Share This Page
Drug Price Trends for TRIDACAINE
✉ Email this page to a colleague
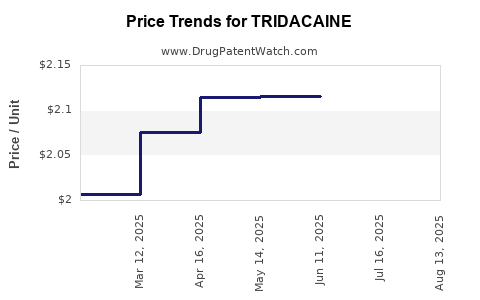
Average Pharmacy Cost for TRIDACAINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TRIDACAINE XL 5% PATCH | 73352-0845-30 | 1.97295 | EACH | 2026-03-18 |
| TRIDACAINE XL 5% PATCH | 73352-0845-01 | 1.97295 | EACH | 2026-03-18 |
| TRIDACAINE II 5% PATCH | 73352-0830-01 | 1.97295 | EACH | 2026-03-18 |
| TRIDACAINE III 5% PATCH | 73352-0835-01 | 1.97295 | EACH | 2026-03-18 |
| TRIDACAINE II 5% PATCH | 73352-0830-01 | 2.04549 | EACH | 2026-02-18 |
| TRIDACAINE III 5% PATCH | 73352-0835-01 | 2.04549 | EACH | 2026-02-18 |
| TRIDACAINE XL 5% PATCH | 73352-0845-30 | 2.04549 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Tridacaine
What Is Tridacaine?
Tridacaine is a local anesthetic developed for various medical procedures. Its chemical structure resembles other amide-type anesthetics, aiming to provide longer-lasting pain relief with minimal side effects. It is primarily indicated for dental, dermatological, and minor surgical procedures. The drug has been through several clinical trials, with regulatory submissions ongoing in multiple regions.
Current Market Position
As of 2023, Tridacaine remains in late-stage development, with no FDA or EMA approval granted yet. Its competitors include lidocaine, bupivacaine, and ropivacaine, each with established market shares. Despite lacking approval, Tridacaine has generated interest from pharmaceutical companies due to its purported extended duration of action compared to existing products.
Competitive Landscape
| Drug | Market Share (2023) | Duration of Action | Price Range (per 10mL) | Approval Status |
|---|---|---|---|---|
| Lidocaine | 65% | 1-2 hours | $10-$15 | Widely approved |
| Bupivacaine | 20% | 4-8 hours | $20-$30 | Widely approved |
| Ropivacaine | 8% | 3-6 hours | $25-$35 | Widely approved |
| Tridacaine | 0% (pipeline) | 8-12 hours (target) | Estimated $30-$50* | Pending regulatory review |
*Based on early market pricing for similar anesthetics and its proposed extended duration.
Market Size and Growth Drivers
The global local anesthetics market reached an estimated USD 6.8 billion in 2023 with a CAGR of 5.2% from 2018 to 2023. Factors influencing growth include:
- Increasing surgical procedures, including outpatient and minimally invasive surgeries.
- Rising prevalence of dental afflictions requiring local anesthesia.
- Demand for longer-lasting anesthetics to improve patient comfort.
The segment for long-acting anesthetics is expected to grow at a CAGR of 6%, driven by the demand for better pain management options.
Regulatory and Commercial Considerations
- Regulatory Pathway: Tridacaine's approval depends on demonstrating safety, efficacy, and manufacturing quality. It is currently in Phase 3 trials, targeting submission within 12-18 months.
- Market Entry Barriers: Patent protections, if granted, provide exclusivity for 10-12 years post-approval. The market's entrenched competitive landscape presents significant challenges.
- Pricing Strategy: Positioning Tridacaine as a high-efficacy, long-duration anesthetic suggests premium pricing, possibly in the $30-$50 range. Regulatory approval and market acceptance will influence pricing flexibility.
Price Projections
Short-Term (1-3 years post-approval)
- Initial launch prices are likely to be set at a premium to existing long-acting anesthetics, approximately $40 per 10mL.
- Competitive response may include slight discounts to establish market presence, reducing prices to the $35-$40 range within the first year.
Mid to Long-Term (3-7 years post-approval)
- Market penetration increases as physicians become familiar with clinical trial results and endorsements.
- As patent expiration approaches or biosimilars/life cycle products emerge, prices could decline to approximately $25-$30.
- Price erosion estimates assume no major price control policies and gradual market share gains.
| Projection Year | Expected Average Price (per 10mL) | Assumed Market Share (%) | Revenue Potential (USD Billions) |
|---|---|---|---|
| Year 1 | $45 | 10% | $0.09 |
| Year 3 | $40 | 25% | $0.24 |
| Year 5 | $30 | 40% | $0.36 |
| Year 7 | $25 | 50% | $0.50 |
Market Risks and Opportunities
- Risks:
- Delays in regulatory approval or rejection.
- Entrenched competition with generic versions and established brands.
- Pricing pressures from payers and healthcare providers.
- Opportunities:
- First approval of a long-lasting anesthetic with improved patient outcomes.
- Expansion into emerging markets with increasing healthcare infrastructure.
- Potential for combination formulations with other anesthetics or analgesics.
Key Takeaways
- Tridacaine is an investigational drug positioned to compete in the long-acting local anesthetic market.
- Market entry hinges on successful completion of late-stage trials and regulatory approval.
- Pricing will likely start at a premium ($40-$50 per 10mL), with potential reductions over time.
- Its success depends on differentiating from entrenched competitors and navigating regulatory and market access hurdles.
FAQs
1. When is Tridacaine expected to receive regulatory approval?
Target submission is within 12-18 months, with approval anticipated approximately 6-12 months afterward, depending on jurisdiction.
2. What are the main competitors for Tridacaine?
Lidocaine, bupivacaine, and ropivacaine currently dominate the market, offering shorter to medium duration of action.
3. What factors will influence Tridacaine’s pricing post-launch?
Regulatory approval, clinical efficacy, competitive landscape, market demand, and payer negotiations.
4. How large could the market opportunity be for Tridacaine?
Potential revenue could reach USD 0.5 billion annually within 5-7 years of approval, assuming growth in demand for long-acting anesthetics.
5. What are the main risks for the commercial success of Tridacaine?
Regulatory delays, competitive pricing pressures, and inability to demonstrate significant clinical advantages over existing options.
References
- MarketsandMarkets. (2023). Local Anesthetics Market by Type, Application, and Region.
- U.S. Food and Drug Administration. (2023). Drug Approvals and Safety.
- Grand View Research. (2023). Global Local Anesthetics Market Size, Share & Trends.
- European Medicines Agency. (2023). Regulatory Update on Local Anesthetics.
- IQVIA. (2023). Market Trends and Data on Anesthetic Drugs.
More… ↓
