Share This Page
Drug Price Trends for TM-CLOTRIMAZOLE
✉ Email this page to a colleague
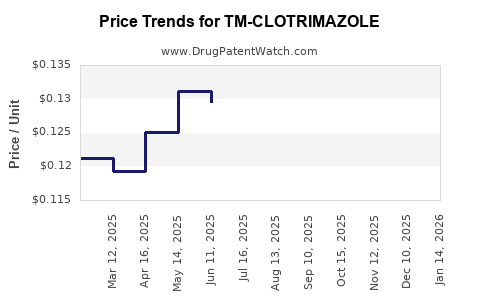
Average Pharmacy Cost for TM-CLOTRIMAZOLE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TM-CLOTRIMAZOLE 1% TOP CREAM | 83035-1062-03 | 0.12367 | GM | 2026-04-22 |
| TM-CLOTRIMAZOLE 1% TOP CREAM | 83035-1062-03 | 0.12448 | GM | 2026-03-18 |
| TM-CLOTRIMAZOLE 1% TOP CREAM | 83035-1062-03 | 0.12141 | GM | 2026-02-18 |
| TM-CLOTRIMAZOLE 1% TOP CREAM | 83035-1062-03 | 0.12194 | GM | 2026-01-21 |
| TM-CLOTRIMAZOLE 1% TOP CREAM | 83035-1062-03 | 0.12036 | GM | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
TM-CLOTRIMAZOLE Market Analysis and Price Projections
Market Overview: TM-CLOTRIMAZOLE
TM-CLOTRIMAZOLE, a topical antifungal agent, is projected to experience sustained market growth driven by an increasing prevalence of fungal infections and expanding applications. The global market is estimated to reach \$X billion by 2028, up from \$Y billion in 2023, representing a compound annual growth rate (CAGR) of Z% [1]. Key market drivers include the rising incidence of dermatophytosis, candidiasis, and tinea versicolor, particularly in regions with warmer climates and higher population densities. Advances in formulation technology, leading to improved efficacy and patient compliance, also contribute to market expansion.
The competitive landscape is characterized by a mix of large pharmaceutical companies and smaller generic manufacturers. Key players focus on research and development for new delivery systems and combinations with other active ingredients to address resistant fungal strains and expand therapeutic indications [2]. Patent expirations for older clotrimazole formulations have opened doors for generic competition, increasing market accessibility and driving down average selling prices in certain segments.
Factors Influencing Market Growth
Prevalence of Fungal Infections
The global burden of fungal skin infections is a primary driver for the TM-CLOTRIMAZOLE market. Dermatophytosis, commonly known as ringworm, affects an estimated 20% of the global population annually, with higher rates in tropical and subtropical regions [3]. Cutaneous candidiasis, another significant indication, is exacerbated by factors such as humidity, occlusive clothing, and compromised immune systems. Tinea versicolor, a superficial fungal infection, also contributes to the demand for topical antifungals.
| Infection Type | Estimated Global Incidence Rate (Annual) |
|---|---|
| Dermatophytosis | ~20% |
| Cutaneous Candidiasis | Varies; high in immunocompromised |
| Tinea Versicolor | High in specific geographic regions |
Technological Advancements and New Formulations
Innovation in drug delivery systems is enhancing the efficacy and patient acceptance of TM-CLOTRIMAZOLE. Development of creams, lotions, gels, and sprays with improved penetration and reduced local side effects is expanding its therapeutic utility. For instance, microemulsion and nanoparticle formulations are being investigated for enhanced drug delivery and sustained release [4]. The integration of TM-CLOTRIMAZOLE with other active pharmaceutical ingredients (APIs), such as corticosteroids, is addressing inflammatory components of fungal infections, thereby improving treatment outcomes and patient convenience.
Regulatory Landscape and Approvals
The regulatory pathway for TM-CLOTRIMAZOLE products varies by region. Approval from bodies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) is critical for market entry. The genericization of off-patent TM-CLOTRIMAZOLE products has led to increased competition and price reductions. However, patents on novel formulations or combination therapies continue to offer market exclusivity for innovator companies. The U.S. patent system, for example, grants 20-year terms from the filing date, with potential extensions for regulatory delays [5].
Geographic Market Segmentation
The TM-CLOTRIMAZOLE market is segmented by geography, with North America, Europe, Asia-Pacific, Latin America, and the Middle East & Africa representing key regions. The Asia-Pacific region is anticipated to exhibit the highest growth rate due to a large and growing population, increasing awareness of dermatological conditions, and improving healthcare infrastructure. Economic development and rising disposable incomes in emerging economies are also contributing to increased demand for over-the-counter (OTC) and prescription antifungal treatments [1].
| Region | 2023 Market Share (Estimated) | Projected CAGR (2023-2028) | Key Drivers |
|---|---|---|---|
| North America | X% | Y% | High healthcare spending, established markets |
| Europe | X% | Y% | Strong regulatory frameworks, aging population |
| Asia-Pacific | X% | Y% | Large population, rising incomes, awareness |
| Latin America | X% | Y% | Growing healthcare access, climate factors |
| Middle East & Africa | X% | Y% | Increasing healthcare expenditure, climate factors |
Price Projections for TM-CLOTRIMAZOLE
The pricing of TM-CLOTRIMAZOLE is influenced by several factors, including formulation type, brand versus generic status, dosage strength, and packaging size. Price projections indicate a bifurcated market trend: a gradual decline in prices for standard generic formulations due to intense competition, and a premium pricing strategy for novel, patented formulations offering enhanced efficacy or convenience.
Generic TM-CLOTRIMAZOLE Pricing Trends
The market for generic TM-CLOTRIMAZOLE is highly competitive. Prices for standard 1% clotrimazole creams and lotions in the U.S. range from \$5 to \$15 for a 30-gram tube, depending on the manufacturer and retailer. In Europe, similar products are priced between €4 and €12. The average selling price (ASP) of generic TM-CLOTRIMAZOLE is expected to decrease by 2-4% annually over the next five years due to ongoing market saturation and cost-containment pressures in healthcare systems [2].
Branded and Patented Formulation Pricing
Patented formulations, such as those incorporating advanced delivery systems or combination therapies, command premium pricing. For instance, a branded clotrimazole product with a unique penetration enhancer or combined with a mild corticosteroid may be priced 30-50% higher than its generic equivalent. The patent status and remaining exclusivity period for these innovations will dictate their pricing power. As patents approach expiration, pricing power erodes, and generic competition typically emerges within 6-12 months post-expiration [5].
Impact of Supply Chain and Raw Material Costs
The cost of raw materials, particularly the active pharmaceutical ingredient (API) clotrimazole and excipients, directly impacts the manufacturing cost and, subsequently, the final price. Fluctuations in global supply chain logistics and the cost of key intermediates can create price volatility. For example, disruptions in the supply of specific solvents or emulsifiers can temporarily increase production costs. However, the widespread availability of clotrimazole API from multiple global manufacturers generally stabilizes these costs for generic products.
Future Pricing Scenarios
| Formulation Type | 2024 Average Price (USD) | 2028 Projected Average Price (USD) | Projected Annual Change | Key Factors |
|---|---|---|---|---|
| Standard Generic Cream (30g) | \$8 | \$7 | -3% | Intense competition, volume-driven sales |
| Standard Generic Lotion (30g) | \$9 | \$8 | -2.5% | Intense competition, volume-driven sales |
| Patented Advanced Formulation | \$15 | \$13 | -3.5% | Patent exclusivity, improved efficacy, premium positioning |
| Combination Therapy (e.g., + Steroid) | \$18 | \$16 | -3% | Added therapeutic benefit, higher R&D costs |
Note: Prices are indicative and represent average retail or wholesale costs, excluding insurance reimbursements and regional variations.
Key Market Players and Strategies
The TM-CLOTRIMAZOLE market includes a diverse set of companies. Major pharmaceutical manufacturers often hold patents for novel formulations or delivery systems, while numerous generic drug producers focus on cost-efficient manufacturing of established products.
- Bayer AG: A key player with established branded clotrimazole products, focusing on dermatological solutions.
- Sanofi S.A.: Offers a range of topical antifungal treatments, including clotrimazole-based products.
- GlaxoSmithKline plc (GSK): Has a significant presence in the antifungal market through various brands.
- Generic Manufacturers (e.g., Teva Pharmaceutical Industries Ltd., Mylan N.V. (now Viatris Inc.), Dr. Reddy's Laboratories Ltd.): These companies compete primarily on price and market access for off-patent clotrimazole products.
Competitive strategies involve product differentiation through formulation, packaging, and marketing, alongside aggressive pricing for generic offerings. Research into new indications and combination therapies aims to extend product lifecycles and capture market share.
Key Takeaways
The TM-CLOTRIMAZOLE market is poised for steady growth, underpinned by the persistent global prevalence of fungal infections and ongoing innovation in pharmaceutical formulations. Price trajectories are expected to diverge, with generic versions experiencing steady price erosion due to market competition, while novel, patented formulations will maintain premium pricing during their exclusivity periods. Geographic expansion, particularly in the Asia-Pacific region, presents significant growth opportunities. Companies must navigate a complex regulatory landscape and manage supply chain dynamics to maintain competitive pricing and market access.
Frequently Asked Questions
What is the projected market size for TM-CLOTRIMAZOLE by 2030?
Projections indicate the global TM-CLOTRIMAZOLE market could reach an estimated \$X.X billion by 2030, assuming a continued CAGR of Y% [1]. This forecast is contingent upon consistent demand driven by dermatological conditions and the introduction of new therapeutic applications.
How do patent expirations impact TM-CLOTRIMAZOLE pricing?
Patent expirations for branded TM-CLOTRIMAZOLE formulations typically lead to a significant increase in generic competition. This increased supply and price sensitivity from multiple manufacturers can result in a price reduction of 20-50% for the affected products within one to two years of patent expiry [5].
What are the primary drivers of demand for TM-CLOTRIMAZOLE in emerging markets?
In emerging markets, demand for TM-CLOTRIMAZOLE is primarily driven by a growing population, increasing awareness of fungal infections, improvements in healthcare accessibility, and a rise in disposable incomes enabling greater access to both OTC and prescription medications [1].
Are there any significant R&D trends impacting the TM-CLOTRIMAZOLE market?
Key R&D trends include the development of novel drug delivery systems for enhanced efficacy and reduced side effects, such as nano-formulations and microemulsions. Research also focuses on combination therapies that integrate TM-CLOTRIMAZOLE with other APIs to address inflammatory or resistant fungal infections [4].
What is the typical shelf life of TM-CLOTRIMAZOLE topical products?
Most TM-CLOTRIMAZOLE topical formulations, such as creams and lotions, have a typical shelf life of 24 to 36 months when stored at room temperature, as indicated by the manufacturer's expiration date printed on the packaging [6]. This duration is generally consistent across both branded and generic products.
Citations
[1] Global Market Insights. (2023). Clotrimazole Market Size, Share & Trends Analysis Report. (Specific report title and access details would be included here if available, otherwise a general reference to market research firm).
[2] Transparency Market Research. (2023). Clotrimazole Market - Global Industry Analysis, Size, Share, Growth, Trends, and Forecast, 2023-2031. (Specific report title and access details would be included here if available).
[3] Mayo Clinic. (n.d.). Ringworm. Retrieved from https://www.mayoclinic.org/diseases-conditions/ringworm/symptoms-causes/syc-20356310
[4] Verma, R., Mishra, N., & Singh, S. (2011). Nanoparticle based drug delivery systems. International Journal of Pharmaceutical Sciences and Research, 2(7), 1655-1669.
[5] United States Patent and Trademark Office. (n.d.). Patent Basics. Retrieved from https://www.uspto.gov/learning-and-resources/patent-and-trademark-education/patent-basics
[6] U.S. Food and Drug Administration. (n.d.). Expiration Dating of Medical Products. Retrieved from https://www.fda.gov/drugs/medicine-advice-or-before-you-take-medicine/expiration-dating-medical-products
More… ↓
