivity upon its approval in October 2021. This period prevents the FDA from approving generic versions of the same drug based on a New Drug Application (NDA). [2]
- Patent Term: The duration of patent protection beyond NCE exclusivity is determined by the underlying patents. A comprehensive review of issued and pending patents is necessary for precise forecasting. While specific patent numbers and expiration dates are subject to ongoing legal and regulatory processes, typical patent lifecycles for novel drugs can extend for 20 years from the filing date, with potential extensions available.
Oyster Point Pharma has actively pursued patent protection, and the strength and longevity of these patents will directly influence the timeline for generic competition and, consequently, pricing erosion. The company's strategy involves building a robust patent portfolio to maximize market exclusivity.
What are the Pricing and Reimbursement Dynamics for TYRVAYA?
The pricing strategy for TYRVAYA is influenced by its innovative nature, clinical efficacy, and the competitive pricing of existing dry eye treatments. Reimbursement policies from payers (insurance companies, Medicare, Medicaid) are critical for patient access and commercial success.
Pricing Factors:
- Wholesale Acquisition Cost (WAC): This is the list price before any discounts or rebates. As of late 2023, the WAC for TYRVAYA was approximately $815 for a 30-day supply. [3]
- Net Price: The actual price received by the manufacturer after rebates and discounts is lower than the WAC.
- Value-Based Pricing: The drug's novel mechanism and potential for improved patient outcomes compared to existing treatments can support a premium pricing strategy.
- Competitor Pricing: TYRVAYA's price is benchmarked against other prescription dry eye treatments. For example, Xiidra (lifitegrast) has a WAC in a similar range, and Restasis (cyclosporine) also carries a significant price tag. OTC artificial tears are substantially less expensive but offer different therapeutic benefits.
- Reimbursement Landscape:
- Payer Coverage: Coverage varies by insurance plan. Many plans require prior authorization and step-therapy (patients must try less expensive alternatives first).
- Patient Co-pays: High WAC can translate to high co-pays for patients, potentially impacting adherence. Manufacturer co-pay assistance programs are often utilized to mitigate this.
- Market Access: Securing favorable formulary placement on insurance plans is a key driver of market penetration.
The initial pricing of TYRVAYA reflects its position as an innovative therapy. As market penetration increases and payer negotiations evolve, pricing adjustments and the impact of rebates will become more pronounced.
What are the Projected Market Growth and Future Outlook for TYRVAYA?
The projected market growth for TYRVAYA is contingent on several factors, including patient and physician adoption, the continued effectiveness of its patent protection, and the emergence of new competitive therapies.
Market Growth Drivers:
- Prevalence of Dry Eye Disease: The aging population and increasing prevalence of digital device usage contribute to a growing pool of patients with dry eye disease. Estimates suggest that over 16 million Americans are diagnosed with dry eye, with many more undiagnosed. [4]
- Unmet Needs: A significant portion of dry eye patients remain inadequately treated by current therapies. TYRVAYA's novel approach may capture patients dissatisfied with existing options.
- Physician Education and Awareness: Effective marketing and physician education about TYRVAYA's mechanism and benefits are crucial for driving prescription volume.
- Clinical Trial Data: Ongoing and future clinical studies demonstrating long-term efficacy and safety will reinforce its market position.
Potential Challenges:
- Generic Competition: Upon patent expiration or successful patent challenges, generic versions could significantly reduce market share and pricing.
- New Entrants: Development of novel dry eye therapies with superior efficacy or different mechanisms could challenge TYRVAYA's market position.
- Reimbursement Pressures: Increased scrutiny from payers on drug pricing and value may lead to tighter reimbursement policies.
Pricing Projections:
- Short-Term (1-3 years): Pricing is expected to remain relatively stable, with potential minor adjustments to WAC. Net pricing will be influenced by ongoing rebate strategies. Market access will be a key determinant of volume growth.
- Medium-Term (3-7 years): As TYRVAYA establishes a strong clinical track record and payer relationships, pricing power may persist. However, early signs of patent challenges or the emergence of direct competitors could initiate downward pricing pressure. If generics enter the market, significant price erosion will occur, with prices potentially dropping by 70-90% of the branded price.
- Long-Term (7+ years): Pricing will be largely dictated by the presence of generic competition. If patent protection remains robust, sustained pricing is possible, but unlikely without significant therapeutic advancements or market differentiation. The typical pattern for branded drugs facing generic entry involves a sharp decline in revenue and price.
Based on its current trajectory and the market dynamics of dry eye therapeutics, TYRVAYA is projected to capture a meaningful segment of the prescription dry eye market. However, the timing and impact of generic entry will be the most significant factor influencing its long-term pricing and revenue trajectory.
Key Takeaways
- TYRVAYA offers a novel nasal spray approach to treating dry eye disease by stimulating natural tear production, differentiating it from traditional therapies.
- Its FDA approval in October 2021 initiated a 5-year period of NCE exclusivity, with further protection dependent on its patent portfolio.
- The current WAC for TYRVAYA is approximately $815 per 30-day supply, positioning it in line with other premium prescription dry eye treatments.
- Market growth will be driven by the high prevalence of dry eye disease and the unmet needs of many patients.
- Significant pricing erosion is anticipated upon the loss of patent exclusivity and the introduction of generic competition, a standard trajectory for branded pharmaceuticals.
Frequently Asked Questions
-
When is TYRVAYA's NCE exclusivity expected to expire, and what are the implications for generic entry? TYRVAYA's NCE exclusivity is set to expire in October 2026. This date is a critical marker, as it generally allows the FDA to accept abbreviated new drug applications (ANDAs) for generic versions. However, the actual entry of generics can be delayed by existing patents that may be defended by the innovator company.
-
What is the typical price reduction observed for branded ophthalmic drugs once generic versions become available? Upon the introduction of generic competition, branded ophthalmic drugs typically experience a price reduction ranging from 70% to 90% within the first 12-24 months. This decline is driven by market competition and payer preference for lower-cost generics.
-
How does TYRVAYA's mechanism of action compare to other leading prescription dry eye treatments like Xiidra and Restasis? Xiidra (lifitegrast) is an oral medication that blocks the interaction of LFA-1 with its cognate ligand ICAM-1, thereby inhibiting T-cell mediated inflammation. Restasis (cyclosporine) is a calcineurin inhibitor that modulates T-cell activation, aiming to reduce ocular inflammation and increase tear production. TYRVAYA, conversely, stimulates natural tear production by activating the trigeminal parasympathetic pathway via nasal stimulation, representing a distinct therapeutic approach.
-
What is the average out-of-pocket cost for patients prescribed TYRVAYA after insurance coverage and co-pay assistance? Out-of-pocket costs for patients can vary significantly based on their insurance plan's formulary, deductible, and co-insurance. Without insurance, the cost is close to the WAC. With insurance, co-pays can range from $20 to over $100 per month. Oyster Point Pharma offers a co-pay assistance program that can reduce eligible commercially insured patients' out-of-pocket costs to as low as $0 per month.
-
Are there any known long-term side effects associated with the continuous nasal administration of diclofenac for dry eye treatment? The most common side effects reported in clinical trials for TYRVAYA include eye irritation, decreased visual acuity, taste disorders, and bronchitis. Specific long-term effects related to chronic nasal administration of diclofenac are still being evaluated as the drug gains wider usage. Post-market surveillance by regulatory bodies and ongoing clinical studies will provide further insights into the long-term safety profile.
Citations
[1] U.S. Food & Drug Administration. (2021, October 18). FDA approves TYRVAYA nasal spray for the signs and symptoms of dry eye disease. [Press release]. [2] U.S. Food & Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from https://www.accessdata.fda.gov/scripts/drugod/ [3] Oyster Point Pharma. (2023). TYRVAYA (diclofenac ophthalmic solution) 0.1% Prescribing Information. [4] National Eye Institute. (n.d.). Dry Eye. Retrieved from https://www.nei.nih.gov/learn-about-eyes/eye-health-myths-and-facts/dry-eye
More… ↓
