Share This Page
Drug Price Trends for TRICITRATES ORAL SOLUTION
✉ Email this page to a colleague
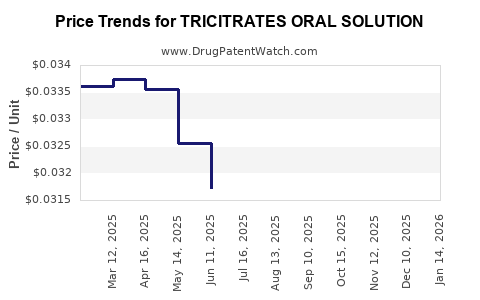
Average Pharmacy Cost for TRICITRATES ORAL SOLUTION
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TRICITRATES ORAL SOLUTION | 00121-0677-16 | 0.03462 | ML | 2026-03-18 |
| TRICITRATES ORAL SOLUTION | 62135-0438-47 | 0.03462 | ML | 2026-03-18 |
| TRICITRATES ORAL SOLUTION | 00121-0677-16 | 0.03321 | ML | 2026-02-18 |
| TRICITRATES ORAL SOLUTION | 62135-0438-47 | 0.03321 | ML | 2026-02-18 |
| TRICITRATES ORAL SOLUTION | 00121-0677-16 | 0.03163 | ML | 2026-01-21 |
| TRICITRATES ORAL SOLUTION | 62135-0438-47 | 0.03163 | ML | 2026-01-21 |
| TRICITRATES ORAL SOLUTION | 00121-0677-16 | 0.02978 | ML | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
TRICITRATES ORAL SOLUTION: MARKET ANALYSIS AND PRICE PROJECTIONS
Tricitrates Oral Solution is a pharmaceutical product subject to patent protections, market dynamics, and pricing fluctuations. This analysis projects market performance and price trends based on current data and anticipated factors.
What is the Current Market Landscape for Tricitrates Oral Solution?
The market for Tricitrates Oral Solution is influenced by its therapeutic indication, the prevalence of associated conditions, and the competitive landscape. While specific market size figures for Tricitrates Oral Solution are not publicly disseminated as a standalone entity, its active ingredients and the conditions they treat offer insight into its potential market positioning.
Tricitrates, typically a combination of citric acid and sodium citrate, is commonly used as an alkalinizing agent to treat metabolic acidosis and to prevent the formation of uric acid and cystine renal calculi. The market for such solutions is driven by the incidence of conditions like chronic kidney disease (CKD), certain types of cancer treatment side effects, and recurrent kidney stone formation.
-
Prevalence of Indications:
- Metabolic Acidosis: This condition is often associated with chronic kidney disease, diabetic ketoacidosis, and certain gastrointestinal disorders. The global prevalence of CKD is estimated to affect 10% of the world’s population, with higher rates in developed countries. [1]
- Renal Calculi: Uric acid and cystine stones constitute a significant portion of kidney stone diagnoses. Approximately 1 in 10 people will experience kidney stones in their lifetime, with recurrence rates as high as 50% within seven years. [2]
-
Competitive Environment: The market for alkalinizing agents and kidney stone prophylaxis includes both prescription medications and over-the-counter (OTC) alternatives. Generic formulations of sodium citrate and citric acid are available, competing with branded products. Furthermore, alternative treatment modalities for metabolic acidosis and kidney stone prevention exist, including other citrate salts, bicarbonate therapies, and dietary interventions.
-
Regulatory Status: Tricitrates Oral Solution is typically regulated as a prescription drug in most major markets. This requires physician oversight and prescription, influencing distribution channels and market access. The regulatory pathway for approval, including post-market surveillance, adds to the cost of bringing and maintaining such a product on the market.
What are the Key Patent Protections and Their Impact?
Patent protection is a critical determinant of market exclusivity and pricing power for pharmaceutical products. The intellectual property landscape for Tricitrates Oral Solution will dictate the period of market dominance for the innovator product and the subsequent entry of generic competitors.
-
Active Ingredient Patents: Patents covering the specific chemical entity or a combination of active ingredients (e.g., tricitrates) would provide broad protection. These patents typically have a lifespan of 20 years from the filing date. However, patent term extensions may be available in certain jurisdictions to compensate for regulatory review periods.
-
Formulation Patents: Patents may also be granted for novel formulations of the active ingredients, such as specific oral solutions with improved stability, palatability, or bioavailability. These can extend market exclusivity beyond the primary active ingredient patent.
-
Method of Use Patents: Patents can protect specific therapeutic uses of Tricitrates Oral Solution, such as its efficacy in treating particular types of metabolic acidosis or preventing specific kinds of kidney stones.
-
Patent Expiry Timeline: A detailed analysis of patent expiry dates for Tricitrates Oral Solution and its key components is crucial for projecting the timeline for generic entry. For example, if the primary patent for a branded Tricitrates Oral Solution expires in 2025, generic manufacturers can legally enter the market thereafter. This entry typically leads to significant price erosion.
-
Impact on Market Exclusivity:
- Pre-Expiry: During the patent-protected period, the innovator company typically enjoys market exclusivity, allowing for premium pricing.
- Post-Expiry: Upon patent expiry, the market opens to generic competition. This generally results in a rapid decline in the price of the drug, often by 50-80% within the first year of generic availability, as multiple manufacturers enter the market.
How Will Pricing Be Influenced in the Coming Years?
The pricing of Tricitrates Oral Solution will be shaped by a confluence of factors, including patent status, market competition, reimbursement policies, and healthcare cost containment efforts.
-
Pre-Generic Competition Phase (Current to Patent Expiry):
- Price Premiums: The innovator product will likely command premium pricing, reflecting R&D investment and market exclusivity.
- Therapeutic Value Pricing: Pricing will be benchmarked against the therapeutic value it provides compared to alternative treatments, considering efficacy, safety, and patient outcomes.
- Reimbursement Rates: Payer formularies and reimbursement rates set by government and private insurers will play a significant role in determining the effective selling price. Negotiated rebates and discounts will reduce the list price.
- Projected Price Range (Innovator): Based on similar prescription alkalinizing agents, the innovator product could range from $50 to $200 per month supply, depending on dosage, patient indication, and market.
-
Post-Generic Competition Phase (Post-Patent Expiry):
- Price Erosion: The introduction of generic versions will lead to substantial price decreases. The speed and magnitude of this erosion depend on the number of generic manufacturers entering the market and the pricing strategies they adopt.
- Generic Price Benchmarking: Generic prices will be driven down by competition, often settling at a fraction of the innovator price.
- Projected Price Range (Generic): Generic Tricitrates Oral Solution is expected to be priced significantly lower, potentially in the range of $10 to $50 per month supply, for multi-tablet or multi-dose formulations.
-
Factors Influencing Future Pricing:
- Cost of Goods Sold (COGS): Fluctuations in the cost of raw materials and manufacturing will impact pricing, particularly for generic versions.
- Healthcare Policy: Government policies on drug pricing, such as price caps or negotiation mandates, could exert downward pressure on prices.
- Market Demand: The sustained or growing demand for treatments of metabolic acidosis and kidney stone prophylaxis will support market volume, but aggressive pricing will be a prerequisite for market share.
- Therapeutic Advancements: Development of superior or more cost-effective alternative treatments could diminish the market share and pricing power of Tricitrates Oral Solution.
What is the Projected Market Growth Trajectory?
The market growth for Tricitrates Oral Solution will be influenced by the interplay of demographic trends, disease prevalence, and the competitive dynamics post-patent expiry.
-
Growth Drivers:
- Aging Population: The increasing global elderly population is a primary driver for conditions like CKD and associated metabolic acidosis.
- Rising Incidence of Chronic Diseases: The prevalence of diabetes and obesity, which are risk factors for CKD and kidney stones, continues to rise globally, fueling demand for related treatments.
- Increased Diagnosis Rates: Improved diagnostic capabilities and greater awareness of metabolic disorders can lead to higher identification and treatment rates.
- Geographic Expansion: Entry into emerging markets with growing healthcare access and infrastructure can expand the patient pool.
-
Growth Restraints:
- Generic Competition: While generic entry expands access and lowers costs, it significantly reduces the overall revenue value of the market for the innovator.
- Cost Containment Pressures: Healthcare systems worldwide are under pressure to control pharmaceutical spending, which can limit price increases and favor more cost-effective treatments.
- Availability of Alternative Therapies: Development and adoption of novel or more effective treatments for the indications of Tricitrates Oral Solution could cannibalize its market share.
-
Projected Market Value:
- Pre-Expiry (e.g., 2024-2027): The market value for the branded product is expected to remain relatively stable or experience modest growth, driven by volume and price, but constrained by the looming patent cliff. If the global market for similar alkalinizing agents is estimated at $500 million to $1 billion annually, Tricitrates Oral Solution could capture a significant portion of this.
- Post-Expiry (e.g., 2028 onwards): The total market value (including generics) is likely to grow in volume but see a sharp decline in overall revenue in the initial years post-expiry due to price competition. Over the longer term, as healthcare systems adopt the lower-cost generics and patient access expands, the volume of treatments will increase, potentially leading to a recovery in total market value, albeit at lower price points. The market could stabilize with an annual value of $300 million to $700 million within 3-5 years post-generic entry, driven by increased patient utilization.
-
Specific Segment Growth: The market for kidney stone prophylaxis may see higher growth rates due to its prevalence and the clear benefit of preventing recurrent, painful episodes.
What are the Key Risks and Opportunities?
Navigating the pharmaceutical market for Tricitrates Oral Solution involves understanding both the potential pitfalls and avenues for growth.
-
Key Risks:
- Intensified Generic Competition: A large number of generic manufacturers entering the market simultaneously can lead to rapid and severe price erosion, impacting profitability for all players.
- Regulatory Scrutiny: Post-market surveillance and potential adverse event reports could lead to labeling changes, warnings, or even product withdrawal, impacting market access and demand.
- Reimbursement Challenges: Changes in payer policies or adverse formulary decisions can restrict patient access and physician prescribing habits.
- Therapeutic Obsolescence: The development of breakthrough therapies that offer superior efficacy or a more convenient administration route could render Tricitrates Oral Solution less competitive.
- Supply Chain Disruptions: Geopolitical events, raw material shortages, or manufacturing issues can impact product availability and pricing.
-
Key Opportunities:
- Emerging Market Expansion: Untapped patient populations in developing economies represent a significant growth opportunity, provided market access strategies are tailored to local healthcare systems and pricing sensitivities.
- Combination Therapies: Exploring potential synergistic effects with other drugs for specific indications could lead to new treatment protocols and patentable claims.
- Lifecycle Management: Developing new formulations (e.g., extended-release, improved taste profiles) or exploring new indications for Tricitrates Oral Solution could extend market exclusivity and value.
- Strategic Partnerships: Collaborating with other pharmaceutical companies for co-promotion, distribution, or R&D can leverage existing infrastructure and market reach.
- Value-Added Services: Offering patient support programs, educational resources, or adherence tools can enhance brand loyalty and market penetration.
Key Takeaways
The market for Tricitrates Oral Solution is characterized by its dependence on patent protection, the prevalence of metabolic acidosis and renal calculi, and a competitive landscape with established generic alternatives. Post-patent expiry, significant price erosion is expected, shifting the market dynamic towards volume-driven sales and cost-effectiveness. Growth will be driven by demographic trends and increasing disease prevalence, offset by healthcare cost containment and therapeutic advancements.
Frequently Asked Questions
-
When is the patent for the primary Tricitrates Oral Solution formulation expected to expire, and what is the projected impact on pricing? The exact patent expiry date is dependent on specific patent filings for branded products. However, upon expiry, a typical price erosion of 50-80% is projected within the first year as generic competition intensifies.
-
What are the primary indications driving demand for Tricitrates Oral Solution, and how prevalent are these conditions globally? The primary indications are metabolic acidosis, often linked to Chronic Kidney Disease (CKD) affecting 10% of the global population, and the prevention of uric acid and cystine renal calculi, experienced by approximately 1 in 10 individuals annually.
-
What is the projected annual market value for Tricitrates Oral Solution both before and after generic entry? Before generic entry, the market for similar alkalinizing agents is estimated between $500 million to $1 billion annually. Post-generic entry, the total market value is projected to stabilize around $300 million to $700 million annually, driven by increased patient utilization at lower price points.
-
Are there any significant regulatory hurdles or policy changes that could impact the future pricing or market access of Tricitrates Oral Solution? Yes, ongoing regulatory scrutiny for drug safety and efficacy, coupled with government-led price control initiatives and payer reimbursement policy changes, could significantly impact market access and pricing.
-
What are the key growth opportunities and risks for manufacturers of Tricitrates Oral Solution in the next five to ten years? Opportunities include expansion into emerging markets and lifecycle management through new formulations. Key risks involve intensified generic competition and the emergence of superior therapeutic alternatives.
Citations
[1] Hill, N. R., St. George, S. V., White, S. L., & Johnson, D. W. (2017). Global prevalence of chronic kidney disease: a systematic review and meta-analysis. Nephrology, 22(9), 695-700.
[2] Scales, C. D., Smith, A. C., Hanley, M. E., Barger, N. D., Butler, R. C., & Albala, D. M. (2012). Prevalence of kidney stones in the United States. European Urology, 62(2), 160-165.
More… ↓
