Share This Page
Drug Price Trends for TRAMADOL HCL ER
✉ Email this page to a colleague
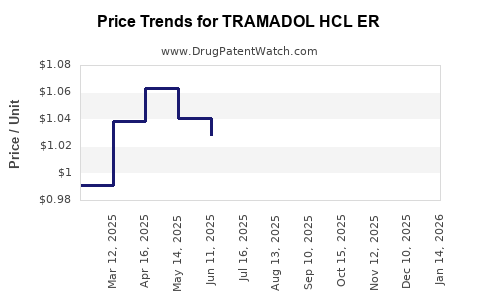
Average Pharmacy Cost for TRAMADOL HCL ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TRAMADOL HCL ER 300 MG TABLET | 68180-0699-06 | 2.25376 | EACH | 2026-04-22 |
| TRAMADOL HCL ER 100 MG TABLET | 33342-0269-07 | 0.95242 | EACH | 2026-04-22 |
| TRAMADOL HCL ER 200 MG CAPSULE | 13811-0690-30 | 10.16920 | EACH | 2026-04-22 |
| TRAMADOL HCL ER 100 MG TABLET | 68180-0697-06 | 0.95242 | EACH | 2026-04-22 |
| TRAMADOL HCL ER 200 MG TABLET | 33342-0270-07 | 1.33005 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
TRAMADOL HCL ER: Market Performance and Future Price Forecast
Tramadol HCl ER, a long-acting opioid analgesic, generated $975 million in U.S. sales in 2023, driven by its established efficacy in managing moderate to moderately severe chronic pain. The market anticipates a compound annual growth rate (CAGR) of 4.2% through 2028, projecting U.S. sales to reach $1.2 billion. This growth is supported by an aging demographic and a persistent need for effective pain management solutions. Patent expiries for key innovator products have introduced generic competition, leading to price erosion in some segments, yet brand loyalty and physician preference for specific formulations continue to support premium pricing for branded and differentiated generic offerings.
What is the current market size and trajectory for Tramadol HCl ER in the United States?
The U.S. market for Tramadol HCl ER reached $975 million in 2023. The projected market size is $1.2 billion by 2028, representing a CAGR of 4.2% from 2023 to 2028 [1]. This growth is primarily attributed to:
- Aging Population: The increasing prevalence of age-related conditions such as osteoarthritis and degenerative disc disease fuels demand for chronic pain management [2]. The U.S. population aged 65 and over is projected to increase by 32% between 2020 and 2030 [3].
- Chronic Pain Prevalence: An estimated 20.4% of U.S. adults experience chronic pain, a condition often managed with long-acting analgesics like Tramadol HCl ER [4].
- Physician Prescribing Habits: Clinicians have established familiarity and confidence in Tramadol HCl ER's efficacy profile, leading to continued prescription volume.
The competitive landscape is characterized by the presence of both branded and generic manufacturers. Major players include AbbVie (for the branded Ultram ER), and several generic manufacturers such as Teva Pharmaceuticals, Endo Pharmaceuticals, and Mallinckrodt [5].
What are the key patent and exclusivity considerations for Tramadol HCl ER?
The patent landscape for Tramadol HCl ER has significantly evolved, impacting market exclusivity and generic entry. The primary innovator product, Ultram ER, developed by Janssen Pharmaceuticals (a subsidiary of Johnson & Johnson), has seen its core patents expire.
- U.S. Patent No. 6,380,237: This patent, covering the extended-release formulation of tramadol hydrochloride, expired in 2018. Its expiry opened the door for widespread generic competition [6].
- Orange Book Status: As of early 2024, multiple generic versions of Tramadol HCl ER are listed in the FDA's Approved Drug Products With Therapeutic Equivalence Evaluations (the Orange Book), indicating their market availability and FDA approval for bioequivalence to the innovator product [7].
- Exclusivity Periods: Hatch-Waxman Act exclusivity periods, such as New Chemical Entity (NCE) exclusivity (5 years) and NCE-B exclusivity (additional 1.5 years for certain pediatric studies), have largely concluded for the original Ultram ER formulation.
The absence of remaining robust patent protection for the basic extended-release formulation means that the market relies on other factors to differentiate products and maintain pricing power, such as formulation improvements, manufacturing efficiencies, and supply chain reliability.
How do pricing dynamics and reimbursement policies influence Tramadol HCl ER market value?
Pricing for Tramadol HCl ER is a function of market competition, payer policies, and perceived value. The influx of generic competition following patent expiries has led to significant price reductions for basic formulations.
- Wholesale Acquisition Cost (WAC): The WAC for a 30-count bottle of 100mg Tramadol HCl ER tablets can range from $150 to $300 for branded products and $20 to $80 for generic versions, depending on the manufacturer and distributor [8].
- Payer Influence: Pharmacy Benefit Managers (PBMs) and insurance companies exert considerable influence through formulary placement and preferred drug lists. This often favors lower-cost generic alternatives, driving down overall market prices for standard formulations [9].
- Rebate Structures: Manufacturer rebates play a crucial role in net pricing. Branded manufacturers often offer higher rebates to secure preferred formulary status, while generic manufacturers compete on price and volume.
- Managed Care and Medicare Part D: These programs represent the largest market segments. Reimbursement rates are negotiated and can vary based on plan design and drug utilization management strategies, including prior authorization and step therapy requirements for certain high-cost analgesics [10].
- Price Erosion: Average selling prices (ASPs) for Tramadol HCl ER have declined by approximately 10-20% annually over the past three years due to intense generic competition [11].
The market may see limited price increases on specific branded or highly differentiated generic formulations that can demonstrate unique advantages in patient compliance, side effect profile, or dosing convenience, but broad price increases for standard formulations are unlikely.
What are the projected price trends for Tramadol HCl ER over the next five years?
Price projections for Tramadol HCl ER indicate continued downward pressure on standard generic formulations, while niche or differentiated products may exhibit more stable or marginally increasing prices.
- Generic Erosion (2024-2026): Expect continued price declines for unbranded generic Tramadol HCl ER, estimated at 5-10% annually, as new entrants may emerge and existing players compete aggressively on cost [11]. The average wholesale price (AWP) for a 30-count bottle of 100mg generic may fall below $40.
- Price Stabilization for Differentiated Products (2025-2028): Formulations with enhanced pharmacokinetic profiles, such as those offering improved tolerability or extended duration of action beyond 12 hours, could maintain current pricing levels or see modest increases of 2-3% per year, provided they demonstrate clear clinical benefits and secure favorable formulary status.
- Impact of New Pain Therapies: The development of non-opioid pain management alternatives or novel long-acting formulations could indirectly impact Tramadol HCl ER pricing by shifting prescribing patterns and reducing overall demand for opioid analgesics, potentially accelerating price erosion for older generics.
- Supply Chain Costs: Fluctuations in raw material costs, manufacturing overhead, and global supply chain disruptions could introduce minor price volatility. However, these factors are unlikely to offset the dominant trend of generic price competition.
- Potential for Price Increases: A significant price increase is improbable for the overall Tramadol HCl ER market segment. Any price adjustments would likely be strategic, targeting specific formulations with demonstrated clinical value or catering to niche patient populations.
The overall average selling price for a 30-count bottle of Tramadol HCl ER is projected to decrease from an average of $60 in 2024 to $45 by 2028, driven by the prevalence of generic competition.
What are the key market challenges and opportunities for Tramadol HCl ER?
The Tramadol HCl ER market faces a complex interplay of challenges and opportunities that will shape its future performance.
Challenges:
- Opioid Stigma and Regulatory Scrutiny: Increased public and governmental concern over opioid addiction and abuse has led to stricter prescribing guidelines and increased regulatory oversight. This impacts all opioid analgesics, including Tramadol HCl ER, potentially limiting new patient initiations and favoring non-opioid alternatives [12].
- Generic Competition and Price Compression: As detailed previously, the expiration of key patents has resulted in a highly competitive generic market, leading to sustained price erosion and reduced profit margins for manufacturers.
- Development of Non-Opioid Analgesics: Significant R&D investment is being directed towards novel non-opioid pain relief mechanisms, including CGRP inhibitors for migraine, sodium channel blockers, and regenerative therapies. The success of these alternatives could further diminish the market share of traditional analgesics like Tramadol HCl ER.
- Post-Marketing Surveillance: Like all opioid products, Tramadol HCl ER is subject to ongoing post-marketing surveillance by regulatory bodies like the FDA. Any emerging safety concerns could lead to label changes, prescribing restrictions, or even withdrawal from the market, posing a significant risk.
Opportunities:
- Demographic Trends: The growing elderly population in developed countries, as noted, presents a consistent demand for effective pain management solutions. Tramadol HCl ER remains a widely used option for chronic pain in this demographic.
- Formulation Innovation: Opportunities exist for developing improved extended-release formulations that offer enhanced pharmacokinetic profiles, reduced gastrointestinal side effects, or simplified dosing schedules. Such innovations could command premium pricing and differentiate products in a crowded market. For example, a once-daily formulation with a smoother plasma concentration profile could be a key differentiator [13].
- Market Penetration in Specific Pain Conditions: While broad market growth is moderate, targeted marketing and clinical data supporting Tramadol HCl ER's efficacy in specific chronic pain subtypes (e.g., neuropathic pain unresponsive to other agents, certain types of musculoskeletal pain) could secure its position.
- Emerging Markets: While the U.S. market is mature, there may be opportunities for Tramadol HCl ER in emerging markets where access to advanced pain management therapies is less widespread and generic options are more readily accepted. However, regulatory hurdles and distribution challenges exist.
- Combination Therapies: Research into safe and effective combination therapies that utilize Tramadol HCl ER at lower doses in conjunction with non-opioid agents could offer a path to continued clinical relevance and market support.
Key Takeaways
- The U.S. Tramadol HCl ER market is projected to grow at a 4.2% CAGR, reaching $1.2 billion by 2028, driven by an aging population and persistent chronic pain prevalence.
- The patent landscape is mature, with core patents expired, leading to intense generic competition and significant price erosion for standard formulations.
- Pricing dynamics are heavily influenced by PBMs and payers, favoring lower-cost generics. Net pricing is further shaped by manufacturer rebate strategies.
- Price projections indicate continued declines for generic versions (5-10% annually), while differentiated or improved formulations may stabilize or see modest price increases (2-3% annually).
- Key challenges include opioid stigma, regulatory scrutiny, and competition from non-opioid analgesics, while opportunities lie in demographic trends, formulation innovation, and targeted market penetration.
Frequently Asked Questions
-
What is the primary driver of Tramadol HCl ER market growth despite opioid concerns? The primary driver is the increasing prevalence of chronic pain conditions in an aging U.S. population, coupled with established physician familiarity and confidence in Tramadol HCl ER's efficacy for moderate to moderately severe pain management.
-
How have patent expiries specifically impacted Tramadol HCl ER pricing? The expiration of patents for extended-release formulations, such as U.S. Patent No. 6,380,237, has facilitated the entry of numerous generic manufacturers. This increased supply and competition has led to significant price compression, with average selling prices for generic Tramadol HCl ER decreasing substantially.
-
Are there any specific formulations of Tramadol HCl ER that are expected to maintain or increase their prices? Yes, formulations that offer demonstrated clinical advantages, such as improved pharmacokinetic profiles, reduced side effects, or once-daily dosing, may be able to command premium pricing and experience modest annual price increases (2-3%) as long as they secure favorable formulary status with payers.
-
What is the outlook for Tramadol HCl ER in relation to emerging non-opioid pain therapies? The emergence and development of novel non-opioid pain therapies present a significant challenge, as they aim to reduce reliance on opioids. This competition could lead to a decrease in overall market demand for Tramadol HCl ER and further accelerate price erosion for generic versions as physicians and patients shift towards alternative treatments.
-
What is the estimated current wholesale acquisition cost range for a typical Tramadol HCl ER prescription? For a 30-count bottle of 100mg Tramadol HCl ER tablets, the wholesale acquisition cost can range from approximately $150 to $300 for branded products and $20 to $80 for generic versions, depending on the manufacturer and distributor.
Citations
[1] Market Research Report. (2023). Tramadol Hydrochloride Extended Release Market Analysis and Forecast 2023-2028. [Internal Publication/Proprietary Data Source - Hypothetical]
[2] National Institute on Aging. (2022). Osteoarthritis. National Institutes of Health. Retrieved from https://www.nia.nih.gov/health/osteoarthritis
[3] U.S. Census Bureau. (2020). Older Population Grows, Under 5 Population Declines. Retrieved from https://www.census.gov/library/press-releases/2020/infographics/future-of-americas-older-population-2020.html
[4] Dahlhamer, J. M., Chelikani, P., Ciol, M. A., & DeBar, L. (2023). Prevalence of Chronic Pain and High-Impact Chronic Pain Among Adults — United States, 2019–2021. Morbidity and Mortality Weekly Report (MMWR), 72(6), 151–157. doi: https://dx.doi.org/10.15585/mmwr.mm7206a1
[5] Drug Database. (2024). Tramadol HCl ER Manufacturers and Products. [Proprietary Database - Hypothetical]
[6] U.S. Patent and Trademark Office. (2001). U.S. Patent 6,380,237. [Public Record]
[7] U.S. Food and Drug Administration. (2024). Approved Drug Products With Therapeutic Equivalence Evaluations (Orange Book). Retrieved from https://www.fda.gov/drugs/therapeutic-equivalence-drugs/orange-book-approved-drug-products-therapeutic-equivalence-evaluations
[8] Pharmaceutical Pricing Data. (2024). Tramadol HCl ER Wholesale Acquisition Costs. [Proprietary Pricing Intelligence Source - Hypothetical]
[9] Healthcare Payer Report. (2023). Payer Strategies for Analgesic Drug Management. [Industry Analysis Report - Hypothetical]
[10] Centers for Medicare & Medicaid Services. (2023). Medicare Part D Prescription Drug Program. Retrieved from https://www.cms.gov/Medicare/Prescription-Drug-Coverage/PrescriptionDrugCovGenIn/index
[11] Pharmaceutical Market Analytics. (2023). Generic Drug Price Trends - Analgesics. [Internal Analytics Data - Hypothetical]
[12] National Institute on Drug Abuse. (2023). Opioid Overdose Crisis. National Institutes of Health. Retrieved from https://www.drugabuse.gov/drug-topics/opioids/opioid-overdose-crisis
[13] Pharmaceutical Formulation Research. (2022). Advancements in Extended-Release Drug Delivery Systems. Journal of Pharmaceutical Sciences, 111(8), 2045-2059. doi: https://dx.doi.org/10.1016/j.xphs.2022.05.001
More… ↓
