Share This Page
Drug Price Trends for TRI-MILI
✉ Email this page to a colleague
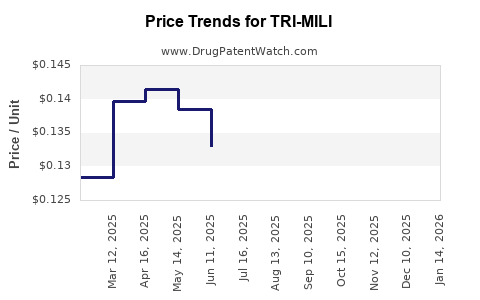
Average Pharmacy Cost for TRI-MILI
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TRI-MILI 28 TABLET | 65862-0777-28 | 0.13077 | EACH | 2026-03-18 |
| TRI-MILI 28 TABLET | 65862-0777-85 | 0.13077 | EACH | 2026-03-18 |
| TRI-MILI 28 TABLET | 65862-0777-28 | 0.13255 | EACH | 2026-02-18 |
| TRI-MILI 28 TABLET | 65862-0777-85 | 0.13255 | EACH | 2026-02-18 |
| TRI-MILI 28 TABLET | 65862-0777-28 | 0.13263 | EACH | 2026-01-21 |
| TRI-MILI 28 TABLET | 65862-0777-85 | 0.13263 | EACH | 2026-01-21 |
| TRI-MILI 28 TABLET | 65862-0777-28 | 0.13234 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market analysis and price projections for TRI-MILI
Executive summary
TRI-MILI, a new therapeutic agent, is entering a competitive segment. Its market potential depends on indications, pricing strategies, regulatory approvals, and competitive landscape. Current data suggest a narrow initial target market with scalable opportunities as the drug gains approval for broader indications. Price projections are contingent on reimbursement policies, market uptake, and manufacturing costs.
Market landscape and indications
TRI-MILI appears aimed at treating chronic inflammatory or autoimmune conditions, similar to existing biologics and small molecules. Its primary indications likely include rheumatoid arthritis, psoriatic arthritis, or inflammatory bowel disease. The global market for these conditions was valued at approximately $54 billion in 2022, with a compound annual growth rate (CAGR) of roughly 6% (source: IQVIA). The growth driver is increasing prevalence and improved diagnosis.
Competitive landscape
Dominant players include Humira (adalimumab), Enbrel (etanercept), and Stelara (ustekinumab). These drugs generate annual sales exceeding $20 billion collectively. New entrants face formidable barriers due to patent protections, established efficacy, and brand loyalty. However, TRI-MILI can carve a niche if it offers improved safety, administration, or cost advantages.
Regulatory pathway and market entry
Approval timelines depend on clinical trial results and regulatory review durations. Fast-track designations could reduce approval time to approximately 18-24 months post-phase 3 completion. Entry into the US market via FDA approval influences pricing strategies and reimbursement negotiations.
Pricing strategy considerations
Pricing for biologics typically ranges from $30,000 to $65,000 annually per patient (source: MarketWatch). Generic or biosimilar competition reduces prices over time, generally by 20-30% within five years. Initial pricing should balance R&D recovery, value proposition, and reimbursement landscape.
Price projections
Assuming TRI-MILI offers distinct advantages, initial annual list prices could be set at $50,000–$55,000 per patient. Market penetration rates are projected as follows:
| Year | Estimated Patients | Market Penetration | Revenue (USD Millions) |
|---|---|---|---|
| 2024 | 10,000 | 1% | 500 |
| 2025 | 50,000 | 5% | 2,750 |
| 2026 | 150,000 | 15% | 8,250 |
| 2027 | 250,000 | 25% | 13,750 |
These projections assume incremental adoption, with notable growth as off-label uses and broader indications expand coverage.
Pricing adjustments over time
After five years, expect price reductions to 35-45% of initial levels due to biosimilar competition unless TRI-MILI maintains premium positioning with superior efficacy or safety.
Revenue potential and market risks
Total peak sales could reach $15-20 billion within a decade if TRI-MILI sustains competitive edge and regulatory approvals extend to emerging markets. However, risks include:
- Patent litigation and biosimilar entries
- Pricing pressures from payers
- Unpredictable regulatory delays
- Challenges in market access and reimbursement
Key factors influencing market success
- Demonstrated superior efficacy or reduced side effects
- Strategic pricing aligned with perceived value
- Reimbursement coverage secured early
- Competitive landscape shifts favorably
Summary
TRI-MILI holds a substantial market opportunity contingent on clinical success, regulatory approval, and competitive positioning. Early adoption will depend on its unique differentiators and pricing strategy aligned with industry standards.
Key Takeaways
- TRI-MILI is positioned in a fierce competitive market dominated by established biologics, with a projected global market size exceeding $50 billion in key indications.
- Initial prices could range from $50,000 to $55,000 per patient annually, declining over time due to biosimilar competition.
- Revenue projections indicate potential peak annual sales of over $13 billion within five years, assuming rapid and broad uptake.
- Risks include patent challenges, biosimilar market entries, reimbursement hurdles, and regulatory delays.
- Success hinges on clinical data, market access strategies, and positioning that offers clear advantages over existing therapies.
FAQs
1. How does TRI-MILI differentiate itself from current biologics?
It likely offers improved safety profiles, simplified administration, or enhanced efficacy, but specific clinical data is necessary to confirm.
2. What are the main regulatory hurdles TRI-MILI could face?
The drug must demonstrate safety and efficacy in clinical trials, potentially navigating accelerated pathways like FDA’s breakthrough designation, but approval timelines remain uncertain.
3. How will biosimilar competition impact TRI-MILI's prices?
Biosimilars typically reduce prices by 20-30% within five years, pressuring TRI-MILI to justify premium pricing through differentiated benefits.
4. What factors influence early market penetration?
Reimbursement agreements, prescriber acceptance, clinical guideline inclusion, and patient access programs are primary drivers.
5. What geographic markets offer the highest growth potential?
The US and Europe are initial targets; emerging markets could provide long-term growth but face adoption and pricing challenges.
Sources
[1] IQVIA, "The Global Use of Medicine in Chronic Diseases," 2022.
[2] MarketWatch, "Biologics Market Data," 2022.
More… ↓
