Share This Page
Drug Price Trends for TOLTERODINE TART ER
✉ Email this page to a colleague
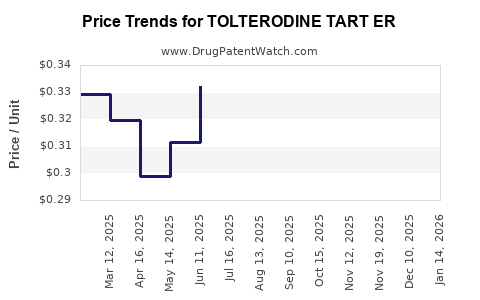
Average Pharmacy Cost for TOLTERODINE TART ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TOLTERODINE TART ER 2 MG CAP | 00093-7163-98 | 0.28211 | EACH | 2026-04-22 |
| TOLTERODINE TART ER 2 MG CAP | 00093-7163-56 | 0.28211 | EACH | 2026-04-22 |
| TOLTERODINE TART ER 4 MG CAP | 70436-0161-06 | 0.22854 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
TOLTERODINE TART ER Market Analysis and Financial Projection
What is Tolterodine Tart ER?
Tolterodine Tart ER (Extended Release) is a long-acting antimuscarinic agent used to treat overactive bladder with symptoms such as urgency, frequency, and incontinence. It functions by blocking muscarinic receptors in the bladder, reducing involuntary contractions. The extended-release formulation offers improved compliance through once-daily dosing.
Market Overview
Current Market Size
As of 2023, overactive bladder (OAB) medications, including Tolterodine Tart ER, generate approximately $3 billion globally. The segment shows consistent growth due to increased prevalence of OAB—estimated at 12-15% in adults over 40—and widening awareness.
Competitive Landscape
Main competitors include:
- Oxybutynin ER
- Solifenacin
- Darifenacin
- Trospium ER
- Mirabegron (a beta-3 adrenergic agonist)
Market shares (2023):
| Drug | Market Share (%) | Notes |
|---|---|---|
| Tolterodine Tart ER | 20 | Mature, established; now facing new entrants |
| Solifenacin | 25 | Leading driver in the segment |
| Mirabegron | 22 | Growing alternative, less anticholinergic effects |
| Others | 33 | Includes oxybutynin ER, darifenacin, trospium |
Key Market Drivers
- Aging populations
- Increased diagnosis rates
- Improved reimbursement policies
- Generic availability reducing costs in mature markets
Challenges
- Competition from newer agents (Mirabegron)
- Side-effect profiles affecting adherence
- Patent expirations potentially lowering prices
Pricing Analysis
Current Price Points
Branded Tolterodine Tart ER
- U.S.: Approximately $125-150 per month (retail)
- Europe: Similar range, with local variations
Generic equivalents
- U.S.: $35-50 per month
- Europe: Slightly lower, depending on local formulary arrangements
Pricing Trends
- Branded formulations maintain premium pricing
- Generics rapidly gained market share post-patent expiry
- Price erosion observed: Up to 75% reduction within 3 years of generic entry
Price Projections
Short-Term (Next 1-2 Years)
- Limited growth expected due to market saturation
- Price stabilization anticipated at current levels
- Generics likely to capture >80% market share
Mid to Long-Term (3-5 Years)
- Potential price declines for brand-name Tolterodine Tart ER to $100-120, driven by generic competition
- Possible stabilization of generic prices around $30-50/month
- Entry of biosimilars or new drugs could alter dynamics
Factors Influencing Prices
- Patent status: Patent expiration expected by 2025
- Regulatory policies: Price controls and value-based pricing models
- Market penetration of alternatives: Mirabegron and others gaining share
- Manufacturing costs: Advances in scale economies could reduce prices
Market Outlook and Opportunities
- Market Penetration: Despite competition, opportunities exist in underserved populations and emerging markets.
- Strategic Positioning: Differentiation through improved pharmacokinetics or combination therapies.
- Partnerships: Licensing and co-marketing agreements, particularly in regions with limited local manufacturing.
Key Takeaways
- The global OAB market exceeds $3 billion, with Tolterodine Tart ER accounting for about 20%.
- The current price in the U.S. is approximately $125-150 retail, with generics dropping to $35-50.
- Patent expiration by 2025 is likely to lead to significant price erosion.
- The market consolidates around newer agents like Mirabegron, impacting long-term pricing.
- Price projections suggest stabilized generic prices at $30-50 within 3-5 years, with branded prices declining proportionally.
FAQs
1. How does Tolterodine Tart ER compare to other OAB treatments?
It offers once-daily dosing and a well-established safety profile, but faces competition from newer drugs with fewer anticholinergic side effects.
2. When is patent expiration expected for Tolterodine Tart ER?
Patent expiration is projected around 2025, after which generic formulations are expected to dominate.
3. What are the major factors affecting drug pricing in this segment?
Patent status, competition from generics, regulatory policies, and adoption rates influence prices.
4. Which regions present key growth opportunities for Tolterodine Tart ER?
Emerging markets with increasing healthcare infrastructure and aging populations show potential for growth.
5. Will new therapies threaten Tolterodine Tart ER’s market share?
Yes, especially drugs like Mirabegron, which have different mechanisms and side effect profiles, are expected to capture a significant share over time.
References
[1] Market Research Future. "Overactive Bladder Market Analysis." 2023.
[2] EvaluatePharma. "Top Selling Drugs & Market Share." 2023.
[3] IQVIA. "Healthcare Data and Price Trends." 2023.
[4] FDA. "Patent and Exclusivity Data for Tolterodine." 2023.
More… ↓
