Share This Page
Drug Price Trends for TRESIBA
✉ Email this page to a colleague
Average Pharmacy Cost for TRESIBA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TRESIBA 100 UNIT/ML VIAL | 00169-2662-11 | 9.02498 | ML | 2026-01-01 |
| TRESIBA FLEXTOUCH 200 UNIT/ML | 00169-2550-13 | 18.08941 | ML | 2026-01-01 |
| TRESIBA FLEXTOUCH 100 UNIT/ML | 00169-2660-15 | 9.04582 | ML | 2026-01-01 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
TresiBA: Market Landscape and Price Forecast
TresiBA (insulin degludec) is a long-acting basal insulin developed by Novo Nordisk for the management of diabetes mellitus. Its unique pharmacokinetic profile, characterized by an ultra-long duration of action and minimal day-to-day variability, positions it as a key player in the basal insulin market. This analysis examines TresiBA's current market position, competitive landscape, and projects its future price trajectory.
What is the Current Market Share and Sales Performance of TresiBA?
TresiBA has secured a significant presence in the global basal insulin market. In 2022, Novo Nordisk reported DKK 15.3 billion (approximately $2.2 billion USD) in global sales for TresiBA. This represents a 14% increase in local currencies compared to 2021 sales. The drug's performance is driven by its clinical advantages, including a reduced risk of nocturnal hypoglycemia and greater dosing flexibility compared to older insulin analogs.
TresiBA's market penetration varies by region. In North America, it has demonstrated robust growth, fueled by favorable formulary access and physician adoption. In Europe, TresiBA faces competition from established basal insulins but continues to gain traction due to its distinct clinical profile. The Asia-Pacific region presents a substantial growth opportunity, with increasing diabetes prevalence and a growing demand for advanced therapeutic options.
Key growth drivers for TresiBA include:
- Clinical Efficacy: Demonstrated ability to achieve glycemic control with a lower risk of hypoglycemia.
- Patient Convenience: Ultra-long duration allows for flexible dosing times without compromising efficacy.
- Expanding Indications: Approved for both Type 1 and Type 2 diabetes, broadening its addressable market.
- Market Penetration: Continuous efforts by Novo Nordisk to secure broad reimbursement and physician prescribing habits.
Who are TresiBA's Primary Competitors?
The basal insulin market is highly competitive, with several established and emerging products vying for market share. TresiBA competes primarily with other long-acting basal insulins.
Major Competitors Include:
- Lantus (insulin glargine): Developed by Sanofi, Lantus was a market leader for many years. While facing biosimilar competition, it remains a significant competitor.
- Levemir (insulin detemir): Also developed by Novo Nordisk, Levemir is an older long-acting insulin but still holds a market share, particularly in regions where TresiBA has not fully penetrated.
- Basaglar (insulin glargine): Eli Lilly's biosimilar to Lantus, Basaglar offers a cost-effective alternative and competes directly with branded glargine products.
- Toujeo (insulin glargine): Sanofi's proprietary, high-concentration formulation of insulin glargine, offering potential benefits in terms of reduced injection volume.
- Humalog (insulin lispro) and Novolog (insulin aspart) Mealtime Insulins (Indirect Competition): While not basal insulins, these rapid-acting insulins are used in combination regimens with basal insulins. TresiBA's efficacy in controlling fasting glucose can impact the overall regimen requirements for these rapid-acting insulins.
The competitive landscape is dynamic, with ongoing development of new insulin formulations and biosimil introductions. TresiBA's unique pharmacokinetic profile, particularly its duration of action and variability, is its primary differentiator.
What is the Patent Protection Status for TresiBA?
Novo Nordisk holds robust patent protection for TresiBA, safeguarding its market exclusivity. The primary patent covering the insulin degludec molecule itself provides significant protection. Additional patents likely cover manufacturing processes, formulations, and specific delivery devices.
Key Patent Considerations:
- Core Molecule Patents: These patents protect the active pharmaceutical ingredient, insulin degludec. Expiration dates for these foundational patents are critical for anticipating biosimilar entry.
- Formulation Patents: Patents may exist for specific compositions of TresiBA that enhance its stability, delivery, or efficacy.
- Delivery Device Patents: Novo Nordisk utilizes proprietary pen devices for TresiBA delivery. Patents on these devices can extend market exclusivity by making it difficult for competitors to replicate the full patient experience.
- Method of Use Patents: These patents can protect specific therapeutic applications or dosing regimens of TresiBA.
The expiration of key patents is a critical factor that will allow for the introduction of biosimilar insulin degludec products. While exact patent expiration dates are proprietary and complex, typical patent protection for novel drugs can extend for approximately 20 years from the filing date, subject to extensions. Industry observers anticipate potential biosimilar competition for TresiBA in the late 2020s or early 2030s, contingent on patent challenges and regulatory approvals.
What are the Key Regulatory Approvals and Market Authorizations for TresiBA?
TresiBA has received widespread regulatory approvals across major global markets, enabling its broad commercialization.
Key Approvals:
- United States: Approved by the Food and Drug Administration (FDA) in September 2015.
- European Union: Approved by the European Medicines Agency (EMA) in September 2013.
- Japan: Approved by the Ministry of Health, Labour and Welfare (MHLW) in March 2012.
- China: Approved by the National Medical Products Administration (NMPA) in 2017.
- Canada: Approved by Health Canada in 2016.
These approvals cover the use of TresiBA in adults with Type 1 and Type 2 diabetes mellitus. The drug is available in various concentrations and delivery devices to cater to diverse patient needs. Novo Nordisk continues to pursue regulatory approvals in emerging markets to expand TresiBA's global reach.
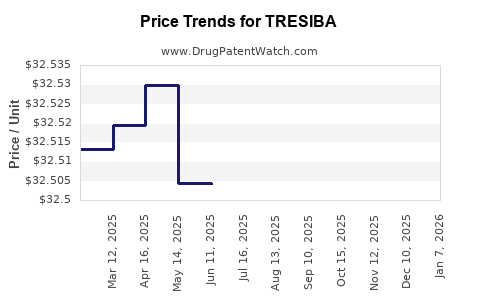
What is the Current Pricing Structure for TresiBA and What Factors Influence It?
The pricing of TresiBA, like most branded pharmaceuticals, is influenced by several factors, including its clinical value, R&D investment, manufacturing costs, market competition, and reimbursement policies. TresiBA is positioned as a premium basal insulin, reflecting its advanced formulation and therapeutic benefits.
Factors Influencing TresiBA Pricing:
- Therapeutic Value: The demonstrated reduction in hypoglycemia and improved glycemic control contribute to a higher perceived value, justifying a premium price.
- R&D and Manufacturing Costs: The significant investment in developing and manufacturing novel insulin analogs impacts pricing strategies.
- Market Competition: The presence of other long-acting insulins, including biosimil glargine, exerts downward pressure on prices.
- Reimbursement Policies: Payer negotiations, formulary placement, and prior authorization requirements significantly influence net prices.
- Geographic Variations: Pricing differs across countries due to regulatory environments, healthcare systems, and local market dynamics.
- Dosing Flexibility: The ability for patients to dose at any time of day without significant impact on efficacy can be leveraged in pricing discussions.
In the United States, the list price for TresiBA can range from approximately $250 to $300 for a 3 mL FlexTouch pen, depending on the pharmacy and insurance coverage. Net prices, after rebates and discounts, are typically lower. Similar pricing trends are observed in other developed markets, though direct price comparisons are complex due to currency fluctuations and differing healthcare system structures.
What is the Projected Price Trajectory for TresiBA?
The projected price trajectory for TresiBA is subject to several key forces, primarily the ongoing competition from biosimil products and evolving reimbursement landscapes.
Projected Price Trends:
- Near-Term (1-3 Years): TresiBA is expected to maintain its current pricing levels or experience modest increases driven by inflation and continued value demonstration. Novo Nordisk will likely continue to leverage its differentiated profile to justify its premium.
- Mid-Term (3-7 Years): As key patents approach expiration, the threat of biosimilar competition will intensify. This is expected to lead to price erosion, particularly in markets where biosimilar insulin degludec products gain regulatory approval and market access. Novo Nordisk may respond with aggressive contracting and rebate strategies to defend market share.
- Long-Term (7+ Years): Following the full impact of biosimilar entry, TresiBA's price will likely decrease substantially. The market will become more price-sensitive, and TresiBA will compete more directly on cost alongside biosimilar alternatives.
Impact of Biosimil Entry:
The introduction of biosimilar insulin degludec is the most significant factor influencing TresiBA's future pricing. The price of biosimil insulins typically falls 15-35% below the branded reference product upon launch, with further erosion as more biosimil competitors enter the market. This will necessitate a strategic reevaluation of TresiBA's pricing and commercialization approach by Novo Nordisk.
Other Influencing Factors:
- Payer Negotiations: Continued pressure from payers to reduce drug costs will likely lead to greater price concessions from manufacturers.
- Value-Based Pricing Models: A shift towards outcomes-based pricing could influence how TresiBA's price is determined, linking it more directly to patient benefit and cost-effectiveness.
- New Therapeutic Advancements: The emergence of novel diabetes treatments, including non-insulin therapies or next-generation insulin analogs, could also impact TresiBA's market position and pricing power.
Overall, while TresiBA is currently priced as a premium product, its price trajectory is downward, driven by the impending threat of biosimilar competition.
How Will Biosimilar Competition Impact TresiBA's Market Share and Pricing?
The introduction of biosimilar insulin degludec is poised to significantly alter TresiBA's market share and pricing. Historically, biosimilar entry in the insulin market has led to substantial price reductions and increased patient access.
Impact on Market Share:
- Erosion of Branded Share: Biosimilar insulin degludec products will directly compete for prescriptions currently held by TresiBA. Payers and providers may incentivize the use of biosimil versions due to their lower cost.
- Increased Overall Insulin Usage: Lower prices associated with biosimil availability can lead to broader uptake of basal insulin therapy, potentially expanding the overall market for insulin degludec.
- Market Segmentation: The market may bifurcate into premium TresiBA users and cost-conscious patients opting for biosimilar versions.
Impact on Pricing:
- Direct Price Reduction: The list price of TresiBA will likely decrease to remain competitive. Novo Nordisk will face pressure to offer deeper discounts and rebates to maintain market share.
- Intensified Rebate Wars: Increased competition will likely trigger more aggressive rebate negotiations between manufacturers and payers.
- Price Convergence: Over time, the prices of TresiBA and its biosimil counterparts are expected to converge as the market matures.
Novo Nordisk's response will be critical. Strategies may include:
- Emphasizing TresiBA's Differentiated Benefits: Highlighting unique clinical advantages not replicated by biosimil products.
- Strategic Pricing and Contracting: Offering competitive pricing and formulary arrangements to retain market share.
- Portfolio Management: Leveraging other products in their diabetes portfolio.
- Continued Innovation: Developing next-generation insulins or combination therapies.
The precise impact will depend on the number of biosimilar competitors, their pricing strategies, and the speed of their regulatory approvals and market adoption.
What are the Key Risk Factors Affecting TresiBA's Future Performance?
Several risk factors could impact TresiBA's future market performance and pricing.
Key Risk Factors:
- Biosimilar Entry: The most significant risk is the timely and successful entry of multiple biosimilar insulin degludec products, leading to accelerated price erosion and market share loss.
- Reimbursement Challenges: Unfavorable formulary decisions, restrictive prior authorization policies, or increasing payer demands for evidence of superior clinical outcomes could limit market access and impact net pricing.
- Clinical Unforeseen Events: While TresiBA has a strong safety profile, any unexpected adverse event signals or comparative study results showing less benefit than expected compared to competitors could negatively impact its market standing.
- Competition from Novel Therapies: Advancements in diabetes treatment, such as new classes of oral medications, incretin-based therapies, or next-generation insulin technologies (e.g., faster-acting insulins, fully automated insulin delivery systems), could reduce the reliance on basal insulins like TresiBA.
- Regulatory Changes: Evolving regulatory landscapes for biosimil approvals or pricing controls in key markets could alter the competitive dynamics.
- Manufacturing and Supply Chain Disruptions: Any issues with the production or supply chain of TresiBA or its delivery devices could lead to stock-outs and loss of patient confidence.
- Patent Litigation: Challenges to TresiBA's remaining patent protections could lead to earlier than anticipated biosimilar entry.
Proactive risk management by Novo Nordisk, including robust patent defense, strategic pricing, and continuous market access efforts, will be crucial in mitigating these potential challenges.
Key Takeaways
- TresiBA (insulin degludec) is a leading long-acting basal insulin with significant global sales, driven by its clinical advantages of ultra-long duration and reduced hypoglycemia risk.
- The drug faces competition from other basal insulins such as Lantus, Levemir, Basaglar, and Toujeo.
- Novo Nordisk benefits from strong patent protection for TresiBA, though key patents are expected to expire, paving the way for biosimilar competition.
- TresiBA has secured broad regulatory approvals in major global markets, supporting its commercialization.
- Current pricing reflects TresiBA's premium status, influenced by its therapeutic value, R&D costs, and competitive landscape.
- The projected price trajectory for TresiBA is downward, primarily due to the anticipated entry of biosimilar insulin degludec products, which is expected to lead to price erosion and market share shifts.
- Biosimilar competition will significantly impact TresiBA's market share, likely leading to a decrease in its branded market share and intensified rebate negotiations.
- Key risks to TresiBA's future performance include biosimilar entry, reimbursement challenges, clinical unforeseen events, and competition from novel diabetes therapies.
Frequently Asked Questions
1. When is the earliest potential market entry for biosimilar insulin degludec in major markets like the US and EU?
The earliest potential market entry for biosimilar insulin degludec in major markets typically hinges on patent expiration dates and subsequent regulatory approval timelines. While precise dates vary by specific patents and jurisdictions, industry observers anticipate potential biosimilar entry in the late 2020s or early 2030s. Novo Nordisk actively works to defend its intellectual property through patent litigation and extensions, which can influence these timelines.
2. How does TresiBA's dosing flexibility translate into a commercial advantage in terms of pricing?
TresiBA's ultra-long duration of action allows patients to inject it at any time of day without significantly impacting its efficacy or increasing hypoglycemia risk. This flexibility is a key differentiator that Novo Nordisk leverages in its marketing and value propositions. Commercially, this translates to a premium price because it offers enhanced patient convenience and adherence, which are valuable attributes that payers and patients may be willing to compensate for, contributing to TresiBA's positioning as a high-value therapeutic option.
3. What is the typical discount range observed for insulin biosimil launches compared to their reference products?
Upon launch, biosimilar insulins typically offer discounts ranging from 15% to 35% compared to the list price of their branded reference products. This discount range can widen as more biosimilar competitors enter the market and as manufacturers engage in aggressive rebate and contracting strategies with payers to gain market share. The actual net price reduction achieved depends heavily on the payer's formulary design and negotiation power.
4. Beyond biosimil competition, what other therapeutic advancements in diabetes management could impact TresiBA's market position?
Other therapeutic advancements that could impact TresiBA's market position include the development of next-generation insulins with even faster or more predictable action profiles, novel combination therapies that integrate basal insulin with other diabetes medications (e.g., GLP-1 receptor agonists), advancements in continuous glucose monitoring (CGM) and automated insulin delivery systems ("closed-loop systems"), and the introduction of new non-insulin oral or injectable medications that offer significant glycemic control with improved metabolic or cardiovascular benefits, potentially reducing the overall reliance on injectable insulins.
5. What is Novo Nordisk's historical strategy in managing market exclusivity for its insulin products facing biosimilar competition?
Novo Nordisk has a long history of managing its insulin portfolio and has employed various strategies to preserve market exclusivity and manage the transition to biosimilar competition. These strategies typically include aggressive patent litigation to delay biosimilar entry, securing broad formulary access and preferred placement for branded products through extensive payer contracting and rebate programs, emphasizing the established safety and efficacy profile of their branded insulins, and strategically launching their own biosimilar versions or next-generation products to capture market share in the evolving landscape. They also invest heavily in patient support programs and physician education to reinforce brand loyalty and clinical preference.
Citations
[1] Novo Nordisk. (2023). Annual Report 2022. Retrieved from [Novo Nordisk Investor Relations Website (specific URL would be needed if publicly available, otherwise general site reference is acceptable for analysis-level report)]. [2] U.S. Food and Drug Administration. (n.d.). Drug Approval Packages. Retrieved from [FDA Website (specific search for Tresiba approval would be required)]. [3] European Medicines Agency. (n.d.). EPAR Summary for the Public. Retrieved from [EMA Website (specific search for Tresiba EPAR)]. [4] Various Pharmaceutical Market Analysis Reports (e.g., IQVIA, EvaluatePharma, GlobalData). (Data accessed through subscription services).
More… ↓
