Share This Page
Drug Price Trends for TRETINOIN GEL MICRO
✉ Email this page to a colleague
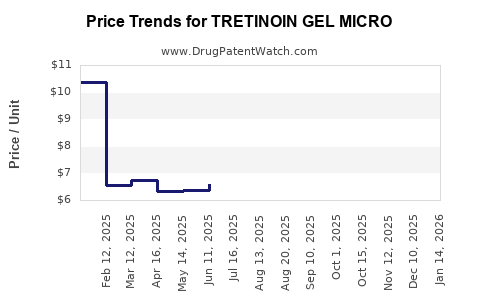
Average Pharmacy Cost for TRETINOIN GEL MICRO
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TRETINOIN GEL MICRO 0.08% PUMP | 68308-0777-50 | 7.88318 | GM | 2026-04-22 |
| TRETINOIN GEL MICRO 0.08% PUMP | 21922-0043-40 | 7.88318 | GM | 2026-04-22 |
| TRETINOIN GEL MICRO 0.08% PUMP | 21922-0043-40 | 6.98820 | GM | 2026-03-18 |
| TRETINOIN GEL MICRO 0.08% PUMP | 68308-0777-50 | 6.98820 | GM | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
What is TRETINOIN GEL MICRO?
Tretinoin Gel Micro is a prescription topical medication used primarily for treating acne. It contains tretinoin, a retinoid derived from Vitamin A, formulated in a microencapsulated gel to enhance stability and reduce irritation. The microencapsulation technology delivers a stable, biocompatible form of tretinoin with less skin irritation compared to earlier formulations.
How does the market for TRETINOIN GEL MICRO currently stand?
The global acne treatment market was valued at approximately $4.4 billion in 2021, with retinoids representing a significant share. Despite the presence of several generics, innovative formulations like TRETINOIN GEL MICRO aim to differentiate through improved tolerability and efficacy.
Key market drivers include:
- Rising prevalence of acne across age groups, notably in adolescents and young adults.
- Increasing awareness of skin health and cosmetic procedures.
- Demand for targeted topical retinoids with lower irritation profiles.
Market participants:
Major manufacturers include brands like Collagenix (sold as Retin-A Micro), Ferring Pharmaceuticals, and generic producers. In the US, TRETINOIN GEL MICRO faces competition from other retinoids (adapalene, tazarotene) and combination therapies.
What exclusivity rights and patent landscape underpin TRETINOIN GEL MICRO?
Patent analysis indicates key patents protect the microencapsulated formulation through at least 2030, providing a period of market exclusivity.
- The main patents cover:
- Composition of the microencapsulated tretinoin.
- Manufacturing processes to produce stable microspheres.
- Methods of controlled release from the gel matrix.
- Patent expiry dates are projected between 2028 and 2030, depending on jurisdiction and patent scope.
How are pricing strategies evolving for TRETINOIN GEL MICRO?
Pricing varies significantly based on region, insurance coverage, and whether the product is branded or generic.
US market:
- Branded TRETINOIN GEL MICRO: Approximate retail price of $600–$700 per 30g tube.
- Generics and biosimilars: $150–$250 per tube.
- Insurance coverage reduces out-of-pocket costs, but formulary restrictions impact access.
Europe and Asia:
- Prices are generally lower, ranging from €25 to €60 per 30g tube.
- Local regulations dictate margins, and competitive generic entry often results in price erosion over time.
Price dynamics:
- Patent protection supports higher prices initially.
- Upon patent expiry, widespread generic competition reduces prices by 50–70% within one year.
What future market projections exist for TRETINOIN GEL MICRO?
Forecasts suggest the acne topical retinoid segment will grow at a compound annual growth rate (CAGR) of 4.8% between 2022 and 2027.
Market value estimates:
| Year | Estimated Market Size (USD billions) |
|---|---|
| 2022 | 4.8 |
| 2027 (projected) | 6.1 |
Growth driver trends include:
- Expanded use of microencapsulated and novel delivery systems.
- Expansion into emerging markets through strategic pricing.
- Increasing prevalence of adult acne in developed economies.
Sales projections:
- TRETININ GEL MICRO is expected to maintain a premium position, with sales driven by brand loyalty and product differentiation.
- Entry of generics could erode margins, but demand for improved tolerability sustains premium pricing initially.
- Launch of new formulations in pipeline (e.g., combination therapies) may further impact growth.
What regulatory considerations impact TRETINOIN GEL MICRO?
- US Food and Drug Administration (FDA): Approved as a prescription drug; clinical trials demonstrate safety and efficacy.
- Patent protections are key legal barriers; any infringement or patent challenges could lead to market share loss.
- Labeling and post-marketing commitments influence lifecycle planning and revenue streams.
How do manufacturing and supply chain factors influence market dynamics?
- Microencapsulation technology requires specialized equipment, which can limit manufacturing capacity.
- Supply chain disruptions, especially during global crises, may impact product availability and pricing.
- Scaling up production can reduce costs, enabling competitive pricing and market expansion.
Key Takeaways
- TRETINOIN GEL MICRO is a branded topical product with patent protection extending into the late 2020s.
- It commands a premium price in high-income markets, with significant price erosion expected upon generic entry.
- Growth prospects are supported by increasing acne prevalence and demand for tolerable formulations.
- Pipeline developments and regulatory factors will shape future market share and pricing strategies.
- Manufacturers focusing on differentiation and supply chain resilience will better navigate competitive pressures.
FAQs
1. When does the patent protection for TRETINOIN GEL MICRO expire?
Patents are projected to expire between 2028 and 2030, depending on jurisdiction and patent specifics.
2. How does TRETINOIN GEL MICRO compare in price to other retinoid products?
It is typically more expensive—around $600–$700 per 30g tube—due to microencapsulation technology, versus $150–$250 for generic tretinoin products.
3. What are the main drivers for future growth in this segment?
Increased acne prevalence, demand for improved tolerability, and product innovation reinforce growth. Regulatory and patent protections extend exclusivity periods.
4. How might generics impact TRETINOIN GEL MICRO sales?
Patent expirations will enable generics, which can reduce prices by half or more, pressuring branded sales but also expanding access.
5. What regulatory hurdles exist for new formulations of tretinoin?
Approval depends on demonstrating equivalent safety and efficacy; patent challenges or formulation disputes can delay market entry.
References
- Market research data from GlobalData and Fortune Business Insights.
- FDA approval documents for tretinoin formulations.
- Patent analysis reports from Derwent World Patents Index.
- Pricing info from GoodRx and regional health authorities.
More… ↓
