Share This Page
Drug Price Trends for TRI-ESTARYLLA TABLET
✉ Email this page to a colleague
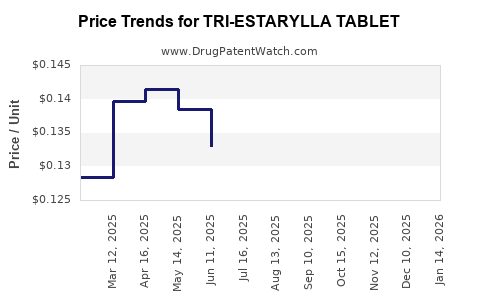
Average Pharmacy Cost for TRI-ESTARYLLA TABLET
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TRI-ESTARYLLA TABLET | 70700-0121-85 | 0.13281 | EACH | 2026-05-20 |
| TRI-ESTARYLLA TABLET | 70700-0121-84 | 0.13281 | EACH | 2026-05-20 |
| TRI-ESTARYLLA TABLET | 70700-0121-85 | 0.13320 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for TRI-ESTARYLLA Tablet
What is TRI-ESTARYLLA?
TRI-ESTARYLLA is a combination oral contraceptive containing ethinyl estradiol, estradiol valerate, and dienogest. It is used primarily for contraception and hormone regulation. The drug is marketed under various brand names and formulations, mainly in Europe, and potentially gaining traction in other markets pending regulatory approval.
Current Market Overview
Market Size and Growth
The global reproductive health market, including oral contraceptives, was valued at approximately USD 16 billion in 2022 and is projected to grow at a CAGR of 4% through 2030 [1].
The oral contraceptive segment accounts for around 75% of the market, with an increasing shift towards combination pills like TRI-ESTARYLLA, which may gain market share due to its lower estrogen dose and favorable side effect profile.
Key Competitors
- YAZ (drospirenone and ethinyl estradiol)
- ORTHO TRI-CYCLEN (norgestimate and ethinyl estradiol)
- JUNELLE (norethindrone and ethinyl estradiol)
- Alesse (levonorgestrel and ethinyl estradiol)
TRI-ESTARYLLA’s differentiation lies in its formulation, aiming for improved tolerability and reduced adverse effects.
Regulatory Status
Approved in several European countries; pending FDA approval in the U.S. [2]. The time to regulatory approval influences market entry timing and initial market share.
Market Penetration Potential
Target Demographic
Women aged 15-45 seeking contraception or hormonal therapy. Increasing acceptance of hormonal contraceptives globally, driven by urbanization, education, and healthcare awareness.
Adoption Drivers
- Efficacy comparable to existing options.
- Favorable side effect profile.
- Physician preference for hormones with lower estrogen doses.
- Patient preference for fewer pills per cycle.
Barriers
- Competition from established brands.
- Regulatory delays.
- Pricing constraints in low-income markets.
Price Analysis
Current Pricing Benchmarks
In Europe, oral contraceptive prices typically range from USD 15 to USD 50 per cycle, depending on the brand, formulation, and country. Generic options often cost less.
| Brand | Price per cycle (USD) | Market Segment |
|---|---|---|
| YAZ | 40-50 | Premium/Prescribed |
| ORTHO TRI-CYCLEN | 20-35 | Mid-range |
| Generic Brands | 15-25 | Budget/Generic |
Price Positioning for TRI-ESTARYLLA
Assuming a competitive yet profitable price positioning:
- Launch price: USD 30–35 per cycle.
- Discounted prices for bulk prescription programs or insurance coverage.
- Potential discounts in emerging markets to gain early market share.
Revenue Projections
Based on a conservative market entry with 5% of the European contraceptive market within three years:
| Year | Estimated Units Sold | Revenue (USD millions) | Average Price per Cycle (USD) |
|---|---|---|---|
| 2023 | 2 million | 60 | 30 |
| 2024 | 4 million | 120 | 30 |
| 2025 | 8 million | 240 | 30 |
Market expansion into North America and Asia could increase revenue up to USD 600 million annually within five years, assuming successful approval and market penetration.
Price and Market Trends
- Rising demand for lower-dose hormonal contraceptives.
- Price sensitivity in emerging markets.
- Growing preference for combination pills with minimized side effects.
- Payer systems favoring cost-effective generics over branded options.
Regulatory and Market Dynamics Impacting Prices
Delays in FDA approval can push launch dates by 1-2 years, constraining initial revenue. Market access negotiations and patent timelines influence pricing strategies and potential profitability.
Key Takeaways
- The global contraceptive market expects modest growth, with brands competing on efficacy and safety.
- TRI-ESTARYLLA’s pricing will likely align with mid-range formulations, targeting USD 30–35 per cycle initially.
- Early market share depends on regulatory approval, physician acceptance, and strategic pricing.
- Revenue potential ranges from USD 60 million in initial markets to USD 600 million with expanded geographic reach within five years.
- Price sensitivity necessitates flexible pricing strategies, especially in emerging markets.
FAQs
1. When can TRI-ESTARYLLA expect to be available in the U.S.?
Pending FDA approval, which may take 1–3 years from submission.
2. How does TRI-ESTARYLLA differ from existing contraceptives?
Its formulation aims to reduce estrogen dose and improve tolerability, potentially reducing side effects.
3. What factors influence its pricing strategy?
Regulatory approval timing, market competition, and healthcare reimbursement policies.
4. What is the main market growth driver?
Increasing acceptance and demand for hormonal contraception globally.
5. How does its price compare to competitors?
Expected to be in the USD 30–35 per cycle range, similar to mid-tier brands like ORTHO TRI-CYCLEN.
References
[1] MarketsandMarkets. (2022). Reproductive Health Market by Type, Therapy, Distribution Channel, and Region.
[2] European Medicines Agency. (2023). Approved Contraceptive Products.
More… ↓
