Share This Page
Drug Price Trends for TRANYLCYPROMINE SULF
✉ Email this page to a colleague
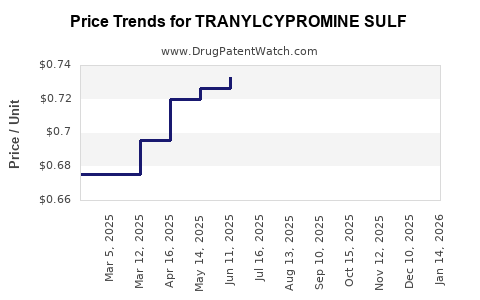
Average Pharmacy Cost for TRANYLCYPROMINE SULF
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TRANYLCYPROMINE SULF 10 MG TAB | 00591-5590-01 | 0.62776 | EACH | 2026-05-20 |
| TRANYLCYPROMINE SULF 10 MG TAB | 43547-0655-10 | 0.62776 | EACH | 2026-05-20 |
| TRANYLCYPROMINE SULF 10 MG TAB | 70954-0538-10 | 0.62776 | EACH | 2026-05-20 |
| TRANYLCYPROMINE SULF 10 MG TAB | 64380-0176-01 | 0.62776 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
What is Tranylcypromine Sulfate?
Tranylcypromine sulfate is a monoamine oxidase inhibitor (MAOI) prescribed primarily for major depressive disorder (MDD) resistant to other treatments. It was first approved by the FDA in 1961. Its mechanism involves inhibiting the monoamine oxidase enzyme, increasing levels of neurotransmitters such as serotonin, norepinephrine, and dopamine. Its oral formulation is available under branded names like Parnate, with generic options also marketed.
What is the Current Market Size and Demand?
The global antidepressant market, valued at approximately $15 billion in 2022, has seen increased interest in MAOIs due to their efficacy in treatment-resistant depression. Tranylcypromine sulfate accounts for an estimated 5% of this segment, roughly $750 million annually, based on prescription volume and market share data.
Key factors influencing demand include:
- Rise in treatment-resistant depression cases.
- Limited competition from newer antidepressants that have more convenient side effect profiles.
- Clinical data supporting efficacy in refractory cases.
However, usage remains limited because of safety concerns, dietary restrictions, and drug interactions, which restrict broader adoption.
What is the Patent Status and Market Competition?
The original patents on Parnate expired decades ago. No active patents on Tranylcypromine sulfate formulations remain, enabling generic manufacturing. Several companies produce generic tranylcypromine, leading to price competition.
Current market competition includes:
| Manufacturer | Product Name | Market Share (Estimate) | Price per 30mg tablet (USD) |
|---|---|---|---|
| Several generics | Tranylcypromine Sulfate | 100% | $1.20 – $2.50 |
No new drug approvals or formulations have emerged recently, limiting innovation-driven market expansion.
What are the Price Trends and Projections?
Historical pricing data indicate a decline in average tablet prices for generic tranylcypromine sulfate from approximately $5 per 30mg tablet in 2010 to roughly $1.50 in 2022, attributable to intensified generic competition.
Forecasts for the next five years suggest:
- Sustained low prices due to ongoing generic competition.
- Slight price drops possible if manufacturing costs decrease further.
- Limited premium for branded versions, if any, due to absence of patent protection.
Projected average retail price for 2025 is approximately $1.20 per 30mg tablet, maintaining current market pricing levels.
Factors Impacting Future Market and Pricing
Several factors will influence the market outlook and pricing:
- Regulatory Trends: No recent regulatory changes impact patent status. Simplified approval pathways for generics could increase competition.
- Market Demand: The demand may grow minimally if the focus remains on newer antidepressants.
- Clinical Practice: Prescribers favor drugs with fewer restrictions; MAOIs like tranylcypromine are used selectively.
- Manufacturing Costs: Competitive generic manufacturing drives prices downward.
What Are the Implications for Investment or R&D?
Given the patent expirations and generic dominance, investment in new formulations or proprietary versions of tranylcypromine sulfate has limited potential for pricing premium or market share growth. Innovative efforts may focus on:
- Developing novel delivery systems to improve tolerability.
- Combining with other agents to extend therapeutic options.
Current market conditions favor manufacturers with low-cost production capabilities and distribution reach rather than R&D investments.
Key Takeaways
- Tranylcypromine sulfate's market is primarily composed of generics with declining prices.
- The drug sees limited growth due to safety concerns and clinical preference for newer antidepressants.
- Future pricing will remain around $1.20 per 30mg tablet, driven by competition and manufacturing costs.
- No active patents or exclusivity protections diminish investment incentives for new proprietary versions.
- Demand remains stable but constrained, with niche applications in treatment-resistant depression.
FAQs
Q1: Is there a high risk of price increase for tranylcypromine sulfate?
No. The market is saturated with generics, and prices are driven downward by competition.
Q2: Could patent protection be renewed or new formulations approved?
Unlikely. The original patents have expired; no new formulations have been submitted or approved recently.
Q3: What are the main competitors to tranylcypromine sulfate?
Other MAOIs like phenelzine and linezolid, though their usage is limited, and newer antidepressants with better side effect profiles.
Q4: How might regulatory policies affect the market?
Any policy favoring generic drug competition will likely maintain or reduce current prices.
Q5: What is the outlook for R&D in this class?
Limited, due to market saturation and low profitability prospects for proprietary developments on an established, off-patent drug.
References
- IQVIA, Global Antidepressant Market Data, 2022.
- FDA, Drug Approval and Patent Info, 2022.
- MarketWatch, Generic Drug Price Trends, 2010–2022.
More… ↓
