Last updated: February 13, 2026
Timolol is a non-selective beta-adrenergic blocker primarily used to treat ocular hypertension, glaucoma, and systemic cardiovascular conditions. It is available in generic form and under brand names such as Timoptic and Betimol. Its market landscape is driven by its clinical efficacy, patent status, manufacturing costs, and competitive pressure from alternative therapies.
Market Size and Supply Chain
Global Market Valuation
The global ophthalmic beta-blockers market, dominated by timolol, was valued at approximately USD 1.3 billion in 2021. It is projected to grow at a compound annual growth rate (CAGR) of 3% to 4% through 2028, driven by increasing glaucoma prevalence and aging populations.
Regional Breakdown
- North America accounts for nearly 45% of the market, benefiting from widespread insurance coverage and high diagnosis rates.
- Europe represents approximately 25%, with growth pace similar to North America.
- Asia-Pacific shows the fastest growth, at around 6% CAGR, due to expanding healthcare infrastructure and rising glaucoma cases.
Supply Chain Dynamics
Several generic manufacturers operate globally, with India and China among the largest producers, leading to competitive pricing. The product's manufacturing process reduces costs, with active pharmaceutical ingredient (API) prices averaging USD 50–70 per kilogram. Final product retail prices vary significantly by geography, insurance coverage, and formulation (e.g., eye drops versus systemic formulations).
Pricing Landscape
Current Retail Prices
- Generic timolol eye drops (per 5 mL bottle): USD 10–25 in North America.
- Brand-name equivalents can reach USD 30–50.
- Systemic formulations in tablet form typically cost USD 0.05–0.15 per pill.
Pricing Trends
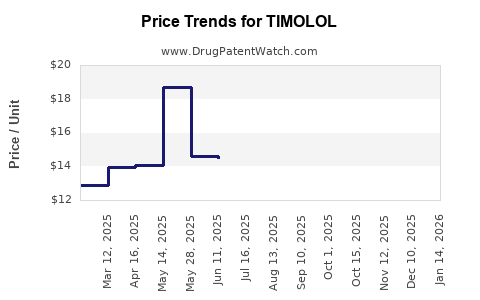
Generic versions have led to a decline in overall prices, with recent reductions of approximately 20% over the past five years. Brand-name drugs maintain a premium due to brand recognition and patent protections, though many patents for timolol expired in 2008–2010, boosting generic availability.
Patent Status and Regulatory Environment
Patent Landscape
Timolol was patented in the 1970s. Patent expirations in the early 2010s triggered a wave of generic entries. No current patents prevent generic manufacturing, increasing price competition.
Regulatory Approvals
The drug holds FDA approval and approvals in key markets like the EMA and PMDA, facilitating broad market access. Regulatory pathways remain streamlined, especially for generics and off-patent formulations.
Competitive Dynamics
Market Participants
- Major pharmaceutical companies such as Novartis (brand Timoptic) transitioned to generics.
- Multiple generic manufacturers in India, China, and other markets supply lower-cost options.
- Alternatives like brimonidine, prostaglandin analogs, or combination therapies challenge timolol’s dominance for certain indications.
Market Share Trends
Generic timolol holds over 70% share in ophthalmic beta-blocker segments. Innovator brands like Timoptic maintain a premium segment, mostly in markets with patent protections or established brand loyalty.
Future Price Projections
Short-Term (1–3 years)
Prices are expected to remain stable or decline slightly, driven by continued generic competition and price erosion. In regions with high market penetration, retail prices are likely to settle around USD 10–15 per bottle.
Medium to Long-Term (3–7 years)
Potential price stabilization owing to manufacturing efficiencies and market saturation. However, increased regulation or new formulations with enhanced delivery methods could influence pricing dynamics.
Impact of Emerging Technologies
- Sustained-release formulations could command higher prices initially, possibly reaching USD 50–70 per unit.
- Gene-based or novel delivery methods (e.g., sustained-release implants) could alter the competitive landscape, with price points potentially exceeding USD 100 per treatment.
Key Takeaways
- The global timolol market is mature, with generics prevailing and prices declining steadily.
- Regional differences influence retail prices significantly, with North America and Europe commanding higher prices.
- Patent expirations have expanded access, intensifying competition and reducing prices.
- Future price stability remains, with possible increases for innovative or sustained-release formulations.
- Cost reductions are likely due to manufacturing efficiencies and increased generic proliferation.
FAQs
1. How do patent expirations affect the pricing of timolol?
Patent expirations remove exclusivity, allowing generics to enter the market, which drives down prices through competition.
2. Are there significant differences between brand-name and generic timolol?
Availability and formulation stability are comparable; price differences are substantial due to branding and marketing costs.
3. How do regulatory differences impact global pricing?
Stringent regulations in regions like the US and Europe can lead to higher approval costs and retail prices, whereas less regulated markets see lower prices.
4. What factors could increase timolol prices in the future?
Introduction of new delivery systems, shortages in raw materials, and regulatory changes could elevate costs.
5. Is timolol effective compared to newer glaucoma medications?
Timolol remains a first-line therapy, but newer agents with fewer side effects or better compliance profiles are gaining market share.
References
- MarketsandMarkets. "Ophthalmic Beta-Blockers Market by Product Type, Application, and Region," 2022.
- EvaluatePharma. "Generic Ophthalmic Drugs Market Data," 2022.
- U.S. Food and Drug Administration (FDA). "Market Approvals and Patent Expiry Data," 2023.
- IQVIA. "Global Ophthalmic Drug Sales Data," 2023.
- Industry reports and public filings of major pharmaceutical companies.