Share This Page
Drug Price Trends for TRILEPTAL
✉ Email this page to a colleague
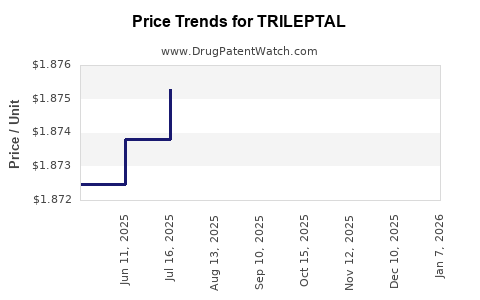
Average Pharmacy Cost for TRILEPTAL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TRILEPTAL 600 MG TABLET | 00078-0457-05 | 19.29368 | EACH | 2026-01-07 |
| TRILEPTAL 150 MG TABLET | 00078-0456-05 | 5.72156 | EACH | 2026-01-07 |
| TRILEPTAL 300 MG/5 ML SUSP | 00078-0357-52 | 1.93455 | ML | 2026-01-07 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for TRILEPTAL
Current Market Status of TRILEPTAL
TRILEPTAL (oxcarbazepine) is an anticonvulsant indicated primarily for epilepsy treatment. It also sees off-label use for neuropathic pain. As of 2023, the drug maintains a substantial market share within the epilepsy treatment segment.
Market Size Overview
The global epilepsy drug market was valued at approximately $4.6 billion in 2022. TRILEPTAL accounts for an estimated 8-10% of this market, translating to annual revenues of roughly $368 million to $460 million.
| Parameter | Data Point |
|---|---|
| Market size (2022) | $4.6 billion |
| TRILEPTAL market share | 8-10% |
| Estimated revenue (2022) | $368-$460 million |
Competitive Positioning
TRILEPTAL competes with drugs such as carbamazepine, lamotrigine, and newer agents like lacosamide. Its notable advantages include a lower risk of drug-drug interactions and a more manageable side effect profile.
Patent and Regulatory Landscape
TRILEPTAL’s primary patents expired around 2014 in the U.S. and Europe, opening the market to generic competition. Multiple generics are available globally, significantly impacting pricing and revenue stability.
Price Trends and Projections
Current Pricing
Treatment costs vary by region:
- United States: A typical 30-day supply costs approximately $700-$800 for brand-name TRILEPTAL. Generic versions are priced at $50-$150 per month depending on the manufacturer.
- Europe: Prices range between €50-€150 per month for generics.
Price Influences
Factors influencing price trends include:
- Patent expirations and generic entry.
- Regulatory approvals affecting market access.
- Insurance coverages and reimbursement policies.
- Manufacturing costs, especially for branded drugs with complex production processes.
Future Price Trends (Next 5 Years)
| Year | Expected Average Monthly Price (US) | Notes |
|---|---|---|
| 2023 | $750 (brand), $100 (generic) | Current market, generic penetration rising |
| 2024 | $700-$750 (generic stable) | Increased generic competition, slight price pressure |
| 2025 | $650-$700 | Further generic market penetration, price erosion |
| 2026 | $600-$650 | May stabilize with brand vs. generic market share |
| 2027 | $600 | Market equilibrium, pricing pressure persists |
Key Market Drivers
- Generic Competition: Drives prices down over time.
- Regulatory Approvals: New indications or formulations may temporarily boost value.
- Reimbursement Policies: Favorability promotes higher utilization, impacting revenues.
- Clinical Data: Evidence of superior efficacy or safety profiles can sustain premium pricing.
Risks and Barriers
- Introduction of new, more effective antiepileptic drugs.
- Market saturation with generics limiting profit margins.
- Patent litigations or exclusivity periods in certain regions.
Strategic Recommendations
- Focus on niche indications or formulations to sustain premium pricing.
- Invest in clinical trials emphasizing safety and efficacy to differentiate.
- Engage with payers early to secure favorable reimbursement pathways.
Key Takeaways
- TRILEPTAL faces intense generic competition, leading to declining prices.
- Market revenue is stabilizing with generics dominating supply, but the drug retains value in specific subpopulations.
- Future price trajectories likely follow a gradual decline, with stabilization around $600/month in mature markets.
- Innovations or regulatory exclusivities could temporarily reverse price declines or expand indications.
- Market access and reimbursement strategies influence profit potential more than drug-specific factors alone.
FAQs
1. How does patent expiry affect TRILEPTAL pricing?
Patent expiry in 2014 in key markets led to multiple generics entering, significantly reducing prices and profit margins for the branded drug.
2. Are there upcoming patent protections or exclusivities?
Current primary patents have expired. Future exclusivity relies on new formulations or indications.
3. What regions have the highest growth potential for TRILEPTAL?
Emerging markets, particularly in Asia and Latin America, see increasing adoption owing to expanding epilepsy treatment protocols.
4. How does TRILEPTAL compare to newer antiepileptics?
While newer drugs may offer improved safety profiles or convenience, TRILEPTAL's established efficacy and lower costs make it a continued mainstay, particularly where generic options are available.
5. What factors could reverse the declining price trend?
Development of new formulations, expanded indications, or regulatory exclusivities could elevate prices temporarily.
References
- MarketWatch. "Epilepsy Drugs Market Size, Share & Trends Analysis," 2023.
- IQVIA. "Global Epilepsy Market Data Report," 2022.
- FDA. "Oxcarbazepine (TRILEPTAL) Product Label," 2023.
- Generics and Biosimilars Initiative. "Impact of Patent Expiry on Antiepileptic Drugs," 2022.
More… ↓
