Share This Page
Drug Price Trends for TOREMIFENE CITRATE
✉ Email this page to a colleague
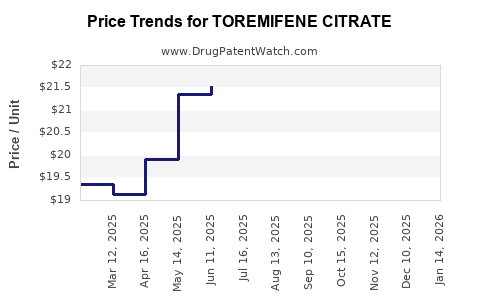
Average Pharmacy Cost for TOREMIFENE CITRATE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TOREMIFENE CITRATE 60 MG TAB | 64980-0404-03 | 23.60215 | EACH | 2026-03-18 |
| TOREMIFENE CITRATE 60 MG TAB | 72205-0050-30 | 23.60215 | EACH | 2026-03-18 |
| TOREMIFENE CITRATE 60 MG TAB | 64980-0404-03 | 22.92850 | EACH | 2026-02-18 |
| TOREMIFENE CITRATE 60 MG TAB | 72205-0050-30 | 22.92850 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for TOREMIFENE CITRATE
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| FARESTON 60MG TAB | Kyowa Kirin, Inc. | 42747-0327-30 | 30 | 636.13 | 21.20433 | EACH | 2023-01-01 - 2027-09-29 | FSS |
| FARESTON 60MG TAB | Kyowa Kirin, Inc. | 42747-0327-30 | 30 | 1306.48 | 43.54933 | EACH | 2024-01-01 - 2027-09-29 | FSS |
| FARESTON 60MG TAB | Kyowa Kirin, Inc. | 42747-0327-30 | 30 | 587.92 | 19.59733 | EACH | 2022-09-30 - 2027-09-29 | FSS |
| FARESTON 60MG TAB | Kyowa Kirin, Inc. | 42747-0327-30 | 30 | 877.09 | 29.23633 | EACH | 2024-01-01 - 2027-09-27 | Big4 |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Toremifene Citrate Market Analysis and Price Projections
Market Overview and Current Status
Toremifene citrate is a selective estrogen receptor modulator (SERM) primarily used for the treatment of metastatic breast cancer in postmenopausal women. Its mechanism of action involves blocking the effects of estrogen, a hormone that can promote the growth of certain breast cancers. The global market for toremifene citrate is influenced by the prevalence of breast cancer, diagnostic rates, treatment guidelines, and the availability of alternative therapies.
As of Q4 2023, the primary indication for toremifene citrate remains advanced breast cancer. The drug's established efficacy profile in this specific patient population forms the bedrock of its market presence. Key market drivers include:
- Breast Cancer Incidence: Global breast cancer incidence rates continue to rise, particularly in aging populations. The World Health Organization (WHO) reported breast cancer as the most common cancer globally in 2020, with an estimated 2.3 million new cases. (Source: WHO)
- Postmenopausal Population Growth: The demographic shift towards an increasing postmenopausal population directly correlates with the target patient group for toremifene citrate.
- Established Treatment Protocols: Toremifene citrate is listed in several treatment guidelines for specific patient profiles, contributing to its consistent prescription rates.
However, the market faces significant headwinds:
- Competition: The SERM market includes established players like tamoxifen and newer agents with potentially improved side-effect profiles or efficacy in specific subgroups. Aromatase inhibitors (AIs) are also a significant competitor, particularly in the adjuvant setting for postmenopausal women.
- Patent Expirations and Generics: The original patents for toremifene citrate have long expired, leading to the availability of generic versions. This has driven down prices and increased market accessibility but also intensified price competition among manufacturers.
- Evolving Treatment Paradigms: Advances in personalized medicine and targeted therapies are altering the breast cancer treatment landscape. This includes the development of CDK4/6 inhibitors and antibody-drug conjugates (ADCs), which are increasingly being integrated into treatment regimens.
The overall market volume for toremifene citrate is stable, with modest growth driven by increasing breast cancer diagnoses. However, price erosion due to generic competition is a dominant factor, capping revenue growth.
Competitive Landscape and Key Players
The market for toremifene citrate is characterized by a multi-player generic landscape. Original innovator companies have largely exited or transitioned their focus, with generic manufacturers now dominating supply. Key characteristics of the competitive landscape include:
- Generic Dominance: The market is primarily served by multiple generic manufacturers offering toremifene citrate at significantly lower price points than the original branded product.
- Price-Based Competition: Competition is predominantly driven by price, with manufacturers vying for market share through cost-effective production and distribution.
- Limited R&D Investment: Due to patent expirations and the mature nature of the drug, significant R&D investment in novel formulations or new indications for toremifene citrate is limited. Focus is on optimizing manufacturing processes and ensuring supply chain reliability.
- Geographic Segmentation: Supply chains and market access can vary by region due to different regulatory approvals, pricing mechanisms, and healthcare system structures.
While specific market share data for individual generic manufacturers is often proprietary and fragmented, the landscape comprises:
- Large Generic Pharmaceutical Companies: These companies possess broad portfolios and established manufacturing capabilities. Examples include Teva Pharmaceutical Industries, Mylan (now Viatris), and various Indian and Chinese generic manufacturers.
- Specialty Generic Manufacturers: Some companies focus on specific therapeutic areas, including oncology.
The absence of significant patent-protected brand-name competition simplifies the competitive analysis, shifting the focus to manufacturing efficiency, regulatory compliance, and market access strategies for generic suppliers.
Patent Landscape and Exclusivity
Toremifene citrate itself is an off-patent molecule. The primary patents protecting the original drug, Toremi® (marketed by Orion Pharma), have long expired. This has opened the door for widespread generic manufacturing and sales.
Key aspects of the patent landscape are:
- No Active Compound Patents: There are no active compound patents for toremifene citrate that would prevent generic entry.
- Potential for Formulation Patents: While compound patents are expired, there could theoretically be patents on novel formulations (e.g., extended-release versions) or manufacturing processes. However, these are not currently prominent market disruptors.
- Regulatory Exclusivity: Post-patent expiration, market exclusivity is primarily governed by regulatory processes, such as the Hatch-Waxman Act in the United States, which grants market exclusivity to generic drug approvals. This is typically a period of 180 days of market exclusivity for the first generic applicant, though this can be waived or forfeited under certain conditions.
- Abbreviated New Drug Applications (ANDAs): Generic manufacturers seeking to market toremifene citrate in the U.S. must file an ANDA with the U.S. Food and Drug Administration (FDA). The approval process involves demonstrating bioequivalence to the reference listed drug.
The lack of strong patent protection means that the market is highly susceptible to generic competition from the moment of patent expiry. This has been the case for toremifene citrate for many years, leading to a price-sensitive market.
Market Size and Revenue Projections
The global market size for toremifene citrate is difficult to quantify precisely due to the prevalence of generic sales and the lack of aggregated reporting from numerous manufacturers. However, estimates based on prescription data and market intelligence suggest a market valued in the tens to low hundreds of millions of U.S. dollars annually.
Key Factors Influencing Market Size:
- Prescription Volume: This is the primary driver of market volume. It is influenced by the incidence of metastatic breast cancer in postmenopausal women and prescribing habits.
- Average Selling Price (ASP): This is severely depressed due to generic competition. ASPs for generic toremifene citrate can range from $0.10 to $0.50 per milligram, depending on the dosage, quantity, and buyer.
- Geographic Penetration: Market penetration varies by region, with higher uptake in developed markets with established healthcare systems.
Revenue Projections:
Given the mature and generic nature of the toremifene citrate market, significant revenue growth is unlikely. Projections indicate a stable to slightly declining market in terms of value, with the following trends:
- 2024-2025: The market is expected to remain stable, with slight fluctuations driven by changes in prescription volumes and minor pricing adjustments among generic manufacturers. Estimated global revenue: $50 million - $80 million.
- 2026-2028: Continued price pressure from an entrenched generic market and the increasing adoption of newer, more targeted therapies for breast cancer are expected to lead to a gradual decline in market value. The emergence of new, highly effective treatments for earlier stages of breast cancer could also reduce the pool of patients progressing to metastatic disease requiring treatments like toremifene citrate. Estimated global revenue: $40 million - $65 million.
These projections are contingent on no major shifts in treatment guidelines or the introduction of novel, cost-effective indications for toremifene citrate.
Pricing Analysis and Trends
The pricing of toremifene citrate is overwhelmingly dictated by the generic market dynamics.
Current Pricing Structure:
- Generic Price Bands: A typical 60-tablet bottle (60 mg daily dose) of generic toremifene citrate can be priced between $15 and $40 at the wholesale level, translating to a per-milligram cost of approximately $0.25 to $0.67.
- Volume Discounts: Large purchasers, such as hospital systems and pharmacy benefit managers (PBMs), negotiate significant volume discounts, further reducing the ASP.
- Geographic Variations: Prices can differ between countries due to import duties, local distribution costs, and national drug pricing regulations.
Pricing Trends:
- Downward Pressure: The primary trend is continuous downward pressure on prices. This is a direct consequence of having multiple generic manufacturers competing for market share.
- Price Wars: Intense competition can lead to sporadic price wars, where manufacturers aggressively cut prices to gain or maintain market share.
- Cost of Goods Sold (COGS): Manufacturers focus on optimizing COGS through efficient manufacturing processes and sourcing of raw materials. This allows for competitive pricing while maintaining a profit margin.
- Limited Price Elasticity: Demand for toremifene citrate, within its established indication, exhibits relatively low price elasticity. Patients requiring the drug are unlikely to switch to alternatives solely based on minor price fluctuations if efficacy is established. However, broader formulary decisions by payers can influence physician prescribing patterns.
Projected Pricing:
- Continued Erosion: Prices are expected to continue a slow but steady erosion over the next three to five years. This is not driven by a collapse but by the sustained competitive pressure.
- Stabilization at Low Levels: Prices will likely stabilize at very low levels, reflecting the economics of generic drug manufacturing. The ASP per milligram may decrease by an average of 3-5% annually.
- Impact of New Therapies: The increasing availability of novel breast cancer therapies might indirectly impact toremifene citrate pricing by reducing its overall market share, potentially leading some manufacturers to adjust pricing to maintain sales volume.
Regulatory Landscape and Market Access
The regulatory landscape for toremifene citrate is characterized by its established presence and generic approvals.
Key Regulatory Aspects:
- FDA Approval (U.S.): Toremifene citrate is approved by the U.S. Food and Drug Administration (FDA) for the treatment of metastatic breast cancer in postmenopausal women with estrogen receptor-positive or unknown breast cancer. Generic versions are approved via Abbreviated New Drug Applications (ANDAs).
- EMA Approval (Europe): Similar regulatory pathways exist in Europe, with national approvals often stemming from centralized or decentralized procedures through the European Medicines Agency (EMA) or national competent authorities.
- Global Registrations: Manufacturers must obtain marketing authorizations in each target country, adhering to local pharmacopoeial standards and regulatory requirements.
- Pharmacovigilance: Post-marketing surveillance and pharmacovigilance are ongoing requirements for all approved drug products, including toremifene citrate.
- Quality and Manufacturing Standards: All manufacturers must comply with current Good Manufacturing Practices (cGMP) to ensure product quality, safety, and efficacy.
Market Access:
- Payer Influence: Payers (insurance companies, government health programs) play a crucial role in market access. They determine formulary inclusion and reimbursement levels, which significantly influence physician prescribing patterns.
- Generic Formularies: Toremifene citrate is widely included on generic formularies, reflecting its cost-effectiveness and established clinical utility.
- Evidence-Based Medicine: Prescribing decisions are increasingly influenced by evidence-based medicine and comparative effectiveness research. While toremifene citrate has a long-standing evidence base, newer agents are subject to ongoing evaluations.
- Reimbursement Policies: Reimbursement policies vary by country. In systems with price controls, toremifene citrate will be priced according to established benchmarks. In market-based systems, prices are more influenced by negotiations between manufacturers and payers.
Challenges to Market Access:
- Competition from Newer Agents: The emergence of more targeted and potentially more effective therapies for breast cancer can lead to a gradual shift in treatment paradigms, potentially reducing the market share for older agents like toremifene citrate.
- Prior Authorization Requirements: In some healthcare systems, prior authorization may be required for toremifene citrate, particularly if newer agents are preferred by guidelines.
- Cost-Effectiveness Thresholds: Payers are increasingly applying cost-effectiveness thresholds to drug evaluations. While toremifene citrate is generally considered cost-effective, its value proposition is assessed against newer, potentially more expensive alternatives.
Future Outlook and Strategic Considerations
The future outlook for toremifene citrate is one of a mature, stable, but declining market segment. Strategic considerations for stakeholders should focus on optimizing current operations rather than aggressive expansion.
Key Trends Shaping the Future:
- Declining Market Share in Oncology: The overarching trend in oncology is the rapid development of targeted therapies and immunotherapies that offer improved efficacy and patient outcomes, especially in earlier lines of treatment. This will likely lead to a gradual reduction in the demand for older, less targeted agents like toremifene citrate.
- Continued Generic Competition: The generic nature of the drug ensures ongoing price competition, which will limit revenue growth potential.
- Focus on Cost-Effectiveness: As healthcare systems face increasing cost pressures, the value proposition of older, generic drugs will remain strong, but their overall market volume will be constrained by the availability of novel, high-value treatments.
- Potential for Niche Indications: While unlikely to drive significant market growth, research into repurposing toremifene citrate for other hormone-sensitive conditions or specific patient subgroups could present minor opportunities. However, significant investment in such research is improbable given the drug's age and generic status.
Strategic Considerations for Stakeholders:
- Generic Manufacturers:
- Operational Efficiency: Focus on optimizing manufacturing costs, supply chain management, and ensuring consistent quality to remain competitive in a price-sensitive market.
- Market Access Optimization: Leverage existing relationships with distributors, wholesalers, and payers to ensure broad market availability and secure favorable pricing where possible.
- Product Portfolio Diversification: For companies with a broad generic portfolio, diversification into newer therapeutic areas or branded generics can mitigate reliance on mature products like toremifene citrate.
- Branded Innovators (Historical/Orion Pharma):
- Focus on Newer Innovations: The strategic imperative is to focus on the development and commercialization of next-generation oncology therapies rather than seeking to revitalize the market for an off-patent molecule.
- Healthcare Providers and Payers:
- Guideline Adherence: Continue to prescribe toremifene citrate according to established guidelines, balancing clinical need with cost-effectiveness.
- Monitor Emerging Therapies: Stay abreast of advancements in breast cancer treatment that may offer superior outcomes, influencing treatment pathways and potentially reducing reliance on older agents.
The market for toremifene citrate will continue to serve a specific patient population for the foreseeable future. However, its role is likely to diminish gradually as newer, more effective therapies become standard of care.
Key Takeaways
- Toremifene citrate is a mature, off-patent SERM primarily used for metastatic breast cancer in postmenopausal women.
- The market is dominated by generic manufacturers, leading to intense price-based competition and significant price erosion.
- Global revenue for toremifene citrate is estimated between $50 million and $80 million in 2024-2025, projected to decline to $40 million - $65 million by 2026-2028.
- Pricing is characterized by low wholesale costs, ranging from $15 to $40 for a 60-tablet bottle, with ongoing downward pressure.
- The regulatory landscape is well-established, with generic approvals via ANDAs in the U.S. and similar pathways globally.
- Market access is influenced by payer formularies and reimbursement policies, with toremifene citrate widely available on generic formularies.
- The future outlook indicates a stable but gradually declining market segment due to competition from newer, targeted oncology therapies.
- Strategic focus for generic manufacturers should be on operational efficiency and cost optimization, while branded innovators should prioritize newer drug development.
Frequently Asked Questions
- What is the primary indication for toremifene citrate? Toremifene citrate is primarily indicated for the treatment of metastatic breast cancer in postmenopausal women with estrogen receptor-positive or unknown breast cancer.
- Has toremifene citrate lost its patent protection? Yes, the original compound patents for toremifene citrate have long expired, allowing for the widespread manufacturing and sale of generic versions.
- What are the main competitors to toremifene citrate in the breast cancer market? Competitors include other selective estrogen receptor modulators (SERMs) like tamoxifen, aromatase inhibitors (AIs), and newer targeted therapies such as CDK4/6 inhibitors and antibody-drug conjugates (ADCs).
- Are there any significant new indications or formulations being developed for toremifene citrate? There is limited R&D investment in new indications or formulations for toremifene citrate due to its mature, off-patent status. The focus is on cost-effective manufacturing and supply.
- What is the projected impact of emerging breast cancer therapies on the toremifene citrate market? The increasing availability and adoption of newer, more effective targeted therapies and immunotherapies are expected to gradually reduce the market share and overall volume of older agents like toremifene citrate.
Citations
[1] World Health Organization. (2020). Breast cancer statistics. Retrieved from https://www.who.int/news-room/fact-sheets/detail/breast-cancer
More… ↓
