Share This Page
Drug Price Trends for TRANEXAMIC ACID
✉ Email this page to a colleague
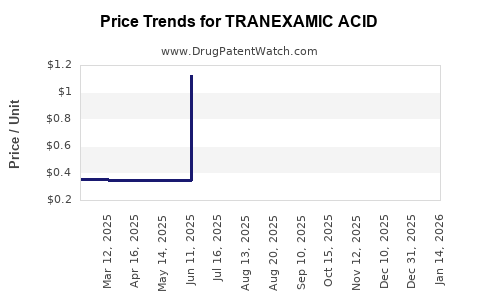
Average Pharmacy Cost for TRANEXAMIC ACID
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TRANEXAMIC ACID 1,000 MG/10 ML | 65145-0106-10 | 0.34808 | ML | 2026-03-25 |
| TRANEXAMIC ACID 650 MG TABLET | 50268-0772-11 | 0.94669 | EACH | 2026-03-18 |
| TRANEXAMIC ACID 650 MG TABLET | 00591-3720-30 | 0.94669 | EACH | 2026-03-18 |
| TRANEXAMIC ACID 650 MG TABLET | 75907-0309-30 | 0.94669 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for TRANEXAMIC ACID
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| TRANEXAMIC ACID 100MG/ML INJ,VIL,10ML | Mylan Institutional LLC | 67457-0197-10 | 10X10ML | 61.80 | 2023-11-15 - 2028-09-28 | FSS | ||
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Tranexamic Acid Market Analysis and Price Projections
Tranexamic acid (TXA) is a synthetic lysine analog used to control bleeding by inhibiting fibrinolysis. Its market is driven by surgical procedures, trauma care, and increasing applications in cosmetic and dermatological treatments. Global demand is projected to grow, influenced by an aging population, rising healthcare expenditures, and expanding indications.
What is the current market size and historical growth of tranexamic acid?
The global tranexamic acid market was valued at approximately USD 500 million in 2023. The market has experienced a compound annual growth rate (CAGR) of 4.5% from 2019 to 2023. Key drivers include a steady increase in the number of surgical procedures worldwide, particularly in orthopedic, cardiothoracic, and gynecological specialties. The growing incidence of trauma cases, requiring rapid hemostatic intervention, also contributes to demand. Furthermore, the expanding use of TXA in topical formulations for hyperpigmentation treatment has opened new market avenues.
What are the primary applications and end-users for tranexamic acid?
Tranexamic acid's primary applications are in preventing and treating excessive bleeding.
Major Applications:
- Surgical Hemostasis: Used in cardiac surgery, orthopedic surgery (e.g., joint replacement), abdominal surgery, and gynecological procedures to reduce blood loss and transfusion requirements.
- Trauma Management: Administered in emergency settings to control bleeding in severe injuries.
- Menorrhagia Treatment: Oral and injectable formulations are prescribed to reduce heavy menstrual bleeding.
- Epistaxis (Nosebleeds): Topical TXA sprays and solutions are used for localized bleeding control.
- Dermatology and Cosmetics: Topical formulations are used to treat melasma, post-inflammatory hyperpigmentation, and other skin discoloration issues.
- Dental Procedures: Used as a mouthwash or topical agent to minimize bleeding after tooth extractions or oral surgeries.
Key End-Users:
- Hospitals and Clinics: The largest segment, utilizing TXA across various surgical departments and emergency services.
- Surgical Centers: Dedicated facilities for elective and specialized surgical procedures.
- Pharmacies: Distributing both prescription and over-the-counter (OTC) TXA products.
- Dermatology Clinics and MedSpas: Employing TXA in cosmetic treatments.
- Ambulatory Care Centers: Providing urgent care and minor surgical services.
What is the projected market growth and forecast for tranexamic acid?
The global tranexamic acid market is forecast to reach approximately USD 750 million by 2029, indicating a projected CAGR of 4.2% from 2024 to 2029. This growth will be sustained by several factors:
- Aging Global Population: An increase in age-related conditions and elective surgeries, particularly in the orthopedic and cardiovascular sectors, will drive demand.
- Advancements in Medical Procedures: Minimally invasive surgical techniques, while often leading to less blood loss, still benefit from TXA's hemostatic properties, especially in complex cases.
- Growing Awareness of TXA's Benefits: Increased clinical studies and physician education are expanding the understanding and adoption of TXA in various medical disciplines.
- Expansion of Cosmetic Applications: The proven efficacy of topical TXA in treating hyperpigmentation is fueling its demand in the burgeoning aesthetic market.
- Emerging Markets: Increased healthcare infrastructure and access to advanced medical treatments in developing economies will contribute to market expansion.
Projected Market Size:
| Year | Market Value (USD Million) |
|---|---|
| 2023 | 500 |
| 2024 | 518 |
| 2025 | 537 |
| 2026 | 557 |
| 2027 | 578 |
| 2028 | 599 |
| 2029 | 620 |
(Note: Values are estimates based on CAGR projections.)
What are the key drivers and restraints impacting the tranexamic acid market?
Key Drivers:
- Increasing Surgical Volume: The rise in elective surgeries, especially orthopedic, cardiac, and gynecological procedures, directly correlates with TXA demand for blood management [1].
- Rising Incidence of Trauma: Global trauma cases, from accidents and natural disasters, necessitate prompt hemostatic agents like TXA.
- Growth in Dermatological Applications: The expanding use of topical TXA for melasma and hyperpigmentation treatments is a significant growth segment [2].
- Favorable Regulatory Landscape: Approvals for new indications and the established safety profile of TXA in many regions support its market penetration.
- Cost-Effectiveness: TXA is generally a more cost-effective hemostatic agent compared to blood transfusions or other advanced therapies, making it a preferred choice in cost-sensitive healthcare systems.
Key Restraints:
- Side Effects and Contraindications: Potential adverse effects, including nausea, vomiting, and thromboembolic events (though rare with proper administration), can limit its use in certain patient populations [3].
- Availability of Alternatives: Other antifibrinolytic agents and hemostatic strategies exist, potentially limiting TXA market share in specific niches.
- Generic Competition and Price Erosion: The presence of multiple generic manufacturers can lead to price pressures, impacting profit margins for some market players.
- Stringent Manufacturing Standards: Maintaining high-quality production standards for pharmaceutical-grade TXA can be a barrier to entry for new manufacturers.
What is the competitive landscape and key players in the tranexamic acid market?
The tranexamic acid market is characterized by a mix of large pharmaceutical companies and specialized manufacturers. Competition is driven by product quality, regulatory approvals, manufacturing capacity, and pricing strategies.
Key Market Players:
- Pfizer Inc.: A major global pharmaceutical company with a significant presence in hemostatic agents.
- Amneal Pharmaceuticals LLC: A prominent generic pharmaceutical manufacturer with a broad portfolio, including TXA.
- Mylan N.V. (now Viatris Inc.): Another leading generic and specialty pharmaceutical company.
- Hikma Pharmaceuticals PLC: A multinational pharmaceutical group with strong manufacturing capabilities.
- Fresenius SE & Co. KGaA: Primarily known for dialysis products, but also involved in injectables and critical care medicines, including TXA.
- Teva Pharmaceutical Industries Ltd.: A global leader in generic and specialty medicines.
- Bayer AG: While not a primary TXA manufacturer, Bayer has a significant presence in women's health, where TXA is used for menorrhagia.
- Sanofi S.A.: A diversified pharmaceutical company with a stake in various therapeutic areas.
- CS Pharma: A specialized pharmaceutical company focusing on critical care and hospital products.
- Taj Pharmaceuticals Ltd.: An Indian pharmaceutical company with global reach.
(Note: This list is not exhaustive but represents key entities influencing the market.)
Market share is fragmented, with leading generic players holding substantial portions due to competitive pricing and widespread distribution networks. Innovation in formulation, particularly for topical and specialized applications, is a key differentiator.
What are the price trends and projections for tranexamic acid?
The pricing of tranexamic acid is influenced by formulation, dosage, region, and whether it is branded or generic.
Current Price Range (Representative):
- Injectable (e.g., 100mg/mL vial): USD 5 - USD 15 per vial (hospitals and institutional pricing may vary significantly).
- Oral Tablets (e.g., 500mg): USD 0.50 - USD 2.00 per tablet (depending on quantity and brand).
- Topical Formulations (e.g., serums, creams): USD 20 - USD 80 per product (varying by concentration, brand, and retail channel).
Price Projections:
The overall price for TXA injections and tablets is expected to remain relatively stable, with a slight downward pressure due to ongoing generic competition. However, slight increases may occur due to rising raw material costs and manufacturing expenses.
- Injectable TXA: Expected to see a CAGR of 1-2% increase in average selling price (ASP) due to operational costs and demand from critical care.
- Oral TXA: Predicted to experience slight price erosion or stability (0-1% CAGR) due to intense generic competition.
- Topical/Cosmetic TXA: This segment may see a more significant price increase (3-5% CAGR) driven by premium branding, specialized formulations, and higher perceived value in the aesthetics market.
Factors influencing price projections:
- Raw Material Costs: Fluctuations in the cost of lysine and other chemical precursors.
- Manufacturing Efficiency: Advances in production technology can lower costs.
- Regulatory Hurdles: Compliance with evolving Good Manufacturing Practices (GMP) can add costs.
- Demand-Supply Dynamics: Regional shortages or surpluses can impact pricing.
- Patent Expirations: While TXA itself is an old molecule, new formulations or delivery systems could be patented, influencing pricing for those specific products.
What are the regulatory considerations and market access challenges for tranexamic acid?
Regulatory approval for tranexamic acid is governed by national health authorities such as the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), and others.
Key Regulatory Considerations:
- API Manufacturing Standards: Active Pharmaceutical Ingredient (API) manufacturers must adhere to stringent GMP guidelines to ensure quality, purity, and safety. Inspections and certifications are critical.
- Drug Master Files (DMFs): Manufacturers of TXA API must maintain DMFs, which are detailed submissions to regulatory agencies containing information about the manufacturing process, facilities, and quality controls.
- Product Registration: Finished dosage forms (tablets, injectables, topical solutions) require product-specific marketing authorization from relevant regulatory bodies. This involves extensive clinical and non-clinical data submission.
- Labeling and Prescribing Information: Adherence to standardized labeling requirements, including indications, contraindications, warnings, and adverse reactions, is mandatory.
- Pharmacovigilance: Post-market surveillance and reporting of adverse events are critical for ongoing market access and patient safety.
Market Access Challenges:
- Reimbursement Policies: While TXA is generally well-reimbursed for approved medical indications, varying healthcare policies across countries can impact patient access and provider adoption.
- Formulary Placement: Securing inclusion in hospital and insurance formularies can be competitive, requiring demonstration of clinical and economic value.
- Generic Competition: For established indications, aggressive pricing from generic manufacturers can challenge market entry for new players.
- Geographic Variations: Different regulatory pathways and market dynamics exist in North America, Europe, Asia-Pacific, and other emerging regions, requiring tailored market access strategies.
- Off-Label Use Concerns: While TXA has off-label uses (e.g., in cosmetics), these are not typically covered by standard medical reimbursement and may face different regulatory scrutiny.
What are the future trends and innovations expected in the tranexamic acid market?
The tranexamic acid market is expected to evolve with advancements in its application and delivery.
Key Future Trends and Innovations:
- Novel Drug Delivery Systems: Research into improved delivery methods for TXA, such as sustained-release formulations or targeted delivery systems, could enhance efficacy and reduce side effects.
- Combination Therapies: Exploring the synergistic effects of TXA when used in combination with other hemostatic agents or therapeutic compounds for enhanced outcomes in complex bleeding scenarios.
- Expanded Dermatological Applications: Further research into TXA's mechanism of action in skin conditions could lead to new cosmetic and dermatological indications, beyond hyperpigmentation. This may include wound healing and anti-inflammatory applications.
- Biologics and Biosimilars: While TXA is a small molecule, the broader trend of biosimilar development might influence market dynamics for complex biologic hemostatic agents, indirectly impacting TXA's positioning.
- Personalized Medicine: Future applications may involve tailoring TXA therapy based on individual patient profiles, genetic predispositions, or specific bleeding risk assessments.
- Digital Health Integration: Wearable devices or diagnostic tools that monitor bleeding risk or TXA efficacy could emerge, integrating TXA management into a broader digital health ecosystem.
- Sustainability in Manufacturing: Increasing focus on greener chemical synthesis processes and sustainable packaging for TXA production and distribution.
Key Takeaways
- The tranexamic acid market is projected to grow to approximately USD 750 million by 2029, driven by surgical demand, trauma care, and expanding cosmetic applications.
- Key market players include established pharmaceutical giants and agile generic manufacturers, with competition focused on quality, cost, and regulatory compliance.
- Injectable and oral formulations are expected to see stable to slightly declining prices due to generic competition, while premium topical formulations are poised for price increases.
- Navigating diverse regulatory pathways and reimbursement policies across global markets presents ongoing challenges for market access.
- Future innovation will likely focus on novel delivery systems, combination therapies, and expanded applications, particularly in dermatology.
Frequently Asked Questions
-
What is the primary medical indication that drives the most significant demand for tranexamic acid? The primary medical indication driving the most significant demand for tranexamic acid is its use in controlling and preventing excessive bleeding during surgical procedures, particularly in cardiac and orthopedic surgeries.
-
Are there any significant patent expiries on tranexamic acid itself that could impact market competition? Tranexamic acid is an old molecule, and its basic compound patents expired long ago. Competition primarily stems from generic manufacturers producing standard formulations. However, new patents may exist for novel delivery systems, combination therapies, or specific therapeutic uses.
-
How does the price of tranexamic acid compare to alternative hemostatic agents? Tranexamic acid is generally considered a cost-effective hemostatic agent. Compared to some recombinant or biologic hemostatic agents, it offers a lower cost per dose, making it a preferred option in many clinical settings, especially where cost containment is a priority.
-
What are the main regulatory hurdles for a new company looking to enter the tranexamic acid manufacturing market? A new company must navigate stringent Good Manufacturing Practices (GMP) for API production, obtain necessary certifications, file comprehensive Drug Master Files (DMFs) with regulatory agencies, and secure marketing authorization for finished dosage forms, which involves demonstrating safety and efficacy.
-
Beyond bleeding control, what is the most rapidly growing non-surgical application for tranexamic acid? The most rapidly growing non-surgical application for tranexamic acid is in dermatology and cosmetic treatments, specifically for addressing hyperpigmentation conditions like melasma and post-inflammatory hyperpigmentation, via topical formulations.
Citations
[1] National Institutes of Health. (2023). Tranexamic Acid for Perioperative Hemorrhage Control. National Library of Medicine. [2] American Academy of Dermatology Association. (2022). Melasma: Diagnosis and Treatment. [3] European Medicines Agency. (2018). Assessment Report: Tranexamic Acid.
More… ↓
