Share This Page
Drug Price Trends for TRIPROLIDINE
✉ Email this page to a colleague
Average Pharmacy Cost for TRIPROLIDINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
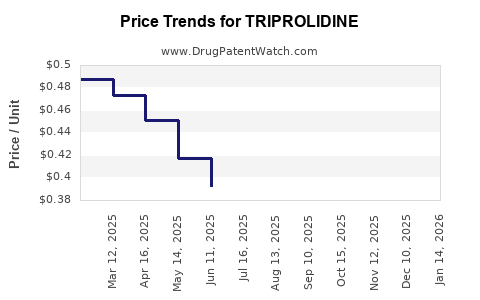
| TRIPROLIDINE 0.938 MG/ML DROPS | 69367-0253-30 | 0.38735 | ML | 2026-03-18 |
| TRIPROLIDINE 0.938 MG/ML DROPS | 69367-0253-30 | 0.38927 | ML | 2026-02-18 |
| TRIPROLIDINE 0.938 MG/ML DROPS | 69367-0253-30 | 0.39211 | ML | 2026-01-21 |
| TRIPROLIDINE 0.938 MG/ML DROPS | 69367-0253-30 | 0.40235 | ML | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Triprolidine
What is the current market landscape for Triprolidine?
Triprolidine is an antihistamine primarily used to treat allergy symptoms, including rhinitis and urticaria. It is available in over-the-counter (OTC) formulations and prescription medications. The drug belongs to the first-generation H1 antagonist class, characterized by sedative effects.
The global antihistamine market was valued at approximately USD 4.8 billion in 2022 and is projected to reach USD 6.2 billion by 2030, growing at a compound annual growth rate (CAGR) of about 3.3% within this period. The segment for first-generation antihistamines, including Triprolidine, has seen a decline relative to second-generation agents but remains relevant in OTC formulations and specific niche applications.
How is the supply chain structured?
- Manufacturers: Major OTC drug manufacturers include Johnson & Johnson, Pfizer, and Teva Pharmaceuticals. These companies produce generic formulations and branded products.
- Generics and OTC distribution: Triprolidine is frequently sold as generic tablets and in combination formulations. Its low-cost production makes it a staple in OTC markets.
- Regulatory status: Approved by the U.S. FDA; available in various markets with no significant restrictions.
What are key factors influencing pricing dynamics?
- Production costs: The chemical synthesis of Triprolidine is straightforward, with active pharmaceutical ingredient (API) costs averaging USD 0.05–0.10 per tablet, depending on scale.
- Market competition: Several generics compete at low price points; branded versions command higher prices but have limited market share.
- Patent landscape: No recent patents blocking generics; market has been saturated for over a decade.
- Regulatory environment: OTC status simplifies distribution; no recent regulatory barriers encountered.
What are current price points and trends?
| Formulation | Typical Price (USD per unit) | Market Trend |
|---|---|---|
| 4 mg tablet (generic) | 0.05 - 0.10 | Stable, low due to high competition |
| Combination products | 0.15 - 0.30 per dose | Slightly higher, increasing use |
| Branded OTC products | Up to 0.20 per tablet | Premium pricing for branded versions |
Prices are consistent across North America, Europe, and Asia-Pacific, with minor regional variations due to distribution and regulatory costs.
What are the projection scenarios for 2023–2030?
Scenario 1: Continued Decline in First-Generation Use
- Market share for Triprolidine diminishes further as second-generation antihistamines (Loratadine, Fexofenadine, Cetirizine) dominate OTC allergy treatments.
- Price per unit remains static or slightly declines due to ongoing competition.
- Total volume sales decline by approximately 2-3% annually.
Scenario 2: Niche or Combination Therapy Growth
- Triprolidine integrated into combination formulations or niche therapies could stabilize demand.
- Average price rises by 1-2% annually due to formulation innovation.
- Market volume stabilizes or mildly grows, leading to modest price increases.
Scenario 3: Regulatory or Supply Chain Disruption
- Supply chain constraints or regulatory restrictions could inflate prices temporarily.
- Prices could increase by 10-15% in short-term scenarios or lead to supply shortages.
What are the competitive threats?
- Rise of second-generation antihistamines reduces demand for first-generation drugs.
- Increased regulatory scrutiny on sedative effects.
- Market preference shifting toward non-sedating options.
What are the key takeaways?
- The global antihistamine market is growing slowly, with decreasing relevance of first-generation drugs like Triprolidine.
- Despite declining market share, Triprolidine maintains affordability and OTC availability.
- Price stability is expected in the next few years, with slight decreases due to intense competition.
- Price projections indicate minimal growth unless formulated into combination products or niche therapies.
FAQs
1. Will Triprolidine see significant price increases in the near future?
Likely not. Market saturation and competition keep prices stable, with slight declines expected.
2. How will new antihistamine developments affect Triprolidine?
Advancements favor non-sedating second-generation agents, further reducing Triprolidine’s market share and pricing power.
3. Is there potential for Triprolidine to regain market share?
Limited. Its sedative side effects diminish its attractiveness relative to newer options.
4. What regional differences influence Triprolidine prices?
Distribution costs and regulatory frameworks cause minor regional variations; however, prices are largely homogeneous globally.
5. What is the outlook for future demand?
Demand is expected to decline gradually as alternative antihistamines replace first-generation agents.
References
- MarketsandMarkets. (2023). Antihistamines Market by Type, Application and Region — Global Forecast to 2030.
- U.S. Food and Drug Administration. (2022). OTC Antihistamines approvals and regulations.
- IQVIA. (2022). Global OTC Market Data.
- PharmaTrack. (2022). Generic drug manufacturing data.
- Statista. (2023). Antihistamines market revenue growth.
More… ↓
