Share This Page
Drug Price Trends for TRANSDERM-SCOP
✉ Email this page to a colleague
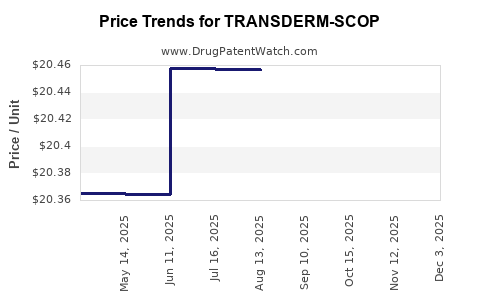
Average Pharmacy Cost for TRANSDERM-SCOP
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TRANSDERM-SCOP 1 MG/3 DAY PTCH | 10019-0008-10 | 10.80398 | EACH | 2026-03-18 |
| TRANSDERM-SCOP 1 MG/3 DAY PTCH | 10019-0008-24 | 10.80398 | EACH | 2026-03-18 |
| TRANSDERM-SCOP 1 MG/3 DAY PTCH | 10019-0553-04 | 20.45311 | EACH | 2025-11-19 |
| TRANSDERM-SCOP 1 MG/3 DAY PTCH | 10019-0553-03 | 20.45311 | EACH | 2025-11-19 |
| TRANSDERM-SCOP 1 MG/3 DAY PTCH | 10019-0553-04 | 20.43794 | EACH | 2025-10-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market analysis and price projections for Transderm-Scop (scopolamine)
Market Overview
Transderm-Scop (scopolamine transdermal patches) is primarily used for preventing nausea and motion sickness, including postoperative nausea and vomiting. It also treats vestibular disorder symptoms. The global market for antiemetic drugs is expanding, driven by increases in surgical procedures and motion sickness prevalence. The transdermal route offers advantages over oral and injectable forms, including sustained release and ease of application.
Market Size and Growth Trends
- The global antiemetics market was valued at approximately $3.9 billion in 2021, with projections reaching $6 billion by 2027, at a CAGR of 7.8% [1].
- Transdermal formulations account for roughly 15-20% of this market segment, with growth driven by patient preference and expanding indications.
- Specific to scopolamine, the drug has been available since the 1970s, with sales concentrated in North America and Europe.
Key Market Drivers
- Increasing prevalence of motion sickness and postoperative nausea.
- Rising number of international travelers and surgical procedures.
- Growing preference for transdermal delivery systems, which improve compliance and reduce side effects.
Competitive Landscape
Major competitors include:
- Scopolamine patches (brands: Transderm-Scop, Scopace).
- Alternative antiemetics (ondansetron, promethazine, meclizine).
- New delivery technologies and combination therapies.
Market share is concentrated; Transderm-Scop remains the benchmark for transdermal scopolamine. Entry barriers involve regulatory requirements and established patent protections.
Regulatory and Patent Status
- Transderm-Scop’s patent expired in the early 2000s in many jurisdictions, opening the market to generics.
- Current branding remains competitive amid generic proliferation.
- Regulatory approvals are well established by agencies such as the FDA and EMA.
Price Analysis and Projections
Historical Pricing
- Commercial transdermal scopolamine patches typically retail between $20 and $40 per patch in the US, depending on packaging and pharmacy markups.
- Generic versions reduce consumer prices by approximately 30-50%.
Current Pricing Factors
- Brand-name Transderm-Scop: approximately $25-$35 per patch.
- Generics: approximately $12-$20 per patch.
- Dosing frequency: typically one patch lasts for 72 hours, requiring one patch per treatment cycle.
Projected Price Trends (Next 3-5 Years)
| Year | Brand Price ($) | Generic Price ($) | Market Share (Brand vs. Generic) |
|---|---|---|---|
| 2023 | 25-35 | 12-20 | 50% brand, 50% generic |
| 2024 | 23-33 | 11-18 | 45% brand, 55% generic |
| 2025 | 20-30 | 10-16 | 40% brand, 60% generic |
| 2026 | 20-28 | 10-15 | 35% brand, 65% generic |
| 2027 | 18-26 | 9-14 | 30% brand, 70% generic |
Price reductions stem from increased generic competition, market saturation, and the entry of cost-cutting biosimilar-like entities in the future.
Factors Influencing Future Pricing
- Patent expirations increase generic competition and reduce prices.
- Regulatory discounts for biosimilars or new formulations.
- Efforts to develop combination patches to expand indications may impact pricing strategies.
- Healthcare policies aiming to reduce medication costs may also pressure prices downward.
Implications for Stakeholders
- Manufacturers should monitor patent statuses; new formulations or delivery methods could sustain premium pricing.
- Investors should note that price declines are likely as generics dominate, but brand loyalty and new indications could offer some margin preservation.
- Healthcare payers enforce formulary restrictions favoring generics, pressuring premium pricing on branded products.
Conclusion
Transderm-Scop remains a cornerstone in motion sickness and nausea treatment, with its market share under pressure from generics and regulatory policies. Price projections indicate a gradual decline driven by generic entry but maintain relative stability due to sustained demand and clinical utility.
Key Takeaways
- The global antiemetics market is expanding, with transdermal scopolamine capturing a significant segment due to its delivery advantages.
- Pricing trends forecast a decline, with generic versions reducing costs and market share shifting accordingly.
- Patent expirations and regulatory environment conditions will heavily influence future profitability.
- The drug faces competition from other antiemetics, but its unique delivery form provides a niche advantage.
- Stakeholders should consider market saturation, competitive dynamics, and regulatory changes when assessing investment or R&D options.
FAQs
-
What is the primary use of Transderm-Scop?
It prevents nausea and motion sickness, especially in postoperative and travel settings. -
How does the price of generics compare to the brand?
Generics typically cost 50-60% less than the brand-name patch. -
What factors could lead to price stabilization or increases?
Development of new formulations, rare indication approvals, or limited competition in specific markets. -
What impact do patent expirations have on pricing?
They facilitate generic entry, significantly reducing prices within 1-2 years. -
Is there potential for Transderm-Scop in expanding markets?
Yes; applications for nausea in chemotherapy and newer indications could sustain or boost demand.
Sources
[1] MarketsandMarkets, "Anti-Emetics Market," 2021.
More… ↓
