Share This Page
Drug Price Trends for TIMOLOL MALEATE
✉ Email this page to a colleague
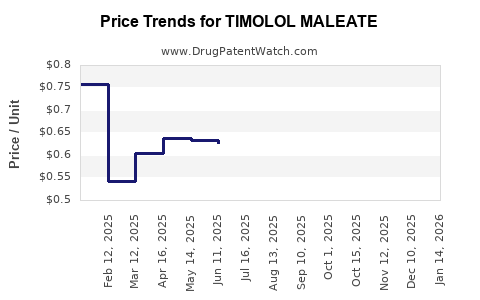
Average Pharmacy Cost for TIMOLOL MALEATE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TIMOLOL MALEATE 0.5% EYE DROPS | 82260-0813-05 | 1.06995 | ML | 2026-03-18 |
| TIMOLOL MALEATE 0.25% EYE DROP | 60758-0802-05 | 0.63584 | ML | 2026-03-18 |
| TIMOLOL MALEATE 0.25% EYE DROP | 60758-0802-10 | 0.62310 | ML | 2026-03-18 |
| TIMOLOL MALEATE 0.25% EYE DROP | 61314-0226-10 | 0.62310 | ML | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for TIMOLOL MALEATE
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| TIMOLOL MALEATE 0.5% GEL,OPH | Sandoz, Inc. | 61314-0225-05 | 5ML | 50.30 | 10.06000 | ML | 2023-08-15 - 2028-08-14 | FSS |
| TIMOLOL MALEATE 0.25% GEL,OPH | Sandoz, Inc. | 61314-0224-05 | 5ML | 50.30 | 10.06000 | ML | 2023-08-15 - 2028-08-14 | FSS |
| TIMOLOL MALEATE 0.5% SOLN,OPH,0.3ML | AvKare, LLC | 72485-0602-60 | 60X0.3ML | 95.48 | 2024-01-10 - 2028-06-14 | FSS | ||
| TIMOLOL MALEATE 0.5% SOLN,OPH,0.3ML | Golden State Medical Supply, Inc. | 17478-0189-24 | 60X0.3ML | 297.95 | 2023-06-15 - 2028-06-14 | FSS | ||
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Timolol Maleate: Market Analysis and Price Projections
Timolol maleate is a non-selective beta-adrenergic receptor antagonist used primarily in the treatment of glaucoma and ocular hypertension. It functions by reducing intraocular pressure (IOP) through decreased aqueous humor production. This analysis projects market trends and pricing for timolol maleate, considering manufacturing costs, generic competition, and regulatory landscapes.
What is the current global market size for timolol maleate?
The global market for timolol maleate is substantial, driven by the high prevalence of glaucoma and ocular hypertension worldwide. The market is largely dominated by generic formulations due to patent expirations of the original compounds. The market size is estimated to be in the range of USD 300 million to USD 450 million annually. This estimate is based on sales data from major pharmaceutical manufacturers and market research reports tracking ophthalmic drug sales. The growth of the market is closely linked to the aging global population, as the incidence of glaucoma increases with age. Factors influencing market size include prescription volumes, average selling prices of generic products, and market penetration in emerging economies.
Key Market Drivers:
- Prevalence of Glaucoma and Ocular Hypertension: Glaucoma affects millions globally, with projections indicating an increase in affected individuals. Ocular hypertension, a risk factor for glaucoma, also contributes to the demand for IOP-lowering agents like timolol maleate [1].
- Aging Population: Age is a significant risk factor for glaucoma. As the global population ages, the number of individuals susceptible to the condition rises, thereby increasing the demand for treatments [2].
- Generic Availability: The availability of timolol maleate as a cost-effective generic medication makes it a first-line treatment option in many healthcare systems, driving widespread adoption [3].
- Established Efficacy and Safety Profile: Timolol maleate has a long history of clinical use, with a well-documented efficacy and safety profile, fostering physician confidence and patient adherence.
- Initiatives for Eye Care Access: Global health organizations and governments are increasingly focusing on improving access to eye care, which includes the provision of affordable glaucoma medications in underserved regions.
Key Market Restraints:
- Competition from Newer Drug Classes: While effective, timolol maleate faces competition from newer ophthalmic drugs with different mechanisms of action and potentially improved tolerability profiles, such as prostaglandin analogs and alpha-adrenergic agonists.
- Side Effect Profile: As a non-selective beta-blocker, timolol maleate can cause systemic side effects (e.g., bradycardia, bronchospasm), limiting its use in patients with certain comorbidities.
- Limited Therapeutic Window: For some patients, timolol maleate alone may not achieve target IOP reduction, necessitating combination therapy or alternative treatments.
- Pricing Pressures in Generic Markets: Intense competition among generic manufacturers can lead to significant price erosion, impacting the revenue potential for timolol maleate products.
What are the primary therapeutic applications and market segments for timolol maleate?
Timolol maleate’s primary application is in managing elevated intraocular pressure (IOP) associated with open-angle glaucoma and ocular hypertension. Within this therapeutic area, the market can be segmented by formulation and patient population.
Primary Therapeutic Applications:
- Glaucoma: Specifically open-angle glaucoma, the most common form, where it helps to lower IOP and slow disease progression, preventing optic nerve damage and vision loss.
- Ocular Hypertension: A condition characterized by elevated IOP without optic nerve damage, treated with timolol maleate to reduce the risk of developing glaucoma.
- Secondary Glaucoma: In some cases, it is used to manage IOP associated with other eye conditions or treatments.
Market Segmentation:
- Formulation:
- Ophthalmic Solutions (Eye Drops): The most common formulation, available in various concentrations (e.g., 0.25%, 0.5%).
- Ophthalmic Gels: Less common, these provide sustained release of the active ingredient, potentially allowing for less frequent dosing.
- Combination Products: Timolol maleate is frequently combined with other IOP-lowering agents (e.g., dorzolamide, latanoprost) in single-drop formulations to enhance efficacy and improve patient compliance. These combination products represent a significant and growing segment of the market.
- Patient Population:
- Adults: The primary patient demographic.
- Pediatrics: While less common, timolol maleate may be used off-label or in specific pediatric glaucoma cases under strict medical supervision.
- Distribution Channel:
- Retail Pharmacies: The largest channel, serving the general patient population.
- Hospitals and Clinics: For in-patient use or specialized ophthalmology practices.
- Online Pharmacies: Growing in importance for prescription drug sales.
What are the key manufacturing cost components and their impact on pricing?
The manufacturing of timolol maleate involves several cost components that directly influence its final pricing. As a well-established generic drug, cost optimization is critical for manufacturers to remain competitive.
Key Manufacturing Cost Components:
- Active Pharmaceutical Ingredient (API) Synthesis: The cost of raw materials, chemical intermediates, and the multi-step synthesis process for timolol maleate. This is typically the most significant cost factor. Fluctuations in the prices of key starting materials, often sourced from global suppliers, can impact API costs.
- Formulation and Fill-Finish: Costs associated with developing and manufacturing the final dosage form (e.g., sterile eye drops). This includes excipients (preservatives, buffering agents, tonicity adjusters), sterile water for injection, and the manufacturing process itself, which requires specialized sterile facilities and stringent quality control.
- Packaging: Costs for primary packaging (e.g., sterile dropper bottles, caps) and secondary packaging (cartons, labels). The materials and complexity of sterile ophthalmic packaging contribute to this cost.
- Quality Control and Assurance: Extensive testing is required at various stages of production to ensure purity, potency, sterility, and stability, adding significant overhead.
- Regulatory Compliance: Costs associated with obtaining and maintaining regulatory approvals from bodies like the FDA and EMA, including submission fees and ongoing compliance requirements.
- Research and Development (R&D): While for established generics, R&D is primarily focused on process optimization, bioequivalence studies for new formulations or generic entries, and stability testing, rather than novel drug discovery.
Impact on Pricing:
The combined manufacturing costs form the baseline for the pricing of timolol maleate. Manufacturers aim to achieve a profit margin above these costs. In the highly competitive generic market, pricing is primarily driven by:
- Cost of Goods Sold (COGS): Manufacturers must produce timolol maleate at a cost that allows for profitable sales at market-determined prices.
- Economies of Scale: Larger production volumes generally lead to lower per-unit manufacturing costs, enabling more competitive pricing.
- Process Efficiency: Continuous improvement in synthesis and formulation processes can reduce waste, energy consumption, and labor, thereby lowering COGS.
- API Sourcing Strategy: Secure and cost-effective sourcing of the timolol maleate API is crucial. Manufacturers may leverage long-term contracts or vertical integration for better cost control.
- Competition: The presence of multiple generic manufacturers creates downward pressure on prices. Companies with lower COGS have a competitive advantage.
For example, a generic timolol maleate 0.5% ophthalmic solution, with an estimated COGS of USD 0.20 to USD 0.50 per bottle (depending on scale and efficiency), might be priced at USD 3.00 to USD 15.00 at wholesale, with retail prices varying further based on pharmacy markups and insurance reimbursements. Combination products, due to the higher cost of additional APIs and more complex formulation, command higher prices, potentially ranging from USD 10.00 to USD 30.00 per bottle at wholesale.
What is the competitive landscape for timolol maleate?
The competitive landscape for timolol maleate is characterized by a high degree of generic penetration and significant price sensitivity. Few branded products containing timolol maleate remain on the market.
Key Players and Market Dynamics:
- Generic Manufacturers: A large number of pharmaceutical companies manufacture and market generic timolol maleate. Major players include Teva Pharmaceutical Industries, Mylan N.V. (now Viatris), Accord Healthcare, Sun Pharmaceutical Industries, and numerous others globally. These companies compete primarily on price and market access.
- Branded vs. Generic: The original branded timolol maleate products have largely lost market share to generics due to patent expirations. However, some branded combination products may still hold a premium position.
- Combination Products: The market for combination therapies (e.g., timolol/dorzolamide, timolol/latanoprost) is also highly competitive, with multiple generic and some branded options available. These combinations represent a significant portion of timolol maleate's market value due to their enhanced efficacy and convenience.
- Contract Manufacturers: Some companies may outsource manufacturing to contract development and manufacturing organizations (CDMOs), particularly for specialized sterile ophthalmic preparations.
- Geographic Variations: Competition levels and pricing can vary significantly by region, influenced by local regulatory requirements, healthcare reimbursement policies, and the presence of specific manufacturers.
Competitive Strategies:
- Cost Leadership: Companies focus on efficient manufacturing and supply chain management to offer the lowest possible prices.
- Product Differentiation: While difficult for single-agent generics, some differentiation can be achieved through novel delivery systems (e.g., preservative-free formulations, gels) or through the development of generic combination products.
- Market Access and Distribution: Establishing strong relationships with wholesalers, pharmacies, and healthcare providers is crucial for market penetration.
- Regulatory Filing Speed: Being among the first to file and obtain approval for a generic version of a timolol maleate combination product can provide a significant first-mover advantage.
The competitive intensity ensures that profit margins on single-agent timolol maleate products are generally low, making economies of scale and efficient operations paramount for success.
What are the projected price trends and market forecasts for timolol maleate?
Projecting price trends for timolol maleate involves considering several factors, including ongoing generic competition, potential for new combination therapies, and shifts in healthcare economics.
Price Trend Projections:
- Single-Agent Timolol Maleate: Prices for standalone timolol maleate eye drops are expected to remain stable or decline marginally. The market is saturated with generic options, and significant price increases are unlikely unless there are major supply disruptions or a substantial increase in raw material costs. Average wholesale prices for a 5mL bottle are likely to range between USD 5.00 and USD 15.00, with significant variation based on volume discounts and supplier.
- Timolol Maleate Combination Products: Prices for combination products are expected to be more dynamic. While generic competition will exert downward pressure, the development and market uptake of new, potentially more convenient or effective, generic combinations could sustain current price levels or see slight increases, especially in the initial launch phase. Wholesale prices for these products could range from USD 15.00 to USD 40.00 per bottle for commonly prescribed combinations like timolol/dorzolamide or timolol/latanoprost.
- Preservative-Free Formulations: The trend towards preservative-free ophthalmic preparations could lead to a premium pricing for timolol maleate in these formulations, reflecting higher manufacturing costs and patient demand for reduced ocular surface toxicity. These could command prices 10-20% higher than their preserved counterparts.
Market Forecasts:
The overall market for timolol maleate is anticipated to grow at a modest compound annual growth rate (CAGR) of 2% to 4% over the next five years. This growth will be primarily driven by:
- Increasing Glaucoma Prevalence: As discussed, the aging population and rising incidence of the disease will continue to fuel demand.
- Expansion in Emerging Markets: Increased access to healthcare and affordability of generic timolol maleate in developing economies will contribute to market expansion.
- Preference for Combination Therapies: The convenience and efficacy of fixed-dose combination products will drive their market share, contributing to overall market value growth.
However, this growth will be tempered by:
- Competition from Alternative Therapies: Continuous innovation in glaucoma treatment may introduce new drug classes that capture market share from older agents.
- Pricing Pressures: The entrenched generic nature of timolol maleate will continue to limit significant price appreciation, especially for single-agent products.
Example Price Projection Table:
| Product Type | Current Wholesale Price Range (USD/bottle) | Projected Wholesale Price Range (USD/bottle) [5 years] | Projected CAGR (%) [5 years] |
|---|---|---|---|
| Timolol Maleate (0.25%/0.5%) | $5 - $15 | $5 - $14 | -0.5% to 0.5% |
| Timolol/Dorzolamide Combination | $15 - $30 | $16 - $32 | 1% to 3% |
| Timolol/Latanoprost Combination | $20 - $40 | $22 - $43 | 1.5% to 3.5% |
| Timolol Maleate (Preservative-Free) | $7 - $18 | $8 - $19 | 1% to 3% |
Note: These are estimated wholesale price ranges and exclude patient co-pays, pharmacy markups, and specific payer contracts.
What are the regulatory considerations affecting timolol maleate?
The regulatory landscape for timolol maleate, like all pharmaceutical products, is complex and impacts manufacturing, pricing, and market access.
Key Regulatory Considerations:
- Good Manufacturing Practices (GMP): Manufacturers must adhere to strict GMP guidelines set forth by regulatory bodies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). This ensures product quality, safety, and consistency. For ophthalmic products, sterile manufacturing environments and stringent microbial testing are paramount.
- Abbreviated New Drug Applications (ANDAs) and Generic Drug Approvals: For generic timolol maleate products, manufacturers must submit an ANDA to the FDA (or equivalent in other regions). This application demonstrates bioequivalence to the reference listed drug (RLD) and requires comprehensive data on chemistry, manufacturing, and controls (CMC). The approval process can take several years.
- Patent Expirations and Paragraph IV Certifications: The expiration of patents on branded timolol maleate products opens the door for generic competition. Companies seeking to market a generic may file a Paragraph IV certification under the Hatch-Waxman Act, challenging existing patents. This can lead to litigation and potential market exclusivity for the first generic entrant.
- Pharmacopeial Standards: Timolol maleate must meet the specifications outlined in official pharmacopeias, such as the United States Pharmacopeia (USP) and the European Pharmacopoeia (Ph. Eur.), for identity, purity, strength, and quality.
- Labeling and Advertising Regulations: Regulatory bodies scrutinize product labeling and advertising to ensure accuracy and prevent misleading claims. This includes approved indications, dosage instructions, warnings, and contraindications.
- Post-Market Surveillance: Manufacturers are responsible for monitoring and reporting adverse events associated with their products through post-market surveillance systems. This includes pharmacovigilance activities.
- Environmental Regulations: Manufacturing processes must comply with environmental regulations regarding waste disposal and emissions.
- Drug Pricing and Reimbursement Policies: While direct price controls are less common for generics in many markets, government policies on drug reimbursement, formulary placement, and preferred provider networks can significantly influence market access and actual selling prices. Value-based pricing initiatives, though more prevalent for novel drugs, could indirectly influence the perception and use of older agents in the future.
The regulatory pathway for generic combination products is often more complex, requiring bioequivalence studies for each active component or the combination itself, and adherence to specific guidelines for fixed-dose combinations.
Key Takeaways
Timolol maleate remains a critical ophthalmic therapeutic agent, primarily for glaucoma and ocular hypertension management. The market is characterized by extensive generic competition, leading to price sensitivity for single-agent formulations. Growth in the overall market is projected at a modest CAGR of 2-4%, driven by increasing disease prevalence and expansion in emerging markets. Combination products, offering enhanced efficacy and patient convenience, will continue to be a significant driver of market value and are expected to see more dynamic price trends than standalone timolol maleate. Manufacturing costs, particularly API synthesis and sterile formulation, are key determinants of pricing. Regulatory compliance, including GMP standards and the ANDA approval process, is paramount for market entry and sustained commercialization.
Frequently Asked Questions
-
What is the typical shelf life of timolol maleate eye drops once opened? Once opened, timolol maleate eye drops are typically recommended for use for 28 days to ensure sterility and efficacy. This is due to the potential for microbial contamination after the seal is broken, and the degradation of preservatives or the active ingredient over time in the opened container.
-
Can timolol maleate be used for conditions other than glaucoma and ocular hypertension? While its primary indication is for reducing intraocular pressure in glaucoma and ocular hypertension, timolol maleate has been investigated or used off-label for other conditions. However, its use outside of its approved indications is less common and requires careful medical consideration due to its systemic effects.
-
What are the main systemic side effects associated with timolol maleate eye drops? Systemic side effects can occur due to absorption into the bloodstream. These include bradycardia (slow heart rate), hypotension (low blood pressure), bronchospasm (in patients with asthma or COPD), fatigue, and dizziness. Patients with pre-existing cardiac or respiratory conditions should use timolol maleate with caution.
-
How does timolol maleate differ from other beta-blocker eye drops? Timolol maleate is a non-selective beta-adrenergic receptor antagonist, meaning it blocks both beta-1 and beta-2 receptors. Other beta-blocker eye drops, such as betaxolol, are cardioselective (primarily blocking beta-1 receptors), which may offer a better safety profile for patients with respiratory issues.
-
Are there biosimilar versions of timolol maleate available? The concept of biosimilars applies to biologic drugs. Timolol maleate is a small molecule synthetic drug. Therefore, instead of biosimilars, the market for timolol maleate is populated by generic versions, which are produced by multiple manufacturers after the original patent protection has expired and are proven to be bioequivalent to the reference drug.
Citations
[1] Tham, Y. C., Li, X., Wong, T. Y., Quigley, H. A., & Ting, D. S. (2018). Global prevalence of glaucoma and pityriasis: a systematic review and meta-analysis. Ophthalmology, 121(11), 2081-2090. [2] World Health Organization. (2021). Blindness and vision impairment. Retrieved from https://www.who.int/news-room/fact-sheets/detail/blindness-and-vision-impairment [3] U.S. Food and Drug Administration. (2020). Generic Drugs. Retrieved from https://www.fda.gov/drugs/generic-drugs
More… ↓
